Abstract
Purpose
Mono- and dual-decorated (DUAL) liposomes (LIP) were prepared, by immobilization of MAb against transferrin (TfR[OX26 or RI7217]) and/or a peptide analogue of ApoΕ3 (APOe) -to target low-density lipoprotein receptor(LPR)-, characterized physicochemically and investigated for BBB-targeting, in-vitro and in-vivo.
Methods
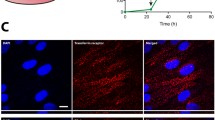
Human microvascular endothelial cells (hCMEC/D3) were used as BBB model, and brain targeting was studied by in-vivo imaging of DiR-labelled formulations (at two doses and surface ligand densities), followed by ex-vivo organ imaging.
Results
LIP diameter was between 100 nm and 150 nm, their stability was good and they were non-cytotoxic. LIP uptake and transport across the hCMEC/D3 cell monolayer was significantly affected by decoration with APOe or MAb, the DUAL exerting an additive effect. Intact vesicle-transcytosis was confirmed by equal transport of hydrophilic and lipophilic labels. In-vivo and ex-vivo results confirmed MAb and DUAL-LIP increased brain targeting compared to non-targeted PEG-LIPs, but not for APOe (also targeting ability of DUAL-LIP was not higher than MAb-LIP). The contradiction between in-vitro and in-vivo results was overruled when in-vitro studies (uptake and monolayer transport) were carried out in presence of serum proteins, revealing their important role in targeted-nanoformulation performance.
Conclusions
A peptide analogue of ApoΕ3 was found to target BBB and increase the targeting potential of TfR-MAb decorated LIP, in-vitro, but not in-vivo, indicating that different types of ligands (small peptides and antibodies) are affected differently by in-vivo applying conditions. In-vitro tests, carried out in presence of serum proteins, may be a helpful predictive “targetability” tool.










Similar content being viewed by others
References
Pardridge WM. Drug transport across the blood–brain barrier. J Cerebral Blood Flow Metabolism. 2012;32:1959–72.
Lu W, Xiong C, Zhang R, Shi L, Huang M, Zhang G, et al. Receptor-mediated transcytosis: A mechanism for active extravascular transport of nanoparticles in solid tumors. J Control Release. 2012;161:959–66.
Paliwal SR, Paliwa R, Agrawa GP, Vyas SP. Targeted breast cancer nanotherapeutics: options and opportunities with estrogen receptors. Critl Rev in Therap Drug Carrier Syst. 2012;29:421–46.
Ghosh SC, Neslihan AS, Klostergaard J. CD44: A validated target for improved delivery of cancer therapeutics. Exp Opinion on Therap Targets. 2012;16:635–50.
Jiang X, Sha X, Xin H, Chen L, Gao X, Wang X, et al. Self-aggregated pegylated poly (trimethylene carbonate) nanoparticles decorated with c(RGDyK) peptide for targeted paclitaxel delivery to integrin-rich tumors. Biomaterials. 2011;32:9457–69.
Wang Z, Yu Y, Dai W, Cui J, Wu H, Yuan L, et al. A specific peptide ligand-modified lipid nanoparticle carrier for the inhibition of tumor metastasis growth. Biomaterials. 2013;34(3):756–64.
Gomes-Da-Silva LC, Santos AO, Bimbo LM, Moura V, Ramalho JS, Pedroso De Lima MC, et al. Toward a siRNA-containing nanoparticle targeted to breast cancer cells and the tumor microenvironment. Int J Pharma. 2012;434:9–19.
Xin H, Sha X, Jiang X, Chen L, Law K, Gu J, et al. The brain targeting mechanism of Angiopep-conjugated poly(ethylene glycol)-co-poly(e{open}-caprolactone) nanoparticles. Biomaterials. 2012;33(5):1673–81.
Pardridge WM. Drug targeting to the brain. Pharm Res. 2007;24:1733–44.
Lalani J, Raichandani Y, Mathur R, Lalan M, Chutani K, Mishra AK, et al. Comparative receptor based brain delivery of tramadol-loaded poly(lactic-co-glycolic acid) nanoparticles. J Biomed Nanotech. 2012;8(6):918–27.
Xia H, Gao X, Gu G, Liu Z, Hu Q, Tu Y, et al. Penetration-functionalized PEG-PLA nanoparticles for brain drug delivery. Int J Pharmac. 2012;436:840–50.
López-Dávila V, Seifalian AM, Loizidou M. Organic nanocarriers for cancer drug delivery. Curr Opinion Pharmacol. 2012;12(4):414–9.
Antimisiaris SG, Kallinteri P, Fatouros D. Liposomes and drug delivery, In: S.C. Gad editor, Pharmaceutical Manufacturing Handbook Production and Processes, John Wiley & Sons, 2008, pp. 443-533.
Markoutsa E, Pampalakis G, Niarakis A, Romero IA, Weksler B, Couraud P-O, et al. Uptake and permeability studies of BBB-targeting immunoliposomes using the hCMEC/D3 cell line. Eur J Pharmaceut Biopharma. 2011;77(2):265–74.
Xiang Y, Liang L, Wang X, Wang J, Zhang X, Zhang Q. Chloride channel-mediated brain glioma targeting of chlorotoxin-modified doxorubicine-loaded liposomes. J Control Release. 2011;152:402–10.
Kluza E, Jacobs I, Hectors SJCG, Mayo KH, Griffoen AW, Strijkers GJ, et al. Dual-targeting of ανβ3 and galectin-1 improves the specificity of paramagnetic/fluorescent liposomes to tumor endothelium in vivo. J Control Release. 2012;158:207–14.
Ying X, Wen H, Lu W-L, Du J, Guo J, Tian W, et al. Dual-targeting daunorubicin liposomes improve the therapeutic efficacy of brain glioma in animals. J Control Release. 2010;141(2):183–92.
Li Y, He H, Jia X, Lu W-L, Lou J, Wei Y. A dual-targeting nanocarrier based on poly(amidoamine) dendrimers conjugated with transferrin and tamoxifen for treating brain gliomas. Biomaterials. 2012;33(15):3899–908.
Kibria G, Hatakeyama H, Ohga N, Hida K, Harashima H. Dual-ligand modification of PEGylated liposomes shows better selectivity and efficient gene delivery. J Control Release. 2011;153(2):141–8.
Bae S, Ma K, Kim TH, Lee ES, Oh KT, Park E-S, et al. Doxorubicin-loaded human serum albumin nanoparticles surface-modified with TNF-related apoptosis-inducing ligand and transferrin for targeting multiple tumor types. Biomaterials. 2012;33(5):1536–46.
Gao H, Qian J, Cao S, Yang Z, Pang Z, Pan S, et al. Precise glioma targeting of and penetration by aptamer and peptide dual-functioned nanoparticles. Biomaterials. 2012;33(20):5115–23.
Papademetriou T, Garnacho C, Schuchman EH, Muro S. In vivo performance of polymer nanocarriers dually-targeted to epitopes of the same or different receptors. Biomaterials. 2013;34:3459–66.
Ulbrich K, Hekmatara T, Herbert E, Kreuter J. Transferrin- and transferring-receptor-antibody-modified nanoparticles enable drug delivery across the blood-brain barrier (BBB). Eur J Pharm Biopharm. 2009;71:251–6.
Schnyder A, Huwyler J. Drug transport to brain with targeted Liposomes. NeuroRx. 2005;2:99–107.
Markoutsa E, Papadia K, Clemente C, Flores O, Antimisiaris SG. Anti-Aβ-MAb and dually decorated nanoliposomes: Effect of Aβ1-42 peptides on interaction with hCMEC/D3 cells. Eur J Pharm Biopharm. 2012;81(1):49–56.
Pardridge WM. Tyrosine hydroxylase replacement in experimental Parkinson’s disease with transvascular gene therapy. NeuroRx. 2005;2(1):129–38.
Re F, Cambianica I, Zona C, Sesana S, Gregori M, Rigolio R, et al. Functionalization of liposomes with ApoE-derived peptides at different density affects cellular uptake and drug transport across a blood-brain barrier model. Nanomed Nanotech Biol Med. 2011;7(5):551–9.
Re F, Cambianica I, Sesana S, Salvati E, Cagnotto A, Salmona M, et al. Functionalization with ApoE-derived peptides enhances the interaction with brain capillary endothelial cells of nanoliposomes binding amyloid-beta peptide. J Biotechnol. 2010;156(4):341–6.
Salvati A, Pitek AS, Monopoli MP, Prapainop K, Baldelli Bombelli F, Hristov DR, et al. Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nature Nanotech. 2013;8:137–43.
Weksler BB, Subileau EA, Perriere N, Charneau P, Holloway K, Leveque M, et al. Blood-brain barrier-specific properties of a human adult brain endothelial cell line. FASEB J. 2005;19:1872–4.
Poller R, Gutman H, Krahenbuhl S, Weksler B, Romero I, Couraud PO, et al. The human brain endothelial cell line hCMEC/D3 as a human blood-brain barrier model for drug transport studies. J Neurochem. 2008;107:1358–68.
Brambilla D, Nicolas J, Le Droumaguet B, Andrieux K, Marsaud V, Couraud P-O, et al. Design of fluorescently tagged poly(alkyl cyanoacrylate) nanoparticles for human brain endothelial cell imaging. Chem Commun. 2010;46:2602–4.
Kaasgaard T, Mouritsen OG, Jørgensen K. Receptor mediated binding of avidin to polymer covered liposomes. J Liposome Res. 2001;11(1):31–42.
Stewart JCM. Colorimetric determination of phospholipids with ammonium ferrothiocyanate. Anal Biochem. 1980;104:10–4.
Kokona M, Kallinteri P, Fatouros D, Antimisiaris SG. Stability of SUV liposomes in the presence of cholate salts and pancreatic lipases: effect of lipid composition. Eur J Pharm Sciences. 2000;9:245–52.
Zhang Y, Zhu C, Pardridge WM. Antisence gene therapy of brain cancer with an artificial virus gene delivery system. Mol Ther. 2002;6:67–72.
Tan PH, Manunta M, Ardjomand N, Xue SA, Larkin DF, Haskard DO, et al. Antibody targeted gene transfer to endothelium. J Gene Med. 2003;5:311–23.
Kalchenko V, Shivtiel S, Malina V, Lapid K, Haramati S, Lapidot T, et al. Use of lipophilic near-infrared dye in whole-body optical imaging of hematopoietic cell homing. J Biomed Opt. 2006;11(5):505–7.
Kreuter J. Influence of the surface properties on nanoparticle mediated transport of drugs to the brain. J Nanosci Nanotechnol. 2004;4:484–8.
Kreuter J, Shamenkov D, Petrov V, Ramge P, Cychutek K, Koch- Brandt C, et al. Apolipoprotein-mediated transport of nanoparticle-bound drugs across the blood–brain barrier. J Drug Target. 2002;10:317–25.
Chang J, Paillard A, Passirani C, Morille M, Benoit J-P, Betbeder D, et al. Transferrin adsorption onto PLGA nanoparticles governs their interaction with biological systems from blood circulation to brain cancer cells. Pharm Research. 2012;29(6):1495–505.
Elias DR, Poloukhtine A, Popik V, Tsourkas A. Effect of ligand density, receptor density, and nanoparticle size on cell targeting. Nanomed Nanotech Biol Med. 2012;9:194–201.
Zheng X, Cheung LS, Schroeder JA, Jiang L, Zohar Y. Cell receptor and surface ligand density effects on dynamic states of adhering circulating tumor cells. Lab Chip. 2011;11(20):3431–9.
Yuan H, Zhang S. Effects of particle size and ligand density on the kinetics of receptor-mediated endocytosis of nanoparticles. Appl Phys Lett. 2010;96(3):0337041–3.
Gunawan RC, Auguste DT. The role of antibody synergy and membrane fluidity in the vascular targeting of immunoliposomes. Biomaterials. 2010;31(5):900–7.
Srivastava RAK, Ito H, Hess M, Srivastava N, Schonfeld G. Regulation of low density lipoprotein receptor gene expression in HepG2 and Caco2 cells by palmitate, oleate, and 25-hydroxycholesterol. J Lipid Research. 1995;36:1434–46.
Keck CM, Jansch M, Müller RH. Protein adsorption patterns and analysis on IV Nanoemulsions—The key factor determining the organ distribution. Pharmaceutics. 2013;5:36–68.
Acknowledgments and Disclosures
E. Markoutsa and K. Papadia equally contributed to this paper. The research leading to these results has received funding from the European Community's Seventh Framework Programme (FP7/2007–2013) under grant agreements n° 212043 (to SGA) and 260524 (to GTS). Authors are grateful to Dr. Pierre-Oliver Couraud (Inserm, Paris, FR) for providing the hCMEC/D3 cell line, and Dr. M Gregori (University Milano-Biccoca, Milan, IT) for her help in MAb thiolation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 125 kb)
Rights and permissions
About this article
Cite this article
Markoutsa, E., Papadia, K., Giannou, A.D. et al. Mono and Dually Decorated Nanoliposomes for Brain Targeting, In Vitro and In Vivo Studies. Pharm Res 31, 1275–1289 (2014). https://doi.org/10.1007/s11095-013-1249-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-013-1249-3