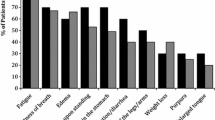
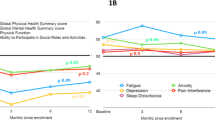
Abstract
Background
Light chain (AL) amyloidosis is a plasma cell neoplasm associated with high early mortality and severe morbidity that can cause severe disability. We explored the impact of AL amyloidosis on symptoms and well-being from the perspectives of patients and health care providers who regularly care for AL patients. We intended to develop a conceptual understanding of patient-reported outcomes in AL amyloidosis to identify the context of use and concept of interest for a clinical outcome assessments tool in this disease.
Method
Twenty patients and ten professionals were interviewed. Patient interviews captured the spectrum of amyloidosis experience including time from diagnosis, type of organ involvement, and presence and type of treatment received. Interviews with professionals included physicians, advanced practice providers, registered nurse, and a patient advocate; these interviews covered similar topics.
Results
The impact of AL amyloidosis on patients’ life was multidimensional, with highly subjective perceptions of normality and meaning. Four major themes from patients and experts included diagnosis of AL amyloidosis, living with AL amyloidosis, symptom burden, and social roles. Barriers to patient-reported outcomes data collection in patients were additionally explored from experts. The themes provide a comprehensive understanding of the important experiences of symptom burden and its impact on daily life from AL amyloidosis patients’ and from the perspectives of professionals who care for patients with AL amyloidosis.
Conclusion
These findings further the conceptual understanding and identification of a preliminary model of concept of interest for development of a clinical outcome assessments tool for AL amyloidosis.


Similar content being viewed by others
References
Bayliss, M., McCausland, K. L., Guthrie, S. D., & White, M. K. (2017). The burden of amyloid light chain amyloidosis on health-related quality of life. Orphanet Journal of Rare Diseases, 12(1), 15. https://doi.org/10.1186/s13023-016-0564-2
Merlini, G. (2017). AL amyloidosis: from molecular mechanisms to targeted therapies. Hematology/The Education Program of the American Society of Hematology. American Society of Hematology. Education Program, 2017(1), 1–12.
Lousada, I., Comenzo, R. L., Landau, H., Guthrie, S., & Merlini, G. (2015). Light chain amyloidosis: Patient experience survey from the Amyloidosis Research Consortium. Advances in Therapy, 32(10), 920–928.
Muchtar, E., Gertz, M. A., Kumar, S. K., Lacy, M. Q., Dingli, D., Buadi, F. K., ... & Dispenzieri, A. (2017). Improved outcomes for newly diagnosed AL amyloidosis between 2000 and 2014: cracking the glass ceiling of early death. Blood, 129(15), 2111–2119. https://doi.org/10.1182/blood-2016-11-751628
Kumar, S. K., Gertz, M. A., Lacy, M. Q., Dingli, D., Hayman, S. R., Buadi, F. K., et al. (2011). Recent improvements in survival in primary systemic amyloidosis and the importance of an early mortality risk score. Mayo Clinic Proceedings, 86(1), 12–18. https://doi.org/10.4065/mcp.2010.0480
Lin, H. M., Seldin, D., Hui, A. M., Berg, D., Dietrich, C. N., & Flood, E. (2015). The patient’s perspective on the symptom and everyday life impact of AL amyloidosis. Amyloid : The International Journal of Experimental and Clinical Investigation : The Official Journal of the International Society of Amyloidosis, 22(4), 244–251. https://doi.org/10.3109/13506129.2015.1102131
Rizio, A. A., White, M. K., McCausland, K. L., Quock, T. P., Guthrie, S. D., Yokota, M., & Bayliss, M. S. (2018). Treatment tolerability in patients with immunoglobulin light-chain amyloidosis. American Health & Drug Benefits, 11(8), 430.
Hsieh, H. F., & Shannon, S. E. (2005). Three approaches to qualitative content analysis. Qualitative Health Research, 15(9), 1277–1288. https://doi.org/10.1177/1049732305276687
Aronson, J. (1995). A pragmatic view of thematic analysis. The Qualitative Report, 2(1), 1–3.
Cella, D., Yount, S., Rothrock, N., Gershon, R., Cook, K., Reeve, B., …PROMIS Cooperative Group. (2007) The patient-reported outcome measurement information system (PROMIS): Progress of an NIH Roadmap Cooperative Agreement during its first two years. Medical Care 45 S3
DeWalt, D. A., Rothrock, N., Yount, S., & Stone, A. A. (2007). Evaluation of item candidates: The PROMIS qualitative item review. Medical Care, 45(5 Suppl 1), S12.
Riley, W. T., Pilkonis, P., & Cella, D. (2011). Application of the National Institutes of Health patient-reported outcomes measurement information system (PROMIS®) to mental health research. The Journal of Mental Health Policy and Economics, 14(4), 201.
Retrieved from http://www.healthmeasures.net/images/PROMIS/manuals/PROMIS_Fatigue_Scoring_Manual.pdf. 2019.
Hays, R. D., Spritzer, K., Amtmann, D., Lai, J. S., DeWitt, E. M., & Rothrock, N. (2013). Upper-extremity and mobility subdomains from the Patient-Reported Outcomes Measurement Information System (PROMIS) adult physical functioning item bank. Archives of Physical Medicine and Rehabilitation, 94(11), 2291–2296. https://doi.org/10.1016/j.apmr.2013.05.014
McCausland, K. L., White, M. K., Guthrie, S. D., Quock, T., Finkel, M., Lousada, I., & Bayliss, M. S. (2018). Light chain (AL) amyloidosis: The journey to diagnosis. Patient, 11(2), 207–216. https://doi.org/10.1007/s40271-017-0273-5
Wright, N. L., Flynn, K. E., Brazauskas, R., Hari, P., & D’Souza, A. (2018). Patient-reported distress is prevalent in systemic light chain (AL) amyloidosis but not determined by severity of disease. Amyloid : The International Journal of Experimental and Clinical investigation : The Official Journal of the International Society of Amyloidosis, 25(2), 129–134. https://doi.org/10.1080/13506129.2018.1486298
Sanchorawala, V., McCausland, K. L., White, M. K., Bayliss, M. S., Guthrie, S. D., Lo, S., & Skinner, M. (2017). A longitudinal evaluation of health-related quality of life in patients with AL amyloidosis: associations with health outcomes over time. British Journal of Haematology, 179(3), 461–470. https://doi.org/10.1111/bjh.14889
McCausland, K. L., Quock, T. P., Rizio, A. A., Bayliss, M. S., White, M. K., Guthrie, S. D., & Sanchorawala, V. (2018). Cardiac biomarkers and health-related quality of life in patients with light chain (AL) amyloidosis. British Journal of Haematology. https://doi.org/10.1111/bjh.15693
D’Souza, A., Magnus, B. E., Myers, J., Dispenzieri, A., & Flynn, K. E. (2020). The use of PROMIS patient-reported outcomes (PROs) to inform light chain (AL) amyloid disease severity at diagnosis. Amyloid : The International Journal of Experimental and Clinical Investigation : The Official Journal of the International Society of Amyloidosis. https://doi.org/10.1080/13506129.2020.1713743
Warsame, R., & D’Souza, A. (2019). Patient reported outcomes have arrived: A practical overview for clinicians in using patient reported outcomes in oncology. Mayo Clinic Proceedings, 94(11), 2291–2301. https://doi.org/10.1016/j.mayocp.2019.04.005
Acknowledgements
The authors acknowledge Ms. Claire Piehowski’s contribution toward data management and coding assistance. Financial support for this study was provided by a K23 HL141445 Mentored Career Development Award. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Author information
Authors and Affiliations
Contributions
Conception/design: AD’S, KEF. Provision of study materials or patients: AD’S, MF. Collection and/or assembly of data: AD’S, JM, RC, KEF. Data analysis and interpretation AD’S, JM, RC, AD, MF, JP, KEF. Manuscript writing: AD’S, JM, RC, AD, MF, JP, KEF. Final approval of manuscript: AD’S, JM, RC, AD, MF, JP, KEF.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
D’Souza, A., Myers, J., Cusatis, R. et al. Development of a conceptual model of patient-reported outcomes in light chain amyloidosis: a qualitative study. Qual Life Res 31, 1083–1092 (2022). https://doi.org/10.1007/s11136-021-02943-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-021-02943-w