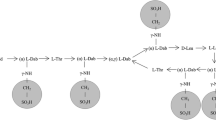
Abstract
Purposes
To evaluate the effects of component contents in different colistin methanesulfonate (CMS) formulas on their clinical pharmacokinetics of the prodrug CMS and the formed colistin.
Methods
Two CMS formulas (CTTQ and Parkedale) were investigated in a single dose, randomized, open-label, crossover study conducted in 18 healthy Chinese subjects. Both CMS formulas met the requirements of European Pharmacopoeia 9.2 with 12.1% difference in the two major active components (CMS A and CMS B). The PK parameters after a single intravenous infusion of CMS at 2.5 mg/kg were calculated and the steady-state plasma colistin concentrations (Css,avg) following multiple dosing, once every 12 h for 7 days, were simulated with the non-compartment model.
Results
The systemic exposure (AUC0-inf) of CMS were 59.49 ± 5.90 h·μg/mL and 51.09 ± 4.70 h·μg/mL, and the AUC0-inf of colistin were 15.39 ± 2.63 h·μg/mL and 12.36 ± 2.10 h·μg/mL for CTTQ and Parkedale, respectively. The ratios (90% CI) of geometric mean of AUC0-inf of CTTQ to Parkedale were 116.38% (112.95%, 119.91%) and 124.49% (120.76%, 128.35%) for CMS and colistin, respectively. The predicted Css,avg (95% CI) were 0.92 (0.85, 0.99) μg/mL and 0.74 (0.69, 0.79) μg/mL for CTTQ and Parkedale, respectively.
Conclusion
The difference in component content in the two CMS formulas had a significant (P < 0.001) impact on the systemic exposure of colistin in human, thus, warranted essential considerations in clinical applications.



Similar content being viewed by others
Abbreviations
- AE:
-
Adverse events
- AKI:
-
Acute kidney injury
- AUC0–12 :
-
Area under the concentration–time curve from time 0 to 12 h after drug administration
- AUC0–24 :
-
Area under the concentration–time curve from time 0 to 24 h after drug administration
- AUC0-inf :
-
Area under the concentration–time curve from time zero to infinity after drug administration
- AUCss,24 :
-
Steady-state 24-h area under the concentration-time curve
- BMI:
-
Body mass index
- CBA:
-
Colistin base activity
- CLr :
-
Renal clearance
- CLt :
-
Total clearance
- Cmax :
-
Peak concentration
- CMS:
-
Colistin methanesulfonate
- CMSA:
-
Colistin A methanesulfonate
- CMSB:
-
Colistin B methanesulfonate
- Css,avg :
-
Average steady-state concentration
- CysC:
-
Cysteine dehydrogenase inhibitor C
- fAUC/MIC:
-
Ratio of area under curve of free colistin over minimum inhibitory concentration
- f m :
-
Fraction of CMS converted into colistin in blood
- MIC:
-
Minimum inhibitory concentration
- NGAL:
-
Neutrophil gelatinase-associated lipocalin
- PK:
-
Pharmacokinetics
- PK/PD:
-
Pharmacokinetic/pharmacodynamic
- SAE:
-
Serious adverse events
- T1/2 :
-
Half time
- TDM:
-
Therapeutic drug monitoring
- Tmax :
-
Time to peak concentration
- Vd :
-
Distribution volume
References
States U. Antibiotic resistance threats in the United States. Content source: Centers for Disease Control and Prevention. 2019.
Hu FP, Guo Y, Yang Y, Zheng YG, Wu S, Jiang XF, et al. Resistance reported from China antimicrobial surveillance network (CHINET) in 2018. Eur J Clin Microbiol Infect Dis. 2019;38(12):2275–81.
Nation RL, Li J. Colistin in the 21st century. Curr Opin Infect Dis. 2009;22(6):535–43.
Chew KL, La MV, Lin RTP, Teo JWP. Colistin and polymyxin B susceptibility testing for crbapenem-resistant and mcr-positive Enterobacteriaceae: comparison of Sensititre, MicroScan, Vitek 2, and Etest with broth microdilution. J Clin Microbiol. 2017;55(9):2609–16.
Cheah SE, Li J, Tsuji BT, Forrest A, Bulitta JB, Nation RL. Colistin and polymyxin B dosage regimens against Acinetobacter baumannii: differences in activity and the emergence of resistance. Antimicrob Agents Chemother. 2016;60(7):3921–33.
Mitsugui CS, Tognim MC, Cardoso CL, Carrara-Marroni FE, Botelho GL. In vitro activity of polymyxins in combination with beta-lactams against clinical strains of Pseudomonas aeruginosa. Int J Antimicrob Agents. 2011;38(5):447–50.
Li J, Nation RL, Milne RW, Turnidge JD, Coulthard K. Evaluation of colistin as an agent against multi-resistant gram-negative bacteria. Int J Antimicrob Agents. 2005;25(1):11–25.
Bergen PJ, Li J, Rayner CR, Nation RL. Colistin methanesulfonate is an inactive prodrug of colistin against Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2006;50(6):1953–8.
Dijkmans AC, Wilms EB, Kamerling IM, Birkhoff W, Ortiz-Zacarias NV, van Nieuwkoop C, et al. Colistin: revival of an old polymyxin antibiotic. Ther Drug Monit. 2015;37(4):419–27.
Beveridge EG, Martin AJ. Sodium sulphomethyl derivatives of polymyxins. Br J Pharmacol Chemother. 1967;29(2):125–35.
Barnett M, Bushby SR, Wilkinson S. Sodium sulphomethyl derivatives of polymyxins. Br J Pharmacol Chemother. 1964;23:552–74.
Li J, Milne RW, Nation RL, Turnidge JD, Coulthard K. Stability of colistin and colistin methanesulfonate in aqueous media and plasma as determined by high-performance liquid chromatography. Antimicrob Agents Chemother. 2003;47(4):1364–70.
Li J, Nation RL, Turnidge JD, Milne RW, Coulthard K, Rayner CR, et al. Colistin: the re-emerging antibiotic for multidrug-resistant gram-negative bacterial infections. Lancet Infect Dis. 2006;6(9):589–601.
Naghmouchi K, Hammami R, Fliss I, Teather R, Baah J, Drider D. Colistin a and colistin B among inhibitory substances of Paenibacillus polymyxa JB05-01-1. Arch Microbiol. 2012;194(5):363–70.
Yu Z, Zhu Y, Fu J, Qiu J, Yin J. Enhanced NADH metabolism involves colistin-induced killing of Bacillus subtilis and Paenibacillus polymyxa. Molecules. 2019;24(3):387.
EDQM. Colistimethate Sodium. EP92. 2017.
Hartzell JD, Neff R, Ake J, Howard R, Olson S, Paolino K, et al. Nephrotoxicity associated with intravenous colistin (colistimethate sodium) treatment at a tertiary care medical center. Clin Infect Dis. 2009;48(12):1724–8.
Tsuji BT, Pogue JM, Zavascki AP, Paul M, Daikos GL, Forrest A, et al. International consensus guidelines for the optimal use of the polymyxins: Endorsed by the American College of Clinical Pharmacy (ACCP), European Society of Clinical Microbiology and Infectious Diseases (ESCMID), Infectious Diseases Society of America (IDSA), International Society for Anti-infective Pharmacology (ISAP), Society of Critical Care Medicine (SCCM), and Society of Infectious Diseases Pharmacists (SIDP). Pharmacotherapy. 2019;39(1):10–39.
Dudhani RV, Turnidge JD, Nation RL, Li J. fAUC/MIC is the most predictive pharmacokinetic/pharmacodynamic index of colistin against Acinetobacter baumannii in murine thigh and lung infection models. J Antimicrob Chemother. 2010;65(9):1984–90.
Dudhani RV, Turnidge JD, Coulthard K, Milne RW, Rayner CR, Li J, et al. Elucidation of the pharmacokinetic/pharmacodynamic determinant of colistin activity against Pseudomonas aeruginosa in murine thigh and lung infection models. Antimicrob Agents Chemother. 2010;54(3):1117–24.
Nation RL, Garonzik SM, Li J, Thamlikitkul V, Giamarellos-Bourboulis EJ, Paterson DL, et al. Updated US and European dose recommendations for intravenous colistin: how do they perform? Clin Infect Dis. 2016;62(5):552–8.
Matzneller P, Gobin P, Lackner E, Zeitlinger M. Feasibility of microdialysis for determination of protein binding and target site pharmacokinetics of colistin in vivo. J Clin Pharmacol. 2015;55(4):431–7.
He H, Li JC, Nation RL, Jacob J, Chen G, Lee HJ, et al. Pharmacokinetics of four different brands of colistimethate and formed colistin in rats. J Antimicrob Chemoth. 2013;68(10):2311–7.
Zhao M, Wu XJ, Fan YX, Guo BN, Zhang J. Development and validation of a UHPLC–MS/MS assay for colistin methanesulphonate (CMS) and colistin in human plasma and urine using weak-cation exchange solid-phase extraction. J Pharmaceut Biomed. 2016;124:303–8.
Zhao M, Wu XJ, Fan YX, Zhang YY, Guo BN, Yu JC, et al. Pharmacokinetics of colistin methanesulfonate (CMS) in healthy Chinese subjects after single and multiple intravenous doses. Int J Antimicrob Agents. 2018;51(5):714–20.
Morais JA, Lobato MR. The new European medicines agency guideline on the investigation of bioequivalence. Basic Clin Pharmacol Toxicol. 2010;106(3):221–5.
Bian XC, Liu XF, Chen YC, Chen DJ, Li J, Zhang J. Dose optimization of colistin combinations against carbapenem-resistant Acinetobacter baumannii from patients with hospital-acquired pneumonia in China by using an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob Agents Chemother. 2019;63(4):e01989–18.
ACKNOWLEDGEMENT AND DISCLOSURES
The authors declare no conflict of interest. The authors would like to thank CHIA TAI TIAN-QING Pharmaceutical Group Co., Ltd. (Jiangsu, China) for kindly providing the CMS formula. This study was supported by Major Research and Development Project of Innovative Drugs, Ministry of Science and Technology (2017ZX09304005). The authors would also like to thank Professor Jian Li, Facility for Anti-Infective Drug Development and Innovation, Victorian College of Pharmacy, Monash University for his advice on PK analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fan, YX., Chen, YC., Li, Y. et al. Effects of Different Component Contents of Colistin Methanesulfonate on the Pharmacokinetics of Prodrug and Formed Colistin in Human. Pharm Res 38, 79–87 (2021). https://doi.org/10.1007/s11095-021-02991-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-021-02991-4