Abstract
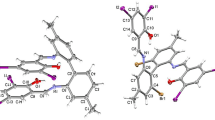
An organic ligand 1-(1H-benzimidazol-1-ylmethyl)-1H-benzotriazole (bta) and two zinc complexes of the composition [Zn(bta)2Hal2] (Hal = Cl, Br) were synthesized. The crystal structure of the complex [Zn(bta)2Cl2] was determined and its features were discussed. According to X-ray diffraction data, the central atom has a tetrahedral environment composed of two nitrogen atoms from two ligand molecules coordinated in monodentate mode and two terminal chloride ions. Density functional theory (DFT) calculations of the ligand and complex [Zn(bta)2Cl2] were carried out. A study of the luminescence properties of the compounds synthesized suggests that excitation of the zinc complexes gives rise to a dual-band luminescence similar to that of the free ligand. Most probably, the emission originates from π—π* and π—π* intra-ligand transitions.
Similar content being viewed by others
References
I. Briguglio, S. Piras, P. Corona, E. Gavini, M. Nieddu, G. Boatto, A. Carta, Eur. J. Med. Chem., 2015, 97, 612.
M. Alpa, H. Göker, R. Brun, S. Yıldız, Eur. J. Med. Chem., 2009, 44, 2002.
S. Sharma, S. Gangal, A. Rauf, Eur. J. Med. Chem., 2009, 44, 1751.
H. T. Abdel-Mohsen, F. A. F. Ragab, M. M. Ramla, H. I. El-Diwani, Eur. J. Med. Chem., 2010, 45, 2336.
K. Starčević, M. Kralj, K. Ester, I. Sabol, M. Grce, K. Pavelić, G. Karminski-Zamola, Bioorg. Med. Chem., 2007, 15, 4419.
K. F. Ansari, C. Lal, Eur. J. Med. Chem., 2009, 44, 4028.
A. Popova, M. Christov, S. Raicheva, E. Sokolova, Corros. Sci., 2004, 46, 1333.
G. Laguzzi, L. Luvidi, Surf. Coat. Tech., 2010, 204, 2442.
P. C. Tway, L. J. Cline Love, J. Phys. Chem., 1982, 86, 5223.
H. Yang, Y.-Q. Ge, J. Jia, J.-W. Wang, J. Lumin., 2011, 131, 749.
Y. Li, J. Wang, W. Guo, C. Gao, Z. Cheng, Instrum. Sci. Technol., 2017, 45, 290.
H. Wang, C. Burda, G. Persy, J. Wirz, J. Am. Chem. Soc., 2000, 122, 5849.
Y.-X. Tan, Y. Zhang, Y.-P. He, Y.-J. Zheng, New J. Chem., 2014, 38, 5272.
S. Biswas, M. Tonigold, M. Speldrich, P. Kögerler, D. Volkmer, Eur. J. Inorg. Chem., 2009, 2009, 3094.
N. T. Abdel-Ghani, M. F. A. El-Ghar, A. M. Mansour, Spectrochim. Acta, Part A, 2013, 104, 134.
T. D. Keene, I. Zimmermann, A. Neels, O. Sereda, J. Hauser, M. Bonin, M. B. Hursthouse, D. J. Price, S. Decurtins, Dalton Trans., 2010, 39, 4937.
E. Apohan, U. Yilmaz, O. Yilmaz, A. Serindag, H. Küçükbay, O. Yesilada, Y. Baran, J. Organomet. Chem., 2017, 828, 52.
M.-X. Yao, M.-H. Zeng, H.-H. Zou, Y.-L. Zhou, H. Liang, Dalton Trans., 2008, 2428.
K. E. Prosser, S. W. Chang, F. Saraci, P. H. Lê, C. J. Walsby, J. Inorg. Biochem., 2017, 167, 89.
R. C. Rocha, H. E. Toma, Inorg. Chim. Acta, 2000, 310, 65.
S.-D. Han, J.-P. Zhao, Y.-Q. Chen, S.-J. Liu, X.-H. Miao, T.-L. Hu, X.-H. Bu, Cryst. Growth Des., 2014, 14, 2.
E. Üstün, A. Özgür, K. A. Coşkun, S. Demir, İ. Özdemir, Y. Tutar, J. Coord. Chem., 2016, 69, 3384.
M. Kosea, V. McKee, Polyhedron, 2014, 75, 30.
S. R. Zheng, S. L. Cai, J. B. Tan, J. Fan, W.-G. Zhang, Inorg. Chem. Commun., 2012, 21, 100.
D.-C. Zhong, J.-H. Deng, X.-Z. Luo, H.-J. Liu, J.-L. Zhong, K.-J. Wang, T.-B. Lu, Cryst. Growth Des., 2012, 12, 1992.
P. A. M. Williams, E. G. Ferrer, M. J. Correa, E. J. Baran, E. E. Castellano, O. E. Piro, J. Chem. Crystallogr., 2004, 34, 285.
Y.-Q. Chen, Y.-K. Qu, G.-R. Li, Z.-Z. Zhuang, Z. Chang, T.-L. Hu, J. Xu, X.-H. Bu, Inorg. Chem., 2014, 53, 8842.
B. S. Barros, J. Chojnacki, A. A. M. Soares, J. Kulesza, L. L. da Luz, S. Alves, Jr., Mater. Chem. Phys., 2015, 162, 364.
Y.-C. Shen, Z.-J. Li, J.-K. Cheng, Y.-Y. Qin, Y.-G. Yao, Inorg. Chem. Commun., 2008, 10, 888.
L. Song, M. Zhao, G. Cui, J. Gao, L. Lin, Acta Crystallogr., Sect. E, 2012, 68, m1179.
X. Wang, N. Ling, Y.-X. Li, S.-L. Zhang, Z. Kristallogr. — New Cryst. Struct., 2018, 233, 215.
J. Zhao, D. Zhao, Y. Zhao, H. Shu, J. Hu, Polyhedron, 2016, 119, 7.
J. Hu, Y. Guo, J. Zhao, J. Zhang, Bioorg. Med. Chem., 2017, 25, 5733.
J. Zhao, S. Li, D. Zhao, S. Chen, J. Hu, J. Coord. Chem., 2013, 66, 1650.
J. Zhao, Y. Guo, J. Hu, H. Yu, S. Zhi, J. Zhang, Polyhedron, 2015, 102, 163.
J. Hu, Y. Zhao, F. Yang, C. Liao, J. Zhao, J. Coord. Chem., 2018, 71, 1368.
J. Zhao, Y. Guo, K. Peng, J. Zhang, C. Zhang, L. Hu, S. Chen, J. Hu, Polyhedron, 2015, 85, 593.
J. Zhao, S. Li, S. Chen, Y. Bai, J. Hu, J. Coord. Chem., 2012, 65, 1201.
J. H. Burckhalter, V. C. Stephens, L. A. R. Hall, J. Am. Chem. Soc., 1952, 74, 3868.
ADF2013, Software for Chemistry & Materials, Theoretical Chemistry, Vrije Universiteit, Amsterdam (The Netherlands), 2013.
J. P. Perdew, K. Burke, M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865.
E. van Lenthe, E. J. Baerends, J. Comput. Chem., 2003, 24, 1142.
E. van Lenthe, A. E. Ehlers, E. J. Baerends, J. Chem. Phys., 1999, 110, 8943.
E. van Lenthe, E. J. Baerends, J. G. Snijders, J. Chem. Phys., 1993, 99, 4597.
E. van Lenthe, E. J. Baerends, J. G. Snjjders, J. Chem. Phys., 1994, 101, 9783.
E. van Lenthe, J. G. Snjjders, E. J. Baerends, J. Chem. Phys., 1996, 105, 6505.
E. van Lenthe, R. van Leeuwen, E. J. Baerends, J. G. Snijders, Int. J. Quantum Chem., 1996, 57, 281.
Bruker AXS Inc. (2000-2012), APEX2 (Version 2.0), SAINT (Version 8.18c), and SADABS (Version 2.11), Bruker Advanced X-ray Solutions, Madison, WI, USA, 2012.
E. Y. Semitut, T. S. Sukhikh, E. Y. Filatov, G. A. Anosova, A. A. Ryadun, K. A. Kovalenko, A. S. Potapov, Cryst. Growth Des., 2017, 17, 5559.
X.-W. Zhu, Y.-J. Zhang, X.-Z. Yang, H.-Y. Qian, C.-X. Zhang, Z. Krist.-New Cryst. St., 2010, 225, 405.
G. te Velde, F. M. Bickelhaupt, E. J. Baerends, C. Fonseca Guerra, S. J. A. van Gisbergen, J. G. Snijders, T. Ziegler, J. Comput. Chem., 2001, 22, 931.
F. M. Bickelhaupt, E. J. Baerends, Rev. Comput. Chem., 2000, 15, 1.
R. F. Bader, in Atoms in Molecules: A Quantum Theory, Clarendon Press, Oxford, 1990, p. 438.
Yu. A. Abramov, Acta Crystallogr., Sect. A, 1997, 53, 264.
D. Cremer, E. Kraka, Croat. Chem. Acta, 1984, 57, 1259.
E.-C. Yang, H.-K. Zhao, B. Ding, X.-G. Wang, X.-J. Zhao, Cryst. Growth Des., 2007, 7, 2009.
M.-C. Hu, Y. Wang, Q.-G. Zhai, S.-N. Li, Y.-C. Jiang, Y. Zhang, Inorg. Chem., 2009, 48, 1449.
T. Wu, D. Li, X.-L. Feng, J.-W. Cai, Inorg. Chem. Commun., 2003, 6, 886.
H.-N. Chang, S.-X. Hou, G.-H. Cui, S.-C. Wang, J. Inorg. Organomet. Polym., 2017, 27, 518.
X.-x. Wang, J.-m. Hao, G.-y. Li, G.-h. Cui, J. Chem. Crystallogr., 2015, 45, 77.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors express their gratitude to the following researchers working at the Nikolaev Institute of Inorganic Chemistry: A. P. Zubareva and V. V. Ankudovich (for carrying out elemental analysis), N. I. Alferova and L. A. Sheludyakova (for IR spectroscopic studies), I. V. Korolkov (for powder XRD study), and A. S. Berezin (for luminescence studies).
This work was financially supported by the Russian Science Foundation (Project No. 18-73-00294).
Russian Chemical Bulletin, International Edition, Vol. 69, No. 10, pp. 1873–1883, October, 2020
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 1873–1883, October, 2020.
Rights and permissions
About this article
Cite this article
Smirnova, K.S., Lider, E.V., Kozlova, S.G. et al. Zinc complexes with 1-(1H-benzimidazol-1-ylmethyl)-1H-benzotriazole: the structure, quantum chemical calculations, and luminescence properties. Russ Chem Bull 69, 1873–1883 (2020). https://doi.org/10.1007/s11172-020-2973-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-020-2973-6