Abstract
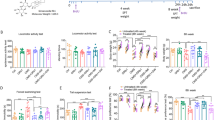
In recent years, the decline of microglia in the hippocampus has been shown to play a role in the development of depression, and its reversal shows marked antidepressant-like effects. β-glucan is a polysaccharide from Saccharomyces cerevisiae and has numerous beneficial effects on the nervous system, including improving axon regeneration and cognition. Considering its immuno-stimulatory activities in cultured microglia and brain tissues, we hypothesize that β-glucan may be a potential candidate to correct the functional deficiency of microglia and thereby alleviate depression-like behaviors in chronically stressed animals. An expected, our results showed that a single injection of β-glucan 5 h before behavioral tests at a dose of 10 or 20 mg/kg, but not at a dose of 5 mg/kg, reversed the depression-like behavior induced by chronic stress in mice in the tail suspension test, forced swimming test, and sucrose preference test. The effect of β-glucan (20 mg/kg) also showed time-dependent properties that were statistically significant 5 and 8, but not 3, hours after drug injection and persisted for at least 7 days. Fourteen days after β-glucan injection, no antidepressant-like effect was observed anymore. However, this effect was overcome by a second β-glucan injection (20 mg/kg) 14 days after the first β-glucan injection. Stimulation of microglia appeared to mediate the antidepressant-like effect of β-glucan, because both inhibition of microglia and their depletion prevented the antidepressant-like effect of β-glucan. Based on these effects of β-glucan, β-glucan administration could be developed as a new strategy for the treatment of depression.






Similar content being viewed by others
Data Availability
Data will be made available on request.
References
Patel D, Kas MJ, Chattarji S, Buwalda B (2019) Rodent models of social stress and neuronal plasticity: relevance to depressive-like disorders. Behav Brain Res 369:111900
Strekalova T, Liu Y, Kiselev D, Khairuddin S, Chiu JLY, Lam J, Chan YS, Pavlov D, Proshin A, Lesch KP, Anthony DC, Lim LW (2022) Chronic mild stress paradigm as a rat model of depression: facts, artifacts, and future perspectives. Psychopharmacology 239:663–693
Hadwen B, Pila E, Thornton J (2022) The associations between adverse childhood experiences, physical and Mental Health, and physical activity: a scoping review. J Phys Act Health 19:847–854
Crowe M, Inder M, Thwaites B (2023) The experience of mood disorder and substance use: an integrative review. J Psychiatr Ment Health Nurs 30:295–308
Afridi R, Suk K (2023) Microglial responses to stress-Induced Depression: causes and consequences. Cells 12:1521
Brisch R, Wojtylak S, Saniotis A, Steiner J, Gos T, Kumaratilake J, Henneberg M, Wolf R (2022) The role of microglia in neuropsychiatric disorders and suicide. Eur Arch Psychiatr Clin Neurosci 272:929–945
Kreisel T, Frank MG, Licht T, Reshef R, Ben-Menachem-Zidon O, Baratta MV, Maier SF, Yirmiya R (2014) Dynamic microglial alterations underlie stress-induced depressive-like behavior and suppressed neurogenesis. Mol Psychiatry 19:699–709
Tong L, Gong Y, Wang P, Hu W, Wang J, Chen Z, Zhang W, Huang C (2017) Microglia loss contributes to the development of Major Depression Induced by different types of chronic stresses. Neurochem Res 42:2698–2711
Gong Y, Tong L, Yang R, Hu W, Xu X, Wang W, Wang P, Lu X, Gao M, Wu Y, Xu X, Zhang Y, Chen Z, Huang C (2018) Dynamic changes in hippocampal microglia contribute to depressive-like behavior induced by early social isolation. Neuropharmacology 135:223–233
Hu P, Wang D, Zhang Y, Cai Z, Ye T, Tong L, Xu X, Lu J, Liu F, Lu X, Huang C (2020) Apoptosis-triggered decline in hippocampal microglia mediates adolescent intermittent alcohol exposure-induced depression-like behaviors in mice. Neuropharmacology 170:108054
Ye T, Wang D, Cai Z, Tong L, Chen Z, Lu J, Lu X, Huang C, Yuan X (2020) Antidepressive properties of macrophage-colony stimulating factor in a mouse model of depression induced by chronic unpredictable stress. Neuropharmacology 172:108132
Netea MG, Gow NA, Munro CA, Bates S, Collins C, Ferwerda G, Hobson RP, Bertram G, Hughes HB, Jansen T, Jacobs L, Buurman ET, Gijzen K, Williams DL, Torensma R, McKinnon A, MacCallum DM, Odds FC, Van der Meer JW, Brown AJ, Kullberg BJ (2006) Immune sensing of Candida albicans requires cooperative recognition of mannans and glucans by lectin and toll-like receptors. J Clin Invest 116:1642–1650
Kokoshis PL, Williams DL, Cook JA, Di Luzio NR (1978) Increased resistance to Staphylococcus aureus Infection and enhancement in serum lysozyme activity by glucan. Science 199:1340–1342
Williams DL, Cook JA, Hoffmann EO, Di Luzio NR (1978) Protective effect of glucan in experimentally induced candidiasis. J Reticuloendothel Soc 23:479–490
Itoh W, Sugawara I, Kimura S, Tabata K, Hirata A, Kojima T, Mori S, Shimada K (1990) Immunopharmacological study of sulfated schizophyllan (SPG). I.—Its action as a mitogen and anti-HIV agent. Int J Immunopharmacol 12:225–233
Masuda Y, Togo T, Mizuno S, Konishi M, Nanba H (2012) Soluble β-glucan from Grifola frondosa induces proliferation and Dectin-1/Syk signaling in resident macrophages via the GM-CSF autocrine pathway. J Leukoc Biol 91:547–556
Baldwin KT, Carbajal KS, Segal BM, Giger RJ (2015) Neuroinflammation triggered by β-glucan/dectin-1 signaling enables CNS axon regeneration. Proc Natl Acad Sci 112:2581–2586
Zhang Q, Zhao W, Hou Y, Song X, Yu H, Tan J, Zhou Y, Zhang HT (2023) β-Glucan attenuates cognitive impairment of APP/PS1 mice via regulating intestinal flora and its metabolites. CNS Neurosci Ther 29:1690–1704
Xu M, Mo X, Huang H, Chen X, Liu H, Peng Z, Chen L, Rong S, Yang W, Xu S, Liu L (2020) Yeast β-glucan alleviates cognitive deficit by regulating gut microbiota and metabolites in Aβ1–42-induced AD-like mice. Int J Biol Macromol 161:258–270
Pan W, Jiang P, Zhao J, Shi H, Zhang P, Yang X, Biazik J, Hu M, Hua H, Ge X, Huang XF, Yu Y (2021) β-Glucan from Lentinula edodes prevents cognitive impairments in high-fat diet-induced obese mice: involvement of colon-brain axis. J Transl Med 19:54
Shi H, Yu Y, Lin D, Zheng P, Zhang P, Hu M, Wang Q, Pan W, Yang X, Hu T, Li Q, Tang R, Zhou F, Zheng K, Huang XF (2020) β-glucan attenuates cognitive impairment via the gut-brain axis in diet-induced obese mice. Microbiome 8:143
Hu M, Zhang P, Wang R, Zhou M, Pang N, Cui X, Ge X, Liu X, Huang XF, Yu Y (2022) Three different types of β-Glucans enhance cognition: the role of the gut-brain Axis. Front Nutr 9:848930
Shah VB, Huang Y, Keshwara R, Ozment-Skelton T, Williams DL, Keshvara L (2008) Beta-glucan activates microglia without inducing cytokine production in Dectin-1-dependent manner. J Immunol 180:2777–2785
Heng Y, Zhang X, Borggrewe M, van Weering HRJ, Brummer ML, Nijboer TW, Joosten LAB, Netea MG, Boddeke EWGM, Laman JD, Eggen BJL (2021) Systemic administration of β-glucan induces immune training in microglia. J Neuroinflammation 18:57
Viau V, Bingham B, Davis J, Lee P, Wong M (2005) Gender and puberty interact on the stress-induced activation of parvocellular neurosecretory neurons and corticotropin-releasing hormone messenger ribonucleic acid expression in the rat. Endocrinology 146:137–146
Heinsbroek RP, Van Haaren F, Van de Poll NE, Steenbergen HL (1991) Sex differences in the behavioral consequences of inescapable footshocks depend on time since shock. Physiol Behav 49:1257–1263
Bollinger JL, Bergeon Burns CM, Wellman CL (2016) Differential effects of stress on microglial cell activation in male and female medial prefrontal cortex. Brain Behav Immun 52:88–97
Wang Y, Hu Z, Liu H, Gu Y, Ye M, Lu Q, Lu X, Huang C (2022) Adolescent microglia stimulation produces long-lasting protection against chronic stress-induced behavioral abnormalities in adult male mice. Brain Behav Immun 105:44–66
Gu Y, Ye T, Tan P, Tong L, Ji J, Gu Y, Shen Z, Shen X, Lu X, Huang C (2021) Tolerance-inducing effect and properties of innate immune stimulation on chronic stress-induced behavioral abnormalities in mice. Brain Behav Immun 91:451–471
Lu X, Liu H, Cai Z, Hu Z, Ye M, Gu Y, Wang Y, Wang D, Lu Q, Shen Z, Shen X, Huang C (2022) ERK1/2-dependent BDNF synthesis and signaling is required for the antidepressant effect of microglia stimulation. Brain Behav Immun 106:147–160
Cai Z, Ye T, Xu X, Gao M, Zhang Y, Wang D, Gu Y, Zhu H, Tong L, Lu J, Chen Z, Huang C (2020) Antidepressive properties of microglial stimulation in a mouse model of depression induced by chronic unpredictable stress. Prog Neuropsychopharmacol Biol Psychiatry 101:109931
Voronova IP, Khramova GM, Kulikova EA, Petrovskii DV, Bazovkina DV, Kulikov AV (2016) 5-HT2A receptors control body temperature in mice during LPS-induced inflammation via regulation of NO production. Pharmacol Res 103:123–131
Dantzer R, Bluthé RM, Layé S, Bret-Dibat JL, Parnet P, Kelley KW (1998) Cytokines and sickness behavior. Ann N Y Acad Sci 840:586–590
Yirmiya R, Pollak Y, Morag M, Reichenberg A, Barak O, Avitsur R, Shavit Y, Ovadia H, Weidenfeld J, Morag A, Newman ME, Pollmächer T (2000) Illness, cytokines, and depression. Ann N Y Acad Sci 917:478–487
Xiao R, Zeng J, Bressler EM, Lu W, Grinstaff MW (2022) Synthesis of bioactive (1→6)-β-glucose branched poly-amido-saccharides that stimulate and induce M1 polarization in macrophages. Nat Commun 13:4661
Shui Y, Hu X, Hirano H, Kusano K, Tsukamoto H, Li M, Hasumi K, Guo WZ, Li XK (2021) β-glucan from Aureobasidium pullulans augments the anti-tumor immune responses through activated tumor-associated dendritic cells. Int Immunopharmacol 101:108265
Stanca S, Rossetti M, Bongioanni P (2023) Astrocytes as Neuroimmunocytes in Alzheimer’s Disease: a biochemical Tool in the Neuron-Glia Crosstalk along the pathogenetic pathways. Int J Mol Sci 24:13880
Duarte N, Shafi AM, Penha-Gonçalves C, Pais TF (2023) Endothelial type I interferon response and brain Diseases: identifying STING as a therapeutic target. Front Cell Dev Biol 11:1249235
Liston A, Dooley J, Yshii L (2022) Brain-resident regulatory T cells and their role in health and Disease. Immunol Lett 248:26–30
Neckles VN, Feliciano DM (2022) From seed to flower: blossoming of microglia in development and brain repair. Cell Tissue Res 387:377–389
Yu CJ, Wang M, Li RY, Wei T, Yang HC, Yin YS, Mi YX, Qin Q, Tang Y (2023) TREM2 and Microglia Contribute to the synaptic plasticity: from Physiology to Pathology. Mol Neurobiol 60:512–523
Pascual O, Ben Achour S, Rostaing P, Triller A, Bessis A (2012) Microglia activation triggers astrocyte-mediated modulation of excitatory neurotransmission. Proc Natl Acad Sci U S A 109:E197–E205
Shinozaki Y, Nomura M, Iwatsuki K, Moriyama Y, Gachet C, Koizumi S (2014) Microglia trigger astrocyte-mediated neuroprotection via purinergic gliotransmission. Sci Rep 4:4329
Delpech JC, Saucisse N, Parkes SL, Lacabanne C, Aubert A, Casenave F, Coutureau E, Sans N, Layé S, Ferreira G, Nadjar A (2015) Microglial activation enhances associative taste memory through purinergic modulation of glutamatergic neurotransmission. J Neurosci 35:3022–3033
Zhang D, Jia Y, Sun X, Li H, Yin M, Li H, Dai L, Han L, Wang L, Qian M, Du J, Zhu K, Bao H (2022) The Dectin-1 receptor signaling pathway mediates the remyelination effect of lentinan through suppression of neuroinflammation and conversion of microglia. J Immunol Res 2022:3002304
Suzuki A, Hara H, Kimura H (2023) Role of the AMPA receptor in antidepressant effects of ketamine and potential of AMPA receptor potentiators as a novel antidepressant. Neuropharmacology 222:109308
Yang PS, Peng HY, Lin TB, Hsieh MC, Lai CY, Lee AS, Wang HH, Ho YC (2020) NMDA receptor partial agonist GLYX-13 alleviates chronic stress-induced depression-like behavior through enhancement of AMPA receptor function in the periaqueductal gray. Neuropharmacology 178:108269
Bao H, Sun L, Zhu Y, Ran P, Hu W, Zhu K, Li B, Hou Y, Nie J, Gao T, Shan L, Du K, Zheng S, Zheng B, Xiao C, Du J (2017) Lentinan produces a robust antidepressant-like effect via enhancing the prefrontal Dectin-1/AMPA receptor signaling pathway. Behav Brain Res 317:263–271
Bao H, Ran P, Zhu M, Sun L, Li B, Hou Y, Nie J, Shan L, Li H, Zheng S, Xu X, Xiao C, Du J (2016) The Prefrontal Dectin-1/AMPA receptor signaling pathway mediates the robust and prolonged antidepressant effect of Proteo-β-Glucan from Maitake. Sci Rep 6:28395
Acknowledgements
The authors would kindly like to thank Dr Yiming Gu for his initial contribution to the construction of the depression model.
Funding
This work was supported by the Natural Science Foundation of Jiangsu Province (BK20221375 and BK20230288), the Nantong Health Commission (QN2023035 and MS2023033), the Six Phase of Jiangsu 333 High-level Talent Training Project (2022: 3-16-661), and the Natural Science Foundation of the Higher Education Institutions of Jiangsu Province (23KJB310020).
Author information
Authors and Affiliations
Contributions
CZ: Methodology, Software, Investigation, Formal analysis, Writing—original draft. ZC: Methodology, Software. QL: Methodology, Software, Investigation, Formal analysis, Data curation. XL: Methodology, Resources, Supervision. WH: Methodology, Software. Rongrong Yang: Methodology, Software. BC: Methodology, Software. CH: Conceptualization, Resources, Supervision. All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, C., Chen, Z., Lu, X. et al. Microglia-Dependent Reversal of Depression-Like Behaviors in Chronically Stressed Mice by Administration of a Specific Immuno-stimulant β-Glucan. Neurochem Res 49, 519–531 (2024). https://doi.org/10.1007/s11064-023-04056-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-023-04056-x