Abstract
Purpose
Distinguishing between primary central nervous system lymphoma (PCNSL) and isocitrate dehydrogenase (IDH)-wildtype glioblastoma is important for therapeutic decision-making. This study aimed to compare the performance of 11C-methionine (MET) and 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET) for distinguishing between these two major malignant brain tumors.
Methods
We retrospectively conducted qualitative and semiquantitative analyses of pre-treatment MET and FDG PET/computed tomography (CT) images of 22 patients with PCNSL and 64 patients with IDH-wildtype glioblastoma. For semiquantitative analysis, we calculated the tumor-to-normal tissue (T/N) ratio by dividing the maximum standardized uptake value (SUV) for the tumor (T) by the average SUV for the normal tissue (N). For performance evaluation, we employed receiver operating characteristic curve analysis and calculated the areas under the curve (AUC) values.
Results
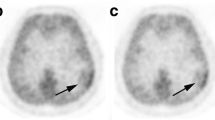
In the qualitative analysis, all PCNSLs and IDH-wildtype glioblastomas were MET-positive, while 95% and 84% of PCNSLs and IDH-wildtype glioblastomas, respectively, were FDG-positive. Eleven patients were excluded from the FDG PET/CT semiquantitative analysis because of hyperglycemia. There was no difference in MET T/N ratio between PCNSL and IDH-wildtype glioblastoma (p = 0.37). FDG T/N ratio was significantly higher in PCNSL than in IDH-wildtype glioblastoma (p < 0.001). The AUC value for distinguishing PCNSL from IDH-wildtype glioblastoma was significantly higher for the FDG T/N ratio (0.871) than for the MET T/N ratio (0.565) (p = 0.0027).
Conclusion
MET PET could detect both PCNSL and IDH-wildtype glioblastoma, but unlike FDG PET, it could not distinguish between these two major malignant brain tumors.




Similar content being viewed by others
Data availability
Data will be made available by the corresponding author upon reasonable request.
References
Ostrom QT, Price M, Neff C, Cioffi G, Waite KA, Kruchko C, Barnholtz-Sloan JS (2023) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2016–2020. Neuro Oncol 25:iv1–iv99. https://doi.org/10.1093/neuonc/noad149
Bühring U, Herrlinger U, Krings T, Thiex R, Weller M, Küker W (2001) MRI features of primary central nervous system lymphomas at presentation. Neurology 57:393–396. https://doi.org/10.1212/wnl.57.3.393
Ferreri AJ, Reni M (2007) Primary central nervous system lymphoma. Crit Rev Oncol Hematol 63(3):257–268. https://doi.org/10.1016/j.critrevonc.2007.04.012
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups, National Cancer Institute of Canada Clinical Trials Group (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996. https://doi.org/10.1056/NEJMoa043330
Verger A, Kas A, Darcourt J, Guedj E (2022) PET imaging in neuro-oncology: an update and overview of a rapidly growing area. Cancers 14(5):1103. https://doi.org/10.3390/cancers14051103
Chen W, Silverman DH (2008) Advances in evaluation of primary brain tumors. Semin Nucl Med 38:240–250. https://doi.org/10.1053/j.semnuclmed.2008.02.005
Kosaka N, Tsuchida T, Uematsu H, Kimura H, Okazawa H, Itoh H (2008) 18F-FDG PET of common enhancing malignant brain tumors. AJR Am J Roentgenol 190:W365–W369. https://doi.org/10.2214/AJR.07.2660
Yamashita K, Yoshiura T, Hiwatashi A et al (2013) Differentiating primary CNS lymphoma from glioblastoma multiforme: assessment using arterial spin labeling, diffusion-weighted imaging and 18F-fluorodeoxyglucose positron emission tomography. Neuroradiology 55:135–143. https://doi.org/10.1007/s00234-012-1089-6
Makino K, Hirai T, Nakamura H, Murakami R, Kitajima M, Shigematsu Y, Nakashima R, Shiraishi S, Uetani H, Iwashita K, Akter M, Yamashita Y, Kuratsu J (2011) Does adding FDG-PET to MRI improve the differentiation between primary cerebral lymphoma and glioblastoma? Observer performance study. Ann Nucl Med 25(6):432–438. https://doi.org/10.1007/s12149-011-0483-1
Hatakeyama J, Ono T, Takahashi M, Oda M, Shimizu H (2021) Differentiating between primary central nervous system lymphoma and glioblastoma: the diagnostic value of combining 18F-fluorodeoxyglucose positron emission tomography with arterial spin labeling. Neurol Med Chir 61(6):367–375. https://doi.org/10.2176/nmc.oa.2020-0375
Gupta T, Manjali JJ, Kannan S, Purandare N, Rangarajan V (2021) Diagnostic performance of pretreatment 18F-fluorodeoxyglucose positron emission tomography with or without computed tomography in patients with primary central nervous system lymphoma: updated systematic review and diagnostic test accuracy meta-analyses. Clin Lymphoma Myeloma Leuk 21(8):497–507. https://doi.org/10.1016/j.clml.2021.03.011
Rosenfeld SS, Hoffman JM, Coleman RE, Glantz MJ, Hanson MW, Schold SC (1992) Studies of primary central nervous system lymphoma with fluorine-18-fluorodeoxyglucose positron emission tomography. J Nucl Med 33:532–536
Dunet V, Pomoni A, Hottinger A, Nicod-Lalonde M, Prior JO (2016) Performance of 18F-FET versus 18F-FDG-PET for the diagnosis and grading of brain tumors: systematic review and meta-analysis. Neuro Oncol 18(3):426–434. https://doi.org/10.1093/neuonc/nov148
Pafundi DH, Laack NN, Youland RS, Parney IF, Lowe VJ, Giannini C, Kemp BJ, Grams MP, Morris JM, Hoover JM, Hu LS, Sarkaria JN, Brinkmann DH (2013) Biopsy validation of 18F-DOPA PET and biodistribution in gliomas for neurosurgical planning and radiotherapy target delineation: results of a prospective pilot study. Neuro Oncol 15(8):1058–1067. https://doi.org/10.1093/neuonc/not002
Law I, Albert NL, Arbizu J et al (2019) Joint EANM/EANO/RANO practice guidelines SNMMI procedure standards for imaging of gliomas using PET with radiolabelled amino acids and [18F]FDG, version 1.0. Eur J Nucl Med Mol Imaging 46:540–557
Ninatti G, Pini C, Gelardi F, Sollini M, Chiti A (2023) The role of PET imaging in the differential diagnosis between radiation necrosis and recurrent disease in irradiated adult-type diffuse gliomas: a systematic review. Cancers 15:364. https://doi.org/10.3390/cancers15020364
Kim YI, Kim Y, Lee JY, Jang SJ (2018) Prognostic value of the metabolic and volumetric parameters of 11C-methionine positron-emission tomography for gliomas: a systematic review and meta-analysis. AJNR Am J Neuroradiol 39:1629–1634. https://doi.org/10.3174/ajnr.A5707
Ogawa T, Kanno I, Hatazawa J et al (1994) Methionine PET for follow-up of radiation therapy of primary lymphoma of the brain. Radiographics 14:101–110. https://doi.org/10.1148/radiographics.14.1.8128041
Kawase Y, Yamamoto Y, Kameyama R, Kawai N, Kudomi N, Nishiyama Y (2011) Comparison of 11C-methionine PET and 18F-FDG PET in patients with primary central nervous system lymphoma. Mol Imaging Biol 13:1284–1289. https://doi.org/10.1007/s11307-010-0447-1
Miyakita Y, Ohno M, Takahashi M, Kurihara H, Katai H, Narita Y (2020) Usefulness of carbon-11-labeled methionine positron-emission tomography for assessing the treatment response of primary central nervous system lymphoma. Jpn J Clin Oncol 50:512–518. https://doi.org/10.1093/jjco/hyaa010
Ahn SY, Kwon SY, Jung SH et al (2018) Prognostic significance of interim 11C-methionine PET/CT in primary central nervous system lymphoma. Clin Nucl Med 43:e259–e264. https://doi.org/10.1097/RLU.0000000000002154
Ishiwata K, Ido T, Vaalburg W (1988) Increased amounts of D-enantiomer dependent on alkaline concentration in the synthesis of L-[methyl-11C] methionine. Int J Rad Appl Instrum A 39:311–314. https://doi.org/10.1016/0883-2889(88)90021-4
Spence AM, Muzi M, Mankoff DA, O’Sullivan SF, Link JM, Lewellen TK, Lewellen B, Pham P, Minoshima S, Swanson K, Krohn KA (2004) 18F-FDG PET of gliomas at delayed intervals: improved distinction between tumor and normal gray matter. J Nucl Med 45(10):1653–1659
Chiavazza C, Pellerino A, Ferrio F, Cistaro A, Soffietti R, Rudà R (2018) Primary CNS lymphomas: challenges in diagnosis and monitoring. BioMed Res Int 2018:3606970. https://doi.org/10.1155/2018/3606970
Stegmayr C, Stoffels G, Kops ER, Lohmann P, Galldiks N, Shah NJ, Neumaier B, Langen KJ (2019) Influence of dexamethasone on O-(2-[18F]-fluoroethyl)-L-tyrosine uptake in the human brain and quantification of tumor uptake. Mol Imaging Biol 21(1):168–174. https://doi.org/10.1007/s11307-018-1221-z
Jo J-C, Yoon DH, Kim S, Lee K, Kang EH, Park JS, Ryu J-S, Huh J, Park C-S, Kim JH et al (2017) Interim 18F-FGD PET/CT may not predict the outcome in primary central nervous system lymphoma patients treated with sequential treatment with methotrexate and cytarabine. Ann Hematol 96:1509–1515
Birsen R, Blanc E, Willems L, Burroni B, Legoff M, Le Ray E, Pilorge S, Salah S, Quentin A, Deau B et al (2018) Prognostic value of early 18F-FDG PET scanning evaluation in immunocompetent primary CNS lymphoma patients. Oncotarget 9:16822–16831
Rozenblum L, Galanaud D, Houillier C, Soussain C, Baptiste A, Belin L, Edeline V, Naggara P, Soret M, Causse-Lemercier V, Willems L, Choquet S, Ursu R, Hoang-Xuan K, Kas A (2023) [18F]FDG PET-MRI provides survival biomarkers in primary central nervous system lymphoma in the elderly: an ancillary study from the BLOCAGE trial of the LOC network. Eur J Nucl Med Mol Imaging 50(12):3684–3696. https://doi.org/10.1007/s00259-023-06334-w
Acknowledgements
Not applicable.
Funding
YY is currently receiving a Grant-in-Aid for Scientific Research (KAKENHI) from the Japan Society for the Promotion of Science (grant number 23K07085). For the remaining authors, there are no funding.
Author information
Authors and Affiliations
Contributions
All authors contributed to the concept and design of the study. The study was designed by TN and YY. Material preparation, data collection, and analysis were performed by TN, KM, YM, MM, HAO, TH and KM. The first draft of the manuscript was written by TN and reviewed by YY and YN. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Ethical approval
This study was approved by the Ethics Committee of the Faculty of Medicine, Kagawa University (approval numbers: 2023-146).
Informed consent
The requirement for obtaining written informed consent was waived owing to the retrospective study design.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Norikane, T., Mitamura, K., Yamamoto, Y. et al. Comparative evaluation of 11C-methionine and 18F-fluorodeoxyglucose positron emission tomography for distinguishing between primary central nervous system lymphoma and isocitrate dehydrogenase-wildtype glioblastoma. J Neurooncol 166, 195–201 (2024). https://doi.org/10.1007/s11060-023-04534-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04534-w