Abstract
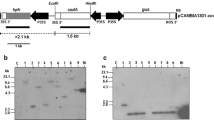
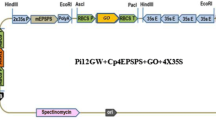
Since the maize transposon Ac can move to a new location within the genome, it has been used in removing selectable markers in transgenic plants. In this paper, we developed an inducible transposon system to truncate a selectable marker in transgenic plants. In this system, the marker gene was accompanied by the inducible transposon, but one end of the transposon was located in the intron of the marker gene. As an example of a marker gene, we isolated the rice 5-enolpyruvylshikimate-3-phosphate synthase (epsps) and modified it for glyphosate tolerance. The transposon contained Ac transposase, which fused with the promoter of the inducible gene for pathogenesis-related protein 1a (PR-1a). This construct was engineered into an expression vector pCAMBIA1300, harboring a hygromycin-resistant gene. The construct was first transformed into rice calli, and transformed plants were selected on hygromycin-containing medium. The stably transformed calli underwent determination for normal transcripts and tolerance to glyphosate. The results were applied to a rice transformation with the same construct, but using glyphosate as the selective agent. By determining the transformation efficiency, T-DNA copy patterns, we demonstrate that the modified epsps could be a suitable selectable marker to create transgenic rice. Furthermore, after obtaining stable transgenic plants and inducing transposition by salicylic acid, the transposon was excised, the marker gene became truncated, and its expression was terminated. This strategy could be applicable to yield self-stabilizing transposon by locating the transposon’s end in the transposase gene’s intron.






Similar content being viewed by others
Abbreviations
- epsps :
-
5-enolpyruvylshikimate-3-phosphate synthase gene
- GOI:
-
Gene of interest
- SA:
-
Salicylic acid
- TPase :
-
Transposase gene
References
Bradshaw LD, Padgette SR, Kimball SL, Wells BH (1997) Perspectives on glyphosate resistance. Weed Technol 11:189–198
Charng Y-C, Pfitzner AJP, Pfitzner UM, Charng-Chang K-F, Chen C-M, Tu J, Kuo T-T (2000) Construction of an inducible transposon, INAc, to develop a gene tagging system in higher plants. Mol Breed 6:353–367
Charng Y-C, Wu G, Hsieh C-S, Chuan H-N, Huang J-Y, Yeh L-C, Shieh Y-H, Tu J (2007) The inducible transposon system for rice functional genomics. Bot Stud 48:1–11
Clemente TE, LaVallee BJ, Howe AR, Conner-Ward D, Rozman RJ, Hunter PE, Broyles DL, Kasten DS, Hinchee MA (2000) Progeny analysis of glyphosate selected transgenic soybeans derived from agrobacterium-mediated transformation. Crop Sci 40:797–803
Ebinuma H, Sugita K, Matsunaga E, Yamakado M (1997) Selection of marker-free transgenic plants using the isopentenyl transferase gene. Proc Natl Acad Sci USA 94:2117–2121
Ebinuma H, Sugita K, Matsunaga E, Endo S, Yamada K, Komamine A (2001) Systems for the removal of a selection marker and their combination with a positive marker. Plant Cell Rep 20:383–392
Endo S, Sugita K, Sakai M, Tanaka H, Ebinuma H (2002) Single-step transformation for generating marker-free transgenic rice using the ipt-type MAT vector system. Plant J 30:115–122
Gruener R, Pfitzner UM (1994) The upstream region of the gene for the pathogenesis-related protein 1a from tobacco responds to environmental as well as to developmental signals in transgenic plants. Eur J Biochem 220:247–255
Hajdukiewicz PTJ, Gilbertson L, Staub JM (2001) Multiple pathways for Cre/lox-mediated recombination in plastids. Plant J 27:161–170
Hare PD, Chua N-H (2002) Excision of selectable marker genes from transgenic plants. Nat Biotechnol 20(6):575–580
Haring MA, Rommens CMT, Nijkamp HJJ, Hille J (1991) The use of transgenic plants to understand transposition mechanisms and to develop transposon tagging strategies. Plant Mol Biol 16:449–461
Hohn B, Levy AA, Puchta H (2001) Elimination of selection markers from transgenic plants. Curr Opin Biotechnol 12:139–143
Howe AR, Gasser CS, Brown SM, Padgette SR, Hart J, Parker GB, Fromm ME, Armstrong CL (2002) Glyphosate as a selective agent for the production of fertile transgenic maize (Zea mays L.) plants. Mol Breed 10:153–164
Hu T, Metz S, Chay C, Zhou HP, Biest N, Chen G, Cheng M, Feng X, Radionenko M, Lu F, Fry J (2003) Agrobacterium-mediated large-scale transformation of wheat (Triticum aestivum L.) using glyphosate selection. Plant Cell Rep 21:1010–1019
Li F, Dey M, He C, Sangwan V, Wu X, Wu R (2003) Rapid PCR-based determination of transgene copy number in rice. Plant Mol Biol Rep 21:73–80
Liu YG, Mitsukawa N, Oosumi T, Whittier RF (1995) Efficient isolation and mapping of Arabidopsis thaliana T-DNA insert junctions by thermal asymmetric interlaced PCR. Plant J 8:457–463
Magnani E, Bartling L, Hake S (2006) From Gateway to MultiSite Gateway in one recombination event. BMC Mol Biol 7:46. doi:10.1186/1471-2199-7-46
Miki B, McHugh S (2004) Selectable marker genes in transgenic plants: applications, alternatives and biosafety. J Biotechnol 107:193–232
Ow DW (2007) GM maize from site-specific recombination technology, what next? Curr Opin Biotechnol 18:115–120
Sambrook J, Russel DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor, New York
Schönbrunn E, Eschenburg S, Shuttleworth WA, Schloss JV, Amrhein N, Evans JNS, Kabsch W (2001) Interaction of the herbicide glyphosate with its target enzyme 5-enolpyruvylshikimate 3-phosphate synthase in atomic detail. Proc Natl Acad Sci USA 98:1376–1380
Sha Y, Li S, Pei Z, Luo L, Tian Y, He C (2004) Generation and flanking sequence analysis of a rice T-DNA tagged population. Theor Appl Genet 108:306–314
Toki S (1997) Rapid and efficient Agrobacterium-mediated transformation in rice. Plant Mol Biol Rep 15:16–21
van Loon LC, van Kammen A (1970) Polyacrylamide disc electrophoresis of the soluble leaf proteins from Nicotiana tabacum var. “Samsun” and “Samsun NN”. II. Changes in protein constitution after infection with tobacco mosaic virus. Virology 40:199–211
White RF (1979) Acetylsalicylic acid (aspirin) induces resistance to tobacco mosaic virus in tobacco. Virology 99:410–412
Zhou H, Arrowsmith JW, Fromm ME, Hironaka CM, Taylor ML, Rodriguez D, Pajeau ME, Brown SM, Santino CG, Fry JE (1995) Glyphosate-tolerant CP4 and GOX genes as a selectable marker in wheat transformation. Plant Cell Rep 15:159–163
Zuo JR, Niu Q-W, Møller SG, Chua N-H (2001) Chemical-regulated, site-specific DNA excision in transgenic plants. Nat Biotechnol 19:157–161
Acknowledgments
We thank Dr. Y. Kishima (Hokkaido University, Japan) for providing the plasmid. This project was supported by the National Science Council (grant no. NSC93-2317-B-002-007) of Taiwan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below are the links to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Charng, YC., Li, KT., Tai, HK. et al. An inducible transposon system to terminate the function of a selectable marker in transgenic plants. Mol Breeding 21, 359–368 (2008). https://doi.org/10.1007/s11032-007-9137-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-007-9137-3