Abstract
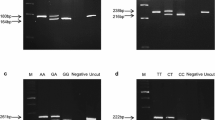
It is necessary that genetic markers or biomarkers can be used to predict resistance towards a wide range of infectious diseases. In the present study, we estimated the potential markers and measured their relationship with heritabilities of a wide range of immune traits. Polymorphisms in exon 13 of Mx1, intron 25 of BAT2 and intron 3 of CXCL12 were identified by sequencing, and the genotypes were analyzed by PCR-RFLP in a resource population composed of 352 pure breed Landrace piglets at days 0, 17 and 32 after birth. Associations of single-nucleotide polymorphisms (SNPs) in these genes with a variety of immunological traits and antibody levels for pig reproduction and porcine respiratory syndrome virus (PRRSV), pseudorabies virus (PRV) and classical swine fever virus (CSFV) were performed. The performance of GG genotype of BAT2 on hemoglobin concentration (HBG) and hematocrit (HCT) of piglets at day 0 was significantly higher than that of the AA and AG individuals. For Mx1, compared with CT genotype, the pigs with TT or CC generated more PRRS antibody at day 0. The piglets with CT genotype had highly significant difference of PRV antibody from those with CC and TT genotypes at day 0. And the piglets with CC genotype had higher level red blood cell count (RBC), hemoglobin concentration (HBG) and hematocrit (HCT) than those with CT and TT genotypes at day 17. For the C7462G SNP in the intron 3 of CXCL12, the PRV antibody level of piglets with the CG genotype were higher than that of piglets with CC and GG genotypes at day 17, and the mean corpuscular volume (MCV) of GG piglets were larger than that of CC and CG individuals at day 0. At the locus 7331 bp in the intron 3 of CXCL12, there were significantly differences of mean corpuscular hemoglobin concentrations (MCHC) at day 0 and white blood cell count (WBC) at day 32, which showed the trend GG or AG>AA, AA>AG or GG, respectively. The pigs with AA or GG genotype had more platelet distribution width (PDW), mean platelet volume (MPV) and platelet-large cell ratio (PLR) at day 17 than those with AG. The results of this study indicated that polymorphisms in Mx1, BAT2 and CXCL12 genes were significantly associated with the immunological traits in Landrace piglets and had potential application value for marker-assisted selection of pig breeding with disease resistance.

Similar content being viewed by others
References
Lunney JK, Ho CS, Wysocki M, Smith DM (2009) Molecular genetics of the swine major histocompatibility complex, the SLA complex. Dev Comp Immunol 33:362–374
Rothbard JB, Gefter ML (1991) Interactions between immunogenic peptides and MHC proteins. Annu Rev Immunol 9:527–565
Sachs DH, Leight J, Cone J, Schwartz S, Stuart L, Rosenburg S (1976) Transplantation in miniature swine. I. Fixation of the major histocompatibility complex. Transplantation 22:559–567
Rothschild MF, Chen HL, Christian LL, Lie WR, Venier L, Cooper M, Briggs C, Warner CM (1984) Breed and swine lymphocyte antigen haplotype differences in agglutination titers following vaccination with B. bronchiseptica. J Anim Sci 59:643–649
Rothschild MF, Hill HT, Christian LL, Warner CM (1984) Genetic differences in serum-neutralization titers of pigs after vaccination with pseudorabies modified live-virus vaccine. Am J Vet Res 45:1216–1218
Lunney JK, Murrel KD (1988) Immunogenetic analysis of Trichiniella spiralis infections in swine. Vet Parasitol 29:179–193
Nunes M, Peelman L, Vaiman M, Bourgeaux N, Chardon P (1994) Characterization of six new loci within the swine major histocompatibility complex class III region. Mamm Genome 5:616–622
Hashimoto M, Nakamura N, Obayashi H, Kimura F, Moriwaki A, Hasegawa G, Shigeta H, Kitagawa Y, Nakano K, Kondo M, Ohta M, Nishimura M (1999) Genetic contribution of the BAT2 gene microsatellite polymorphism to the age-at-onset of insulin-dependent diabetes mellitus. Hum Genet 105:197–199
Lindenmann J, Lane CA, Hobson D (1963) The resistance of A2G mice to myxoviruses. J Immunol 90:942–951
Chieux V, Hober D, Harvey J, Lion G, Lucidarme D, Forzy G, Duhamel M, Cousin J, Ducoulombier H, Wattré P (1998) The MxA protein levels in whole blood lysates of patients with various viral infections. J Virol Methods 70:183–191
Haller O, Staeheli P, Kochs G (2007) Interferon-induced Mx proteins in antiviral host defense. Biochimie 89:812–818
Ko JH, Jin HK, Asano A, Takada A, Ninomiya A, Kida H, Hokiyama H, Ohara M, Tsuzuki M, Nishibori M, Mizutani M, Watanabe T (2002) Polymorphisms and the differential antiviral activity of the chicken Mx gene. Genome Res 12:527–530
Morozumi T, Sumantri C, Nakajima E, Kobayashi E, Asano A, Oishi T, Mitsuhashi T, Watanabe T, Hamasima N (2001) Three types of polymorphisms in exon 14 in porcine Mx1 gene. Biochem Genet 39:251–260
Oh JW, Drabik K, Kutsch O, Choi C, Tousson A, Benveniste EN (2001) CXC chemokine receptor 4 expression and function in human astroglioma cells. J Immunol 166:2695–2704
Singh S, Sadanandam A, Singh RK (2007) Chemokines in tumor angiogenesis and metastasis. Cancer Metastasis Rev 26:453–467
Raman D, Baugher PJ, Thu YM, Richmond A (2007) Role of chemokines in tumor growth. Cancer Lett 256:137–165
Viola A, Luster AD (2008) Chemokines and their receptors: drug targets in immunity and inflammation. Annu Rev Pharmacol Toxicol 48:171–197
Juarez J, Bendall L, Bradstock K (2004) Chemokines and their receptors as therapeutic targets: the role of the SDF-1/CXCR4 axis. Curr Pharm Des 10:1245–1259
Busillo JM, Benovic JL (2007) Regulation of CXCR4 signaling. Biochim Biophys Acta 1768:952–963
Marchese A, Chen C, Kim YM, Benovic JL (2003) The ins and outs of G protein-coupled receptor trafficking. Trends Biochem Sci 28:369–376
Smith MC, Luker KE, Garbow JR, Prior JL, Jackson E, Piwnica-Worms D, Luker GD (2004) CXCR4 regulates growth of both primary and metastatic breast cancer. Cancer Res 64:8604–8612
Payne AS, Cornelius LA (2002) The role of chemokines in melanoma tumor growth and metastasis. J Invest Dermatol 118:915–922
Locati M, Murphy PM (1999) Chemokines and chemokine receptors: biology and clinical relevance in inflammation and AIDS. Annu Rev Med 50:425–440
Ma G, Huang J, Sun N, Liu X, Zhu M, Wu Z, Zhao S (2008) Molecular characterization of the porcine GBP1 and GBP2 genes. Mol Immunol 45:2797–2807
Sambrook J, Fritschi EF, Maniatis T (1989) Molecular cloning: a laboratorymanual. Cold Spring Harbor Laboratory Press, New York
Svejgaard A, Platz P, Ryder LP (1983) HLA and disease 1982–a survey. Immunol Rev 70:193–218
Singal DP, Li J, Zhu Y (2000) HLA class III region and susceptibility to rheumatoid arthritis. Clin Exp Rheumatol 18:485–491
Gong YF, Lu X, Wang ZP, Li S, Qiu XT, Zhang Q (2008) Detection of Quantitative Trait Loci Affecting Haematological Traits on Swine Chromosomes 2, 7 and 8. Progress in Biochemistry and Biophysics 35:1291–1297
Le Bon A, Tough DF (2002) Links between innate and adaptive immunity via type I interferon. Curr Opin Immunol 14:432–436
Samuel CE (2001) Antiviral actions of interferons. Clin Microbiol Rev 14:778–809
Muller-Doblies D, Ackermann M, Metzler A (2002) In vitro and in vivo detection of mx gene products in bovine cells following stimulation with alpha/beta interferon and viruses. Clin Diagn Lab Immunol 9:1192–1199
Arnheiter H, Skuntz S, Noteborn M, Chang S, Meier E (1990) Transgenic mice with intracellular immunity to influenza virus. Cell 62:51–61
Haller O, Stertz S, Kochs G (2007) The Mx GTPase family of interferon- induced antiviral proteins. Microbes Infect 9:1636–1643
Asano A, Ko JH, Morozumi T, Hamashima N, Watanabe T (2002) Polymorphisms and the antiviral property of porcine Mx1 protein. J Vet Med Sci 64:1085–1089
Palm M, Garigliany MM, Cornet F, Desmecht D (2010) Interferon-induced Sus scrofa Mx1 blocks endocytic traffic of incoming influenza A virus particles. Vet Res 41:29
Zhang X, Shin J, Molitor TW, Schook LB, Rutherford MS (1999) Molecular responses of macrophages to porcine reproductive and respiratory syndrome virus infection. Virology 262:152–162
Klein RS, Rubin JB (2004) Immune and nervous system CXCL12 and CXCR4: parallel roles in patterning and plasticity. Trends Immunol 25:306–314
D’Apuzzo M, Rolink A, Loetscher M, Hoxie JA, Clark-Lewis I, Melchers F, Baggiolini M, Moser B (1997) The chemokine SDF-1, stromal cell-derived factor 1, attracts early stage B cell precursors via the chemokine receptor CXCR4. Eur J Immunol 27:1788–1793
Nagasawa T, Kikutani H, Kishimoto T (1994) Molecular cloning and structure of a pre-B-cell growth-stimulating factor. Proc Natl Acad Sci USA 91:2305–2309
Nanki T, Lipsky PE (2000) Cutting edge: stromal cell-derived factor-1 is a costimulator for CD4_ T cell activation. J Immunol 164:5010–5014
Ara T, Itoi M, Kawabata K, Egawa T, Tokoyoda K, Sugiyama T, Fujii N, Amagai T, Nagasawa T (2003) A role of CXC chemokine ligand 12/stromal cell-derived factor-1/pre-B cell growth stimulating factor and its receptor CXCR4 in fetal and adult T cell development in vivo. J Immunol 170:4649–4655
Onai N, Zhang Y, Yoneyama H, Kitamura T, Ishikawa S, Matsushima K (2000) Impairment of lymphopoiesis and myelopoiesis in mice reconstituted with bone marrow-hematopoietic progenitor cells expressing SDF-1-intrakine. Blood 96:2074–2080
Rossi D, Zlotnik A (2000) The biology of chemokines and their receptors. Ann Rev Immunol 18:217–242
Guo Y, Hangoc G, Bian H, Pelus LM, Broxmeyer HE (2005) SDF-1/CXCL12 enhances survival and chemotaxis of murine embryonic stem cells and production of primitive and definitive hematopoietic progenitor cells. Stem Cells 23:1324–1332
Acknowledgments
Financial assistance from 863 High Technology and Development Project of China (2006AA10Z195, 2007AA10Z152), Ministry of Science and Technology (2009ZX08009-142B, S2010GR0947) and Ministry of Agriculture in China (2010-Z16) are greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
S.J. Wang and W.J. Liu contributed equally.
Rights and permissions
About this article
Cite this article
Wang, S.J., Liu, W.J., Sargent, C.A. et al. Effects of the polymorphisms of Mx1, BAT2 and CXCL12 genes on immunological traits in pigs. Mol Biol Rep 39, 2417–2427 (2012). https://doi.org/10.1007/s11033-011-0992-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-0992-y