Abstract
Purpose
The aim of this study is to investigate the association between postoperative tumor volume and overall survival (OS) of O6-methylguanine DNA methyltransferase (MGMT)-unmethylated glioblastoma patients.
Methods
One hundred-twenty-six patients with MGMT-unmethylated glioblastoma who were treated either with surgical resection or needle biopsy between 2006 and 2015 were included in this retrospective cohort. Pre- and postcontrast T1 weighted images were evaluated in order to determine pre- and postoperative contrast-enhancing tumor volumes (CE-TV). Cox regression models adjusted for other significant prognostic factors were used to investigate the association between postoperative tumor volume and survival.
Results
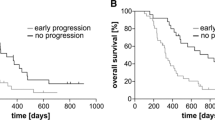
Complete resection of CE-TV was significantly associated with longer OS in the univariate analysis (HR 0.61; 95% CI 0.40–0.94; p = 0.02). However, this fact could not be confirmed after adjusting the model for other relevant prognostic factors (HR 1.01; 95% CI 0.65–1.55; p = 0.962). Postoperative CE-TV was significantly associated with survival in both univariate (HR: 1.04; 95% CI 1.025–1.055; p < 0.001) and multivariate analyses (HR: 1.027; 95% CI 1.005–1.049; p = 0.014).
Conclusions
Although complete resection of tumor tissue was not significantly associated with longer OS in MGMT-unmethylated GBM patients, maximum safe resection should always be attempted, since postoperative tumor volume is strongly associated with OS.


Similar content being viewed by others
References
Wick W, Weller M, Van Den Bent M, Sanson M, Weiler M, Von Deimling A, Plass C, Hegi M, Platten M, Reifenberger G (2014) MGMT testing-the challenges for biomarker-based glioma treatment. Nat Rev Neurol 10:372–385
Donson AM, Addo-Yobo SO, Handler MH et al (2007) MGMT promoter methylation correlates with survival benefit and sensitivity to temozolomide in pediatric glioblastoma. Pediatr Blood Cancer 48:403–407. https://doi.org/10.1002/pbc.20803
Schaub C, Schäfer N, Mack F et al (2016) The earlier the better? Bevacizumab in the treatment of recurrent MGMT-non-methylated glioblastoma. J Cancer Res Clin Oncol 142:1825–1829. https://doi.org/10.1007/s00432-016-2187-3
Binabaj MM, Bahrami A, ShahidSales S et al (2018) The prognostic value of MGMT promoter methylation in glioblastoma: a meta-analysis of clinical trials. J Cell Physiol 233:378–386. https://doi.org/10.1002/jcp.25896
Everhard S, Kaloshi G, Crinière E et al (2006) MGMT methylation: a marker of response to temozolomide in low-grade gliomas. Ann Neurol 60:740–743. https://doi.org/10.1002/ana.21044
Hegi ME, Diserens A-C, Godard S et al (2004) Clinical trial substantiates the predictive value of O-6-methylguanine-DNA methyltransferase promoter methylation in glioblastoma patients treated with temozolomide. Clin Cancer Res 10:1871–1874
Taylor JW, Schiff D (2015) Treatment considerations for MGMT-unmethylated glioblastoma. Curr Neurol Neurosci Rep 15:507. https://doi.org/10.1007/s11910-014-0507-z
Hegi ME, Diserens A-C, Gorlia T et al (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003. https://doi.org/10.1056/NEJMoa043331
England TN (2000) Numb Er 19 response of gliomas to alkylating agents. Methods 343:4–8
Kitange GJ, Carlson BL, Schroeder MA et al (2009) Induction of MGMT expression is associated with temozolomide resistance in glioblastoma xenografts. Neuro Oncol 11:281–291. https://doi.org/10.1215/15228517-2008-090
Sanai N, Polley M-Y, McDermott MW et al (2011) An extent of resection threshold for newly diagnosed glioblastomas. J Neurosurg 115:3–8. https://doi.org/10.3171/2011.2.JNS10998
Oppenlander ME, Wolf AB, Snyder LA et al (2014) An extent of resection threshold for recurrent glioblastoma and its risk for neurological morbidity. J Neurosurg 120:846–853. https://doi.org/10.3171/2013.12.JNS13184
Bloch O, Han SJ, Cha S et al (2012) Impact of extent of resection for recurrent glioblastoma on overall survival. J Neurosurg 117:1032–1038. https://doi.org/10.3171/2012.9.JNS12504
Chaichana KL, Cabrera-Aldana EE, Jusue-Torres I et al (2014) When gross total resection of a glioblastoma is possible, how much resection should be achieved? World Neurosurg 82:e257–e265. https://doi.org/10.1016/j.wneu.2014.01.019
Orringer D, Lau D, Khatri S et al (2012) Extent of resection in patients with glioblastoma: limiting factors, perception of resectability, and effect on survival. J Neurosurg 117:851–859. https://doi.org/10.3171/2012.8.JNS12234
Kreth FW, Thon N, Simon M et al (2013) Gross total but not incomplete resection of glioblastoma prolongs survival in the era of radiochemotherapy. Ann Oncol 24:3117–3123. https://doi.org/10.1093/annonc/mdt388
Bette S, Barz M, Wiestler B et al (2018) Prognostic value of tumor volume in glioblastoma patients: size also matters for patients with incomplete resection. Ann Surg Oncol 25:558–564. https://doi.org/10.1245/s10434-017-6253-0
Chaichana KL, Jusue-Torres I, Navarro-Ramirez R et al (2014) Establishing percent resection and residual volume thresholds affecting survival and recurrence for patients with newly diagnosed intracranial glioblastoma. Neuro Oncol 16:113–122. https://doi.org/10.1093/neuonc/not137
Grabowski MM, Recinos PF, Nowacki AS et al (2014) Residual tumor volume versus extent of resection: predictors of survival after surgery for glioblastoma. J Neurosurg 121:1115–1123. https://doi.org/10.3171/2014.7.JNS132449
Mayhew TM, Olsen DR (1991) Magnetic resonance imaging (MRI) and model-free estimates of brain volume determined using the Cavalieri principle. J Anat 178:133–144
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. https://doi.org/10.1056/NEJMoa043330
Kebir S, Stummer W, Steinbach JP et al (2017) P16.11 complete resection is not associated with improved survival in MGMT non-methylated glioblastoma. Results from the GLARIUS trial. Neuro Oncol 19:iii112–iii112. https://doi.org/10.1093/neuonc/nox036.427
Delgado-López PD, Corrales-García EM (2016) Survival in glioblastoma: a review on the impact of treatment modalities. Clin Transl Oncol 18:1062–1071. https://doi.org/10.1007/s12094-016-1497-x
Hadjipanayis CG, Widhalm G, Stummer W (2015) What is the surgical benefit of utilizing 5-aminolevulinic acid for fluorescence-guided surgery of malignant gliomas? Neurosurgery 77:663–673. https://doi.org/10.1227/NEU.0000000000000929
Suchorska B, Weller M, Tabatabai G et al (2016) Complete resection of contrast-enhancing tumor volume is associated with improved survival in recurrent glioblastoma—results from the DIRECTOR trial. Neuro Oncol 18:549–556. https://doi.org/10.1093/neuonc/nov326
Li YM, Suki D, Hess K, Sawaya R (2016) The influence of maximum safe resection of glioblastoma on survival in 1229 patients: can we do better than gross-total resection? J Neurosurg 124:977–988. https://doi.org/10.3171/2015.5.JNS142087
Haj A, Doenitz C, Schebesch KM et al (2018) Extent of resection in newly diagnosed glioblastoma: impact of a specialized neuro-oncology care center. Brain Sci 8:1–10. https://doi.org/10.3390/brainsci8010005
Chen L, Mao Y (2014) Gross total resection plays a leading role in survival of patients with glioblastoma multiforme. World Neurosurg 82:e105–e107. https://doi.org/10.1016/j.wneu.2014.04.074
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Arthur H.A. Sales, Stefanie Bette, Melanie Barz, Thomas Huber, Benedikt Wiestler, Yu-Mi Ryang, Stephanie E.Combs, Friederike Schmidt-Graf, Bernhard Meyer, and Jens Gempt: SB, JG, and BM work as consultants for Brainlab (Brainlab AG, Munich). TH works as consultant for Smart Reporting GmbH (Munich, Germany).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sales, A.H.A., Bette, S., Barz, M. et al. Role of postoperative tumor volume in patients with MGMT-unmethylated glioblastoma. J Neurooncol 142, 529–536 (2019). https://doi.org/10.1007/s11060-019-03124-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-019-03124-z