Abstract
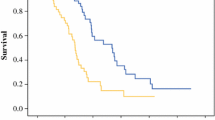
To evaluate the prognostic value of fluid-attenuated inversion recovery (FLAIR) signal intensity of postoperative cavity on progression free survival (PFS) and overall survival (OS) in patients with high-grade gliomas (HGG). This study retrospectively enrolled 45 consecutive HGG patients. These patients had chemoradiotherapy after gross-total resection of tumors. Quantitative analysis of the FLAIR signal intensity in postoperative cavity and background was made. We evaluated the threshold value, accuracy, sensitivity, specificity, and survival state with this technique. The patients who progressed and patients who did not progress were 33 and 12 cases separately. The ratio of postoperative cavity and background (C–B) on FLAIR sequence in patients who progressed was higher than that of patients who did not progress (P = 0.014). The PFS of the patients who progressed was shorter than that of patients who did not progress (P = 0.008). The area under ROC curve, threshold, sensitivity, specificity of C–B ratio for predicting tumor progression were 0.875, 62.3, 69.7, 0.84, and 0.50% respectively. The PFS of lower signal group was much longer than that of higher signal group (P = 0.004). The OS of the patients with higher signal was shorter than that of patients with lower signal (P = 0.034). The increase of gray value of FLAIR in postoperative cavity may be used as an imaging marker for predicting tumor progression.






Similar content being viewed by others
References
Huber T, Bette S, Wiestler B et al (2016) Fractional anisotropy correlates with overall survival in glioblastioma. World Neurosurg 95:525–534
Majós C, Cos M, Castañer S et al (2016) Early post-operative magnetic resonance imaging in glioblastoma: correlation among radiological findings and overall survival in 60 patients. Eur Radiol 26:1048–1055
Chang PD, Chow DS, Yang PH, Filippi CG, Lignelli A (2017) Predicting glioblastoma recurrence by early changes in the apparent diffusion coefficient value and signal intensity on FLAIR images. AJR Am J Roentgenol 208:57–65
Roldan-Valadez E, Rios C, Motola-Kuba D, Matus-Santos J, Villa AR, Moreno-Jimenez S (2016) Choline-to-N-acetyl aspartate and lipids-lactate-to-creatine ratios together with age assemble a significant Cox’s proportional-hazards regression model for prediction of survival in high-grade gliomas. Br J Radiol 89(1067):20150502
Kolakshyapati M, Adhikari RB, Karlowee V et al (2017) Nonenhancing peritumoral hyperintense lesions on diffusion-weighted imaging in glioblastoma; a novel diagnostic and specific prognostic indicator. J Neurosurg 31:1–12
Kim R, Choi SH, Yun TJ et al (2017) Prognosis prediction of non-enhancing T2 high signal intensity lesions in glioblastoma patients after standard treatment: application of dynamic contrast-enhanced MR imaging. Eur Radiol 27:1176–1185
Leiva-Salinas C, Schiff D, Flors L, Patrie JT, Rehm PK (2017) FDG PET/MR imaging coregistration helps predict survival in patients with gliobastoma and radiologic progression after standard of care treatment. Radiology 283:508–514
Bette S, Huber T, Gempt J et al (2017) Local fractional anisotropy is reduced in areas with tumor recurrence in glioblastoma. Radiology 283:499–507
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972
Cui Y, Tha KK, Terasaka S et al (2016) Prognostic imaging biomarkers in glioblastoma: development and independent validation o the basis of multiregion and quantitative analysis of MR images. Radiology 278:546–553
Çoban G, Mohan S, Kural F, Wang S, O’Rourke DM, Poptani H (2015) Prognostic value of dynamic susceptibility contrast-enhanced and diffusion-weighted MR imaging in patients with glioblastomas. AJNR Am J Neuroradiol 36:1247–1252
Kim HS, Kim JH, Kim SH, Cho KG, Kim SY (2010) Posttreatment high-grade glioma: usefulness of peak height position with semiquantitative MR perfusion histogram analysis in an entire contrast-enhanced lesion for predicting volume fraction of recurrence. Radiology 256:906–915
Winterstein M, Münter MW, Burkholder I, Essig M, Kauczor HU, Weber MA (2010) Partially resected glioma: diagnostic performance of fluid-attenuated inversion recovery MR imaging for detection of progression. Radiology 254:907–916
Sarbu N, Oleaga L, Valduvieco I, Pujol T, Berenguer J (2016) Increased signal intensity in FLAIR sequences in resection cavity can predict progression and progression-free survival in glioma. Neurocirugia 27:269–276
Ito-Yamashita T, Nakasu Y, Mitsuya K, Mizokami Y, Namba H (2013) Detection of tumor progression by signal increase on fluid-attenuated inversion recovery magnetic resonance images in the resection cavity of high-grade glioma. Neurol Med Chir 53:496–500
Mori H, Kunimatsu A, Abe Oet al (2012) Diagnostic ability of fluid-attenuated inversion recovery MR imaging to detect remnant or recurrent meningiomas after resection. Neuroradiol J 25:163–171
Poussaint TY, Vajapeyam S, Ricci KI et al (2016) Apparent diffusion coefficient histogram metrics correlate with survival in diffuse intrinsic pontine glioma: a report from the Pediatric Brain Tumor Consortium. Neuro Oncol 18:725–734
Han K, Ren M, Wick W et al (2014) Progression-free survival as a surrogate endpoint for overall survival in glioblastoma: a literature-based meta-analysis from 91 trials. Neuro Oncol 16:696–706
Kim BS, Kim SK, Choi SH et al (2015) Prognostic implication of progression pattern after anti-VEGF bevacizumab treatment for recurrent malignant gliomas. J Neurooncol 124:101–110
Dalesandro MF, Andre JB (2016) Posttreatment evaluation of brain gliomas. Neuroimaging Clin N Am 26:581–599
Bette S, Gempt J, Huber T et al (2016) FLAIR signal increase of the fluid within the resection cavity after glioma surgery: generally valid as early recurrence marker? J Neurosurg 21:1–9
Chang K, Zhang B, Guo X et al (2016) Multimodal imaging patterns predict survival in recurrent glioblastoma patients treated with bevacizumab. Neuro Oncol 18:1680–1687
Park JE, Kim HS, Park KJ, Kim SJ, Kim JH, Smith SA (2016) Pre- and posttreatment glioma: comparison of amide proton transfer imaging with MR spectroscopy for biomarkers of tumor proliferation. Radiology 278:514–523
Zhou J, Tryggestad E, Wen Z et al (2011) Differentiation between glioma and radiation necrosis using molecular magnetic resonance imaging of endogenous proteins and peptides. Nat Med 17:130–17134
Toyonaga T, Yamaguchi S, Hirata K et al (2017) Hypoxic glucose metabolism in glioblastoma as potential prognostic factor. Eur J Nucl Med Mol Imaging 44:611–619
Piroth MD, Galldiks N, Pinkawa M et al (2016) Relapse patterns after radiochemotherapy of glioblastoma with FET PET-guided boost irradiation and simulation to optimize radiation target volume. Radiat Oncol 11:87–96
Kil WJ, Tofilon PJ, Camphausen K (2012) Post-radiation increase in VEGF enhances glioma cell motility in vitro. Radiat Oncol 22:7–25
Neill E, Luks T, Dayal M et al (2017) Quantitative multi-modal MR imaging as a non-invasive prognostic tool for patients with recurrent low-grade glioma. J Neurooncol 132:171–179
Hsu CC, Watkins TW, Kwan GN, Haacke EM (2016) Susceptibility-weighted imaging of glioma: update on current imaging status and future directions. J Neuroimaging 26:383–390
McDonald RJ, McDonald JS, Dai D et al (2017) Comparison of gadolinium concentrations within multiple rat organs after intravenous administration of linear versus macrocyclic gadolinium chelates. Radiology. https://doi.org/10.1148/radiol.2017161594
Dekkers IA, Roos R, van der Molen AJ (2017) Gadolinium retention after administration of contrast agents based on linear chelators and the recommendations of the European Medicines Agency. Eur Radiol. https://doi.org/10.1007/s00330-017-5065-8
Merkel A, Soeldner D, Wendl C et al (2017) Early postoperative tumor progression predicts clinical outcome in glioblastoma-implication for clinical trails. J Neurooncol 132:249–254
Romano A, Calabria LF, Tavanti F et al (2013) Apparent diffusion coefficient obtained by magnetic resonance imaging as a prognostic marker in glioblastomas: correlation with MGMT promoter methylation status. Eur Radiol 23:513–520
Acknowledgements
We would like to thank Dr. Duo Gao for language editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
This work was carried at the Department of Medical Imaging, The Second Hospital of Hebei Medical University, Shijiazhuang, Hebei Province, China.
Rights and permissions
About this article
Cite this article
Quan, GM., Zheng, YL., Yuan, T. et al. Increasing FLAIR signal intensity in the postoperative cavity predicts progression in gross-total resected high-grade gliomas. J Neurooncol 137, 631–638 (2018). https://doi.org/10.1007/s11060-018-2758-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2758-z