Abstract
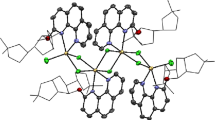
The complex salt {[CuL2][Cu4I6]•MeCN}n (1) and the compound [Cu4L3I4]•3 MeCN (2) (L is a chiral ligand bearing a natural monoterpene (+)-3-carene moiety) were synthesized. The crystal structures of compounds 1 and 2 were determined by X-ray diffraction. The structure of compound 1 consists of complex cations [CuL2]2+ (N3O2 polyhedron is a trigonal bipyramid) and CuI coordination polymers (CuI4 polyhedron is a tetrahedron) as anions. The experimental magnetic moment μeff at 300 K is 1.90 μB, which is consistent with the X-ray diffraction data and the assumption that compound 1 is mixed-valence. The structure of compound 2 comprises a tetranuclear CuI complex, in which three Cu atoms are coordinated by two N atoms of the ligand L and two I atoms, and the fourth Cu atom is coordinated by four I atoms (coordination polyhedra are distorted tetrahedra). Compounds L and 2 were found to influence the viability of human laryngeal carcinoma cells (Hep2). The IC50 value for complex 2 (13.0±1.7 μM) is substantially smaller than IC50 for compound L (30.5±0.5 μM).
Similar content being viewed by others
References
S. Sharma, M. Chauhan, A. Jamsheera, S. Tabassum, F. Arijmand, Inorg. Chim. Acta, 2017, 458,8.
G. Muller, Dalton Trans., 2009, 9692.
T. Wu, X.-Z. You, P. Boun, Coord. Chem. Rev., 2015, 284,1.
J. Grassous, Chem. Soc. Rev., 2009, 38, No 3,830.
R. Carr, N.H. Evans, D. Parcer, Chem. Soc. Rev., 2012, 41, 7673.
K. Inoue, Sh.-I. Ohkoshi, H. Imai, Magnetism: Molecules to Materials V, Eds J. S. Miller, M. Drillon, Wiley-VCH VerlagGmbH, Weinheim (FRG), 2002,42.
X.-L. Li, K. Chen, Y. Liu, Z.-X. Wang, T.-W. Wang, J.-L. Zuo, Y.-Z. Li, Y. Wang, J. S. Zhu, J.-M. Liu, Y. Song, X.-Z. You, Angew. Chem., Int. Ed., 2007, 46, 6820.
A. von Zelewsky, O. Mamula, J. Chem. Soc., Dalton Trans., 2000,219.
O. Mamula, A. von Zelewsky, Coord. Chem. Rev., 2003, 242,87.
A. V. Tkachev, Mendeleev Chem. J., 1998, 42,42.
S. V. Larionov, A. V. Tkachev, Mendeleev Chem. J., 2004, 48,154.
S. V. Larionov, Russ. J. Coord. Chem., 2012, 38,1.
G. Muller, J.-C. G. Bünzli, J. P. Riehl, D. Suhr, A. von Zelewsky, H. Mürner, Chem. Commun., 2002, 1522.
J. L. Lunkley, D. Shirotani, K. Yamanari, S. Kaizaki, G. Muller, Inorg. Chem., 2011, 50, 12724.
K. D. Oyler, F. J. Coughlin, S. Bernhard, J. Am. Chem. Soc., 2007, 129,210.
S. V. Larionov, Z. A. Savelreva, R. F. Klevtsova, L. A. Glinskaya, E. M. Uskov, S. A. Popov, A. V. Tkachev, Russ. J. Struct. Chem., 2010, 51,519.
S. V. Larionov, Z. A. Savelreva, R. F. Klevtsova, L. A. Glinskaya, E. M. Uskov, M. I. Rakhmanova, S. A. Popov, A. V. Tkachev, Russ. J. Struct. Chem., 2011, 52,531.
T. E. Kokina, L. A. Glinskaya, A. V. Tkachev, V. F. Plyusnin, Yu. V. Tsoy, I. Yu. Bagryanskaya, E. S. Vasilyev, D. A. Piryazev, L. A. Sheludyakova, S. V. Larionov, Polyhedron, 2016, 117,437.
T. E. Kokina, L. A. Glinskaya, E. S. Vasiliev, M. I. Rakhmanova, S. V. Makarova, D. A. Piryazev, I. V. Korol´kov, A. V. Tkachev, S. V. Larionov, Russ. J. Struct. Chem., 2017, 58, 1027.
C. Duncan, A. R. White, Metallomics, 2012, 4,127.
C. Santini, M. Pelei, V. Gandin, M. Porchia, F. Tisato, C. Marzano, Chem. Rev., 2014, 114,815.
D. Krajciová, M. Melník, E. Havránek, A. Forgácsová, P. Mikus, J. Coord. Chem., 2014, 67, 1493.
M. Wehbe, A. W. Y. Leung, M. J. Abrams, C. Orvig, M. B. Bally, Dalton Trans., 2017, 46, 10758.
P. Jia, R. Ouyang, P. Cao, X. Tong, X. Zhou, T. Lei, Yu. Zhao, N. Guo, H. Chang, Yu. Miao, Sh. Zhou, J. Coord. Chem., 2017, 70, 2175.
T. A. Fernandes, F. Mendes, A. P. S. Roseiro, I. Santos, M. F. N. N. Carvalho, Polyhedron, 2015, 87,215.
M. E. S. Serra, D. Murtinho, Z. N. da Rocha, A. S. Pires, J. G. Baptista, A. M. Abrantes, M. Laranjo, J. E. Casalta-Lopes, M. F. Botelho, A. A. C. C. Pais, S. C.C. Nunes, H. D. Burrows, T. Costa, Polyhedron, 2017, 137,147.
B.-L. Fei, Z.-X. Huang, W.-Sh. Xu, D.-D. Li, Ya. Lu, W.-L. Gao, Yu. Zhao, Yu Zhang, Q.-B. Liu, J. Photochemistry Photobiology, B: Biology, 2016, 160,43.
N. Armaroli, G. Accorsi, F. Cardinali, A. Listorti, Top. Curr. Chem., 2007, 280,69.
P. Ford, E. Cariarti, J. Bourassa, Chem. Rev., 1999, 99, 3625.
C. Marzano, M. Pellei, F. Tisato, C. Santini, Anti-Cancer Agents in Medicinal Chemistry, 2009, 9,185.
R. Galindo-Murillo, J. C. Garcia-Ramos, L. Ruiz-Azuara, T. E. Cheatham, F. Cortés-Guzmán, Nucleic Acids Res., 2015, 43, 5364.
Y. Wei, N. Yoshikai, J. Am. Chem. Soc., 2013, 135, 3756.
A. Nurtaeva, E. M. Holt, Acta Cryst., 1998, C54,594.
S.-L. Li, R. Zhang, J.-J. Hou, X.-M. Zhang, Inorg. Chem. Commun., 2013, 32,12.
A. V. Artem´ev, J. A. Eremina, E. V. Lider, O. V. Antonova, E. V. Vorontsova, I. Yu. Bagryanskaya, Polyhedron, 2017, 138,218.
P. A. Petukhov, A. V. Tkachev, Tetrahedron, 1997, 53, 2535.
V. V. Zakharychev, A. V. Kuzenkov, Chem. Heterocycl. Compd., 2007, 43,989.
G. M. Sheldrick, Acta Crystallogr. Sect. C: Cryst. Struct. Commun., 2015, 71, 3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on the materials of the XXVII International Chugaev Conference on Coordination Chemistry (October 2–6, 2017; Nizhny Novgorod, Russia).
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 7, pp. 1251–1260, July, 2018.
Rights and permissions
About this article
Cite this article
Kokina, T.E., Glinskaya, L.A., Piryazev, D.A. et al. Synthesis and structures of CuI,II complexes with a 2,2´-bipyridine derivative bearing a (+)-3-carene moiety. Russ Chem Bull 67, 1251–1260 (2018). https://doi.org/10.1007/s11172-018-2209-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-018-2209-1