Abstract
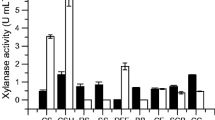
Biotechnology offers innovative alternatives for industrial bioprocesses mainly because it uses enzymes that biodegrade the hemicellulose releasing fermentable sugars. Caulobacter crescentus (C. crescentus) has seven genes responsible for xylanolytic cleavage, 5 to β-xylosidases (EC 3.2.1.37) and 2 for endoxylanases, like xynA2 (CCNA_03137) that encodes Xylanase II (EC 3.2.1.8) of the glycohydrolases-GH10 group. The xynA2 gene was amplified by PCR, cloned into the pTrcHisA vector e efficiently overexpressed in E. coli providing a His-tag fusion protein. Recombinant xylanase (XynA2) was purified by affinity chromatography using a nickel sepharose column and exhibited a single 43 kDa band on SDS-PAGE gel. XynA2 showed an optimum alkaline pH (8) and stability at alkaline pH for 24 h. Although C. crescentus is mesophilic, XynA2 has optimum temperature of 60 °C and is thermo-resistance at 65 °C. XynA maintains 66% of the enzymatic activity at high temperatures (90 °C) without being denatured.The enzyme displayed a xylanolitic activity free of cellulase to xylan from beechwood and it was not inhibited in the presence of 50 μmol mL−1 of xylose. In addition, dithiothreitol (DTT) induced XynA2 activity, as it improved its kinetic parameters by lowering the KM (5.78 μmol mL−1) and increasing the KCat/KM ratio (1.63 U s−1). Finally, C. crescentus XynA2 efficiently hydrolyzed corn straw with high release of reducing sugars that can be applied in different branches of the industry.






Similar content being viewed by others
References
Hoffmam ZB, Zanphorlin LM, Cota J, Diogo JA, Almeida GB, Damásio ARL, Squina F, Murakami MT, Ruller R (2016) Xylan-specific carbohydrate-binding module belonging to family 6 enhances the catalytic performance of a GH11 endo-xylanase. New Biotechnol 33:467–472. https://doi.org/10.1016/j.nbt.2016.02.006
Nunes RM, Guarda EA, Serra JCV, Martins ÁA (2013) Resíduos agroindustriais : potencial de produção do etanol de segunda geração no Brasil. Rev Lib 14:113–123
Annamalai N, Thavasi R, Jayalakshmi S, Balasubramanian T (2009) Thermostable and alkaline tolerant xylanase production by Bacillus subtilis isolated form marine environment. Indian J Biotechnol 8:291–297
Corrêa JM, Mingori MR, Gandra RF, Loth EA, Seixas FAV, Simão RDCG (2014) Depletion of the xynB2 gene upregulates β-xylosidase expression in C. crescentus. Appl Biochem Biotechnol 172:1085–1097. https://doi.org/10.1007/s12010-013-0549-8
Graciano L, Corrêa JM, Vieira FGN, Bosetto A, Loth EA, Kadowaki MK, Gandra RF, Simão RCG (2015) Cloning and Expression of the xynA1 gene encoding a Xylanase of the GH10 group in Caulobacter crescentus. Appl Biochem Biotechnol 175:3915–3929. https://doi.org/10.1007/s12010-015-1560-z
Corrêa JM, Graciano L, Abrahão J, Loth EA, Gandra RF, Kadowaki MK, Henn C, Simão RDCG (2012) Expression and characterization of a GH39 β-xylosidase II from Caulobacter crescentus. Appl Biochem Biotechnol 168:2218–2229. https://doi.org/10.1007/s12010-012-9931-1
Corrêa JM, Christi D, Della Torre CL, Henn C, da Conceição-Silva JL, Kadowaki MK, Simão RCG (2016) High levels of β-xylosidase in Thermomyces lanuginosus: potential use for saccharification. Braz J Microbiol 47:680–690. https://doi.org/10.1016/j.bjm.2016.04.028
Liu G, Qin Y, Li Z, Qu Y (2013) Development of highly efficient, low-cost lignocellulolytic enzyme systems in the post-genomic era. Biotechnol Adv 31:962–975. https://doi.org/10.1016/j.biotechadv.2013.03.001
Bibi Z, Nawaz MA, Waqas M, Aman A, Qader SA (2019) Significance of metal ions, solvents and surfactants to improve the xylan degrading behavior of β-1,4-D-xylanohydrolase from Geobacillus stearothermophilus KIBGE-IB29. Biocatal Agric Biotechnol 17:242–246. https://doi.org/10.1016/j.bcab.2018.11.028
Kumar V, Dangi AK, Shukla P (2018) Engineering thermostable microbial xylanases toward its industrial applications. Mol Biotechnol 60:226–235. https://doi.org/10.1007/s12033-018-0059-6
Poindexter JS (1981) The Caulobacters: ubiquitous unusual bacteria. Microb Rev 45:123–179
Liu X, Liu Y, Jiang Z, Liu H, Yang S, Yan Q (2018) Biochemical characterization of a novel xylanase from Paenibacillus barengoltzii and its application in xylooligosaccharides production from corncobs. Food Chem 264:310–318. https://doi.org/10.1016/j.foodchem.2018.05.023
CAZy, Carbohydrate-Active enzymes Database (2018). https://www.cazy.org/. Accessed 16 Oct 2017
Chang S, Guo Y, Wu B, He B (2017) Extracellular expression of alkali tolerant xylanase from Bacillus subtilis Lucky in E. coli and application for xylooligosaccharides production from agro-industrial waste. Int J Biol Macromol 96:249–256. https://doi.org/10.1016/j.ijbiomac.2016.11.032
Li L, Liang B, Shi J, Li F, Mascini M, Liu A (2012) A selective and sensitive D-xylose electrochemical biosensor based on xylose dehydrogenase displayed on the surface of bacteria and multi-walled carbon nanotubes modified electrode. Biosens Bioelectron 33:100–105. https://doi.org/10.1016/j.bios.2011.12.027
Kumar V, Marín-Navarro J, Shukla P (2016) Thermostable microbial xylanases for pulp and paper industries: trends, applications and further perspectives. World J Microbiol Biotechnol 32(2):25–34. https://doi.org/10.1007/s11274-015-2005-0
Heinen PR, Henn C, Peralta RM, Bracht A, Simão CG, Conceição-Silva JL, Polizeli MLTM, Kadowaki MK (2014) Xylanase from Fusarium heterosporum: properties and influence of thiol compounds on xylanase activity. Afr J Biotechnol 13:1047–1055. https://doi.org/10.5897/AJB2013.13282
Marks ME, Castro-Rojas CM, Teiling C, Du L, Kapatral V, Walunas TL, Crosson S (2010) The genetic basis of laboratory adaptation in Caulobacter crescentus. J Bacteriol 192:3678–3688. https://doi.org/10.1128/JB.00255-10
Justo PI, Corrêa JM, Maller A, Kadowaki MK, da Conceição-Silva JL, Gandra RF, Simão RCG (2015) Antonie van Leeuwenhoek. Int J Gen Mol Microbiol 108:993–1007. https://doi.org/10.1007/s10482-015-0552-x
Graciano L, Corrêa JM, Gandra RF, Seixas FAV, Kadowaki MK, Sampaio SC, da Conceição-Silva JL, Osaku CA (2012) The cloning, expression, purification, characterization and modeled structure of Caulobacter crescentus β-xylosidase I. World J Microbiol Biotechnol 28:2879–2888. https://doi.org/10.1007/s11274-012-1099-x
Nierman WC, Feldblyum TV, Laub MT, Paulsen IT, Nelson KE, Eisen J, Heidelberg JF, Alley MRK, Ohta N, Maddock JR, Potocka I, Nelson WC, Newton A, Stephens C, Phadke ND, Ely B, DeBoy RT, Dodson RJ, Durkin AS, Gwinn ML, Haft DH, Kolonay JF, Smit J, Craven MB, Khouri H, Shetty J, Berry K, Utterback T, Tran K, Wolf A, Vamathevan J, Ermolaeva M, White O, Salzberg SL, Venter JC, Shapiro L, Fraser CM (2001) Complete genome sequence of Caulobacter crescentus. Proc Natl Acad Sci 98:4136–4141. https://doi.org/10.1073/pnas.061029298
Hughes V, Jiang C, Brun YV (2012) Caulobacter crescentus. Curr Biol 22:507–509. https://doi.org/10.1016/j.cub.2012.05.036
Chen W, Kuo T (1993) A simple and rapid method for the preparation of gram-negative bacterial genomic DNA. Nucleic Acids Res 21:2260–2260. https://doi.org/10.1093/nar/21.9.2260
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: A laboratory manual, 2nd edn. Cold Spring Harbor press, New York
NCBI, National Center for Biotechnology Information, (2017). https://www.ncbi.nlm.nih.gov/blast/. Accessed 12 Oct 2017
E. Myers, S. Altschul, W. Gish, W. Miller, D. Lipman, NCBI, BLAST 2.2.29, (2014) https://blast.ncbi.nlm.nih.gov. Accessed 12 Oct 2017
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Miller GL (1959) Use of Dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Davis BJ (1964) Disc electrophoresis - II Method and application to human serum proteins. Ann N Y Acad Sci 122:404–427. https://doi.org/10.1111/j.1749-6632.1964.tb14213.x
McIlvaine TC (1921) A buffer solution for colorimetric comparison. J Biol Chem 49:183–186
Kozlowski LP (2016) IPC - isoelectric point calculator. Biol Direct 11:1–16. https://doi.org/10.1186/s13062-016-0159-9
R Development Core Team (2018) R: a language and environment for statistical computing 3.5.1, Vienna, Austria: R Foundation for Statistical Computing. www.r-project.org. Accessed 28 Feb 2020
Gessesse A, Mamo G (1998) Purification and characterization of an alkaline xylanase from alkaliphilic Micrococcus sp. AR-135. J Ind Microbiol Biotechnol 20:210–214. https://doi.org/10.1038/sj.jim.2900503
Dodd D, Cann IKO (2009) Enzymatic deconstruction of xylan for biofuel production. GCB Bioenergy 1:2–17. https://doi.org/10.1111/j.1757-1707.2009.01004.x
Mander P, Choi YH (2014) PGYS Choi JH Hong, SS Cho, JC Yoo, Biochemical characterization of xylanase produced from Streptomyces sp. CS624 using an agro residue substrate. Process Biochem 49:451–456. https://doi.org/10.1016/j.procbio.2013.12.011
Corradini FAS, Baldez TO, Milessi TSS, Tardioli PW, Ferreira AG, Giordano RC, Giordano RLC (2018) Eucalyptus xylan: an in-house-produced substrate for xylanase evaluation to substitute birchwood xylan. Carbohydr Polym 197:167–173. https://doi.org/10.1016/j.carbpol.2018.05.088
Bosetto A, Justo PI, Zanardi B, Venzon SS, Graciano L, Santos EL, Simão RCG (2016) Research progress concerning fungal and bacterial β-xylosidases. Appl Biochem Biotechnol 178:766–795. https://doi.org/10.1007/s12010-015-1908-4
Collins T, Gerday C, Feller G (2005) Xylanases, xylanase families and extremophilic xylanases. FEMS Microbiol Rev 29:3–23. https://doi.org/10.1016/j.femsre.2004.06.005
Rättö M, Poutanen K, Viikari L (1992) Production of xylanolytic enzymes by an alkalitolerant Bacillus circulans strain. Appl Microbiol Biotechnol 37:470–473. https://doi.org/10.1007/BF00180971
Kumar V, Singh PK, Shukla P (2018) Thermostability and Substrate specificity of GH-11 xylanase from Thermomyces lanuginosus VAPS24. Indian J Microbiol 58(4):515–519. https://doi.org/10.1007/s12088-018-0751-z
Dutta T, Sengupta R, Sahoo R, Ray SS, Bhattacharjee A, Ghosh S (2007) A novel cellulase free alkaliphilic xylanase from alkali tolerant Penicillium citrinum: production, purification and characterization. Lett Appl Microbiol 44:206–211. https://doi.org/10.1111/j.1472-765X.2006.02042.x
Silva LAO, Terrasan CRF, Carmona EC (2015) Purification and characterization of xylanases from Trichoderma inhamatum. Electron J Biotechnol 18:307–313. https://doi.org/10.1016/S0960-8524(97)00052-7
Knob A, Beitel SM, Fortkamp D, Terrasan CRF, Almeida AF (2013) Production, purification, and characterization of a major Penicillium glabrum xylanase using Brewer’s spent grain as substrate. Biomed Res Int 2013:1–8. https://doi.org/10.1155/2013/728735
Vieira WB, Moreira LRDS, Neto AM, Filho EXF (2007) Production and characterization of an enzyme complex from a new strain of Clostridium thermocellum with emphasis on its xylanase activity. Braz J Microbiol 38:237–242. https://doi.org/10.1590/S1517-83822007000200009
Cai ZW, Ge HH, Yi ZW, Zeng RY, Zhang GY (2018) Characterization of a novel psychrophilic and halophilic β-1,3-xylanase from deep-sea bacterium, Flammeovirga pacifica strain WPAGA1. Int J Biol Macromol 118:2176–2184. https://doi.org/10.1016/j.ijbiomac.2018.07.090
Kumar V, Shukla P (2018) Extracellular xylanase production from T lanuginosus VAPS24 at pilot scala and thermostability enhancement by immobilization. Process Biochem 71:53–60. https://doi.org/10.1016/j.procbio.2018.05.019
Shao W, Xue Y, Wu A, Kataeva I, Pei J, Wu H, Wiegel J (2011) Characterization of a novel β-xylosidase, XylC, from Thermoanaerobacterium saccharolyticum JW/SL-YS485. Appl Environ Microbiol 77:719–726. https://doi.org/10.1128/AEM.01511-10
Abd El Aty AA, Saleh SAA, Eid BM, Ibrahim NA, Mostafa FA (2018) Thermodynamics characterization and potential textile applications of Trichoderma longibrachiatum KT693225 xylanase. Biocatal Agric Biotechnol 14:129–137. https://doi.org/10.1016/j.bcab.2018.02.011
Teng C, Yan Q, Jiang Z, Fan G, Shi B (2010) Production of xylooligosaccharides from the steam explosion liquor of corncobs coupled with enzymatic hydrolysis using a thermostable xylanase. Bioresour Technol 101:7679–7682. https://doi.org/10.1016/j.biortech.2010.05.004
Teng C, Jia H, Yan Q, Zhou P, Jiang Z (2011) High-level expression of extracellular secretion of a β-xylosidase gene from Paecilomyces thermophila in Escherichia coli. Bioresour Technol 102:1822–1830. https://doi.org/10.1016/j.biortech.2010.09.055
Kumar V, Kumar A, Chhabra D, Shukla P (2019) Improved biobleaching of mixed hardwood pulp and process optimization using novel GA-ANN and GA-ANFIS hybrid statistical tools. Bioresour Technol 271:274–282. https://doi.org/10.1016/j.biortech.201809.115
Kumar V, Chhabra D, Shukla P (2017) Xylanase production from Thermomyces lanuginosus VAPS-24 using low cost agroindustrial residues via hybrid optimization tools and its potential use for saccarification. Bioresour Technol 243:1009–1019. https://doi.org/10.1016/j.biortech.2017.07.094
Funding
D. Jacomini, L. Bussler and J.M. Corrêa were fellows of CAPES, the Coordination of Improvement of Higher Education Personnel.
Author information
Authors and Affiliations
Contributions
The manuscript corresponds to an original work of the authors and was not previously submitted to Molecular Biology Reports or elsewhere. This work was carried out in collaboration among all authors whose names appears on the submission. Authors DJ and LB performed the experiments and wrote the first draft of the manuscript. Author JMC wrote the protocol. Authors MKK and AM designed the study and performed the statistical analysis. Authors DJ and JLCS managed the literature searches. Author RCGS managed the critical analyses of the study and wrote the final version of the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The present research was performed in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jacomini, D., Bussler, L., Corrêa, J.M. et al. Cloning, expression and characterization of C. crescentus xynA2 gene and application of Xylanase II in the deconstruction of plant biomass. Mol Biol Rep 47, 4427–4438 (2020). https://doi.org/10.1007/s11033-020-05507-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05507-2