Abstract
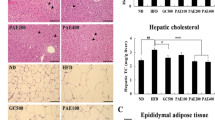
Previous studies have demonstrated that the kiwifruit peel, which is usually discarded by consumers and factories, has the highest polyphenol content among all parts of the kiwifruit. To maximize the utilization of these waste resources, the aim of this study was to examine the regulatory effects of polyphenols extracted from kiwifruit peel (KPE) on lipid metabolism and investigate their underlying mechanisms. Thirty-two male Sprague‒Dawley rats were divided into four groups: those fed a normal diet, those fed a high-fat (HF) diet, and those fed a HF diet with a low dose of KPE solution (50 mg/kg) or a high dose of KPE (100 mg/kg) by gavage. The findings of the study revealed that KPE effectively reduced body weight gain and the increases in triglycerides and total cholesterol in serum induced by the HF diet (HFD). Additionally, KPE supplementation led to a significant decrease in hepatic fat accumulation, potentially by increasing hepatic oxidation abilities. Hepatic lipidomics demonstrated that KPE influenced various metabolic pathways, including linoleic acid metabolism, steroid biosynthesis, and the biosynthesis of unsaturated fatty acids in HFD-induced rats, which were associated with the downregulation of FATP2, ACC, FAS, GPAT, DGTA1, DGTA2, and PPARγ expression as well as the upregulation of AMPK, PGC-1α, CPT-1, and PPARα expression. These findings suggest that KPE has considerable regulatory effects in rats with dyslipidaemia, which may provide supporting information for the reuse of kiwifruit peel.
Graphical Abstract







Similar content being viewed by others
Data Availability
The data presented in this study are available on request from the corresponding author.
References
Pirillo A, Casula M, Olmastroni E, Norata G, Catapano A (2021) Global epidemiology of dyslipidaemias. Nat Rev Cardiol 18:689–700. https://doi.org/10.1038/s41569-021-00541-4
Wannamethee S, Shaper A, Ebrahim S (2000) HDL-cholesterol, total cholesterol, and the risk of stroke in middle-aged british men. Stroke 31:1882–1888. https://doi.org/10.1161/01.STR.31.8.1882
Katundu K, Mukhula V, Phiri T, Phiri C, Filisa F, Chipewa P, Chirambo G, Mipando M, Mwandumba H, Muula A (2022) High prevalence of dyslipidemia among persons with diabetes mellitus and hypertension at a tertiary hospital in Blantyre, Malawi. BMC Cardiovasc Disord 22:557. https://doi.org/10.1186/s12872-022-03011-y
Barouki R, Gluckman P, Grandjean P, Hanson M, Heindel J (2012) Developmental origins of non-communicable disease: implications for research and public health. Environ. Health 11:42. https://doi.org/10.1186/1476-069x-11-42
Cory H, Passarelli S, Szeto J, Tamez M, Mattei J (2018) The role of polyphenols in human health and food systems: a mini-review. Front Nutr 5:87. https://doi.org/10.3389/fnut.2018.00087
Ding S, Jiang H, Fang J (2018) Regulation of immune function by polyphenols. J Immunol Res 2018:1264074. https://doi.org/10.1155/2018/1264074
Zhai X, Lin D, Zhao Y, Li W, Yang X (2018) Enhanced anti-obesity effects of bacterial cellulose combined with konjac glucomannan in high-fat diet-fed C57BL/6J mice. Food Funct 9:5260–5272. https://doi.org/10.1039/c8fo01211c
Caceres C, Kim M, Bae M, Pham T, Lee Y, Hu S, O'Neill E, Kim B, Park Y, Lee J (2021) The effect of cranberry consumption on lipid metabolism and inflammation in human apo A-I transgenic mice fed a high-fat and high-cholesterol diet. Br J Nutr 126:183–190. https://doi.org/10.1017/s0007114520004080
Das M, Geetha V, Zarei M, Harohally N (2022) Modulation of obesity-associated metabolic dysfunction by a novel lipophilic fraction obtained from Agaricus bisporus. Life Sci 305:120779. https://doi.org/10.1016/j.lfs.2022.120779
Cheynier V (2012) Phenolic compounds: from plants to foods. Phytochem Rev 11(2):153–177. https://doi.org/10.1007/s11101-012-9242-8
Yuan M, Chen X, Su T, Zhou Y, Sun X (2021) Supplementation of kiwifruit polyphenol extract attenuates high fat diet induced intestinal barrier damage and inflammation via reshaping gut microbiome. Front Nutr 8:702157. https://doi.org/10.3389/fnut.2021.702157
Wood D, Cristina P, Teixeira L, Gambero A, Villena J, Lima M (2018) Effects of polyphenols on thermogenesis and mitochondrial biogenesis. Int J Mol Sci 19(9):2757. https://doi.org/10.3390/ijms19092757
Duarte L, Gasaly N, Poblete C, Uribe D, Echeverria F, Gotteland M, Garcia-Diaz D (2021) Polyphenols and their anti-obesity role mediated by the gut microbiota: a comprehensive review. Rev Endocr Metab Disord 22:367–388. https://doi.org/10.1007/s11154-020-09622-0
Rodriguez I, Vauzour D, Minihane A (2016) Polyphenols and non-alcoholic fatty liver disease: impact and mechanisms. Proc Nutr Soc 75:47–60. https://doi.org/10.1017/S0029665115004218
Santhakumar A, Bulmer A, Singh I (2014) A review of the mechanisms and effectiveness of dietary polyphenols in reducing oxidative stress and thrombotic risk. J Hum Nutr Diet 27:1–21. https://doi.org/10.1017/10.1111/jhn.12177
Nazzaro F, Fratianni F, De Vincenzo V, Battistelli A, Da Adriano G, Coppola R (2020) Polyphenols, the new frontiers of prebiotics. In Advances in food and nutrition research, 1st edn.Elsevier, Amsterdam, pp 35-89
Yahfoufi N, Alsadi N, Jambi M, Matar C (2018) The immunomodulatory and anti-inflammatory role of polyphenols. Nutrients 10:1618. https://doi.org/10.3390/nu10111618
Yan Z, Zhong Y, Duan Y, Chen Q, Li F (2020) Antioxidant mechanism of tea polyphenols and its impact on health benefits. Anim Nutr 6:115–123. https://doi.org/10.1016/j.aninu.2020.01.001
Steinberg R, Hardie G (2023) New insights into activation and function of the AMPK. Nat Rev Mol Cell Biol 24:255–272. https://doi.org/10.1038/s41580-022-00547-x
Zang M, Xu S, Maitland K, Zuccollo A, Hou X, Jiang B, Wierzbicki M, Verbeuren J, Cohen A (2006) Polyphenols stimulate AMP-Activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic LDL receptor-deficient mice. Diabetes 55:2180–2191. https://doi.org/10.2337/db05-1188
Felix A, Takahashi N, Takahashi M, Katsumata R, Satoh R, Soon T, Miyajima K, Nakae D, Inoue H, Uehara M (2017) Extracts of black and brown rice powders improve hepatic lipid accumulation via the activation of PPARα in obese and diabetic model mice. Biosci Biotechnol Biochem 81:2209–2211. https://doi.org/10.1080/09168451.2017.1372178
Wu C, Lin M, Wang H, Yang M, Jou M, Wang C (2011) Rutin inhibits oleic acid induced lipid accumulation via reducing lipogenesis and oxidative stress in hepatocarcinoma cells. J Food Sci 76:65–72. https://doi.org/10.1111/j.1750-3841.2010.02033.x
Wu C, Ou T, Chang C, Chang X, Yang M, Wang C (2014) The polyphenol extract from Sechium edule shoots inhibits lipogenesis and stimulates lipolysis via activation of AMPK signals in HepG2 cells. J Agric Food Chem 62:750–759. https://doi.org/10.1021/jf404611a
Mulvihill E, Assini J, Lee J, Allister E, Sutherland B, Koppes J, Sawyez C, Edwards J, Telford D, Charbonneau A (2011) Nobiletin attenuates VLDL overproduction, dyslipidemia, and atherosclerosis in mice with diet-induced insulin resistance. Diabetes 60:1446–1457. https://doi.org/10.2337/db10-0589
Funding
This research was funded by the National Natural Science Foundation of China (NSFC) (32060537), the Science and Technology Support Project in Guizhou Province (Qian ke he Jichu ZK [2022]360).
Author information
Authors and Affiliations
Contributions
Conceptualization, Y.Z., and X.S.; Methodology, T.S; Software, Y.Z., X.C., and T.S.; Validation, Y.Z., and X.C.; Data curation, T.S. and M.Y.; Writing-original draft preparation, Y.Z. and X.C.; Writing-review and editing, Y.Z. and X.C.; Funding acquisition, Y.Z. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
The experimental procedures were conducted by the National Institute of Health’s Guide for the Care and Use of Laboratory Animals and approved by the Ethical Committee on Animal Research at the Guizhou Medical University (No. 1702105).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, Y., Chen, X., Su, T. et al. Kiwifruit Peel Extract Improves the Alterations in Lipid Metabolism in High-fat Diet-fed Model Rats. Plant Foods Hum Nutr 79, 113–119 (2024). https://doi.org/10.1007/s11130-023-01132-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-023-01132-z