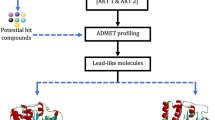
Abstract
Breast and stomach cancer is reported as a leading cause for human mortality across the world. The overexpression of receptor tyrosine kinase (RTK) proteins, namely the human epidermal growth factor receptor2 (HER2) and the vascular endothelial growth factor receptor2 (VEGFR2), is reported to be responsible for development and metastasis of breast and stomach cancer. Although several synthetic tyrosine kinase inhibitors (TKIs) as drug candidates targeting RTK—HER2 and VEGFR2 are currently available in the market, these are expensive with the reported side effects. This confers an opportunity for development of alternative novel tyrosine kinase inhibitors (TKIs) for RTK—HER2 and VEGFR2 receptors from the botanical sources. In the present study, we characterized 47 bioactive phytocompounds from the methanol extracts of the rhizomes of Asiatic traditional medicinal herbs—Panax bipinnatifidus and Panax pseudoginseng, of Indian Himalayan landraces using HPLC, GC–MS and high-sensitivity LC–MS tools. We performed molecular docking and molecular dynamics simulation analysis using Schrödinger suite 2020-3 to confirm the TKI phytocompounds showing the best binding affinity towards RTK—HER2 and VEGFR2 receptors. The results of molecular docking studies confirmed that the phytocompound (ligand) luteolin 7-O-glucoside (IHP15) showed the highest binding affinity towards receptor HER2 (PDB ID: 3PP0) with docking score and Glide g score (G-Score) of – 13.272, while chlorogenic acid (IHP12) showed the highest binding affinity towards receptor VEGFR2 (PDB ID: 4AGC) with docking score and Glide g score (G-Score) of – 10.673. Molecular dynamics (MD) simulation analysis carried out for 100 ns has confirmed strong binding interaction between the ligand and receptor complex [luteolin 7-O-glucoside (IHP15) and HER2 (PDB ID: 3PP0)] and is found to be stabilized within 40 to 100 ns of MD simulation, whereas ligand–receptor complex [chlorogenic acid (IPH12) and VEGFR2 (PDB ID: 4AGC)] also showed strong binding interaction and is found to be stabilized within 18–30 ns but slightly deviated during 100 ns of MD simulation. In silico ADME-Tox study using SwissADME revealed that the ligands luteolin 7-O-glucoside (IHP15) and chlorogenic acid (IHP12) have passed majority parameters of the common drug discovery rules. The present study has confirmed luteolin 7-O-glucoside (IHP15) and chlorogenic acid (IHP12) as potential tyrosine kinase inhibitors (TKIs) which were found to inhibit RTKs—HER2 and VEGFR2 receptor proteins, and thus paving the way for development of alternative potential TKIs (drug molecules) for treatment of HER2- and VEGFR2-positive breast and stomach cancer.
Graphic abstract











Similar content being viewed by others
Abbreviations
- ADME-Tox :
-
Adsorption, digestion, metabolism, excretion and toxicity
- GC-MS:
-
Gas chromatography mass spectrometry
- HER2:
-
Human epidermal growth factor receptor2
- HPLC:
-
High-performance liquid chromatography
- IHP:
-
Indian Himalayan Panax
- LC-MS:
-
Liquid chromatography mass spectrometry
- MDS:
-
Molecular dynamics simulation
- MERPB:
-
Methanol extract of rhizome of Panax bipinnatifidus
- MERPB:
-
Methanol extract of rhizome of Panax pseudoginseng
- NADPH oxidase:
-
Nicotinamide adenine dinucleotide phosphate oxidase
- NRTK:
-
Non-receptor tyrosine kinase
- PDB ID:
-
Protein Data Bank identity
- RMSD:
-
Root mean square deviation
- RMSF:
-
Root mean square fluctuation
- RTK:
-
Receptor tyrosine kinase
- TK:
-
Tyrosine kinase
- TKI:
-
Tyrosine kinase inhibitor
- VEGFR2:
-
Vascular endothelial growth factor receptor2
References
Mallath MK, Taylor DG, Badwe RA et al (2014) The growing burden of cancer in India: epidemiology and social context. Lancet Oncol 15:e205–e212. https://doi.org/10.1016/S1470-2045(14)70115-9
Hyuna S, Jacques F, Rebecca L. S, Mathieu L, Isabelle S, Ahmedin J, Freddie B (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3): 209–249. doi: https://doi.org/10.3322/caac.21660
Liling H, Shiyu J (2020) Yuankai S (2020) Tyrosine kinase inhibitors for solid tumors in the past 20 years (2001–2020). J Hemato Oncol 13:143. https://doi.org/10.1186/s13045-020-00977-0
Bhattacharyya GS, Doval DC, Desai CJ et al (2020) Overview of breast cancer and implications of overtreatment of early-stage breast cancer: an Indian perspective. JCO Glob Oncol 6:789–798. https://doi.org/10.1200/GO.20.00033
Dean-Colomb W, Esteva FJ (2008) HER2-positive breast canceignallintin and beyond. Eur J Cancer 44:2806–2812. https://doi.org/10.1016/j.ejca.2008.09.013
Das SK, Deka SJ, Paul D et al (2021) In-silico based identification of phytochemicals from Houttuynia cordata Thunb. as potential inhibitors for overexpressed HER2 and VEGFR2 cancer genes. J Biomol Struct Dynamics 1–14. https://doi.org/10.1080/07391102.2021.1891136
Iqbal N, Iqbal N (2014) Human epidermal growth factor receptor 2 (HER2) in cancers: overexpression and therapeutic implications. Mol Biol Int 2014:852748. https://doi.org/10.1155/2014/852748
Yarden Y (2001) Biology of HER2 and its importance in breast cancer. Oncology 61:1–13. https://doi.org/10.1159/000055396
CDER-FDA: Center for Drug Evaluation and Research—Food & Drug Administration. Silver Spring, MD 20993 (USA). http://www.fda.gov
Guo S, Colbert LS, Fuller M et al (2010) Vascular endothelial growth factor receptor-2 in breast cancer. Biochim Biophys Acta (BBA) Rev Cancer 1806:108–121. https://doi.org/10.1016/j.bbcan.2010.04.004
Ernst P (1999) The role of inflammation in the pathogenesis of gastric cancer. Aliment Pharmacol Ther 13:13–18. https://doi.org/10.1046/j.1365-2036.1999.00003.x
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420:860–867. https://doi.org/10.1038/nature01322
Maeda S, Omata M (2008) Inflammation and cancer: role of nuclear factor-kappaB activation. Cancer Sci 99:836–842. https://doi.org/10.1111/j.1349-7006.2008.00763.x
Ferrara N, Adamis AP (2016) Ten years of anti-vascular endothelial growth factor therapy. Nat Rev Drug Discov 15(6):385–403
Qin S, Li A, Yi M et al (2019) Recent advances on anti-angiogenesis receptor tyrosine kinase inhibitors in cancer therapy. J Hematol Oncol 12(1):27
Choudhari AS, Mandave PC, Deshpande M et al (2020) Phytochemicals in cancer treatment: from preclinical studies to clinical practice. Front Pharmacol 10:1614. https://doi.org/10.3389/fphar.2019.01614
Frodin DG, Govaerts R (2003) World checklist and bibliography of Araliaceae: 1–444. The Board of Trustees of the Royal Botanic Gardens, Kew (publ. 2004). http://wcsp.science.kew.org
Ratan ZA, Haidere MF, Hong YH et al (2020) Pharmacological potential of ginseng and its major component ginsenosides. J Ginseng Res 45(2):199–210. https://doi.org/10.1016/j.jgr.2020.02.004
Bahukhandi A, Upadhyay S, Bisht K (2021) Panax ginseng CA Meyer. In: Naturally occurring chemicals against Alzheimer’s disease. Elsevier, pp 217–223
Das SK, Das TJ, Tshering D et al (2019) Ethnobotanical notes on significant food and medicinal flora used by the indigenous Monpa and Nyishi communities of Arunachal Pradesh, India. Pleione 13:291–304. https://doi.org/10.26679/Pleione.13.2.2019.291-304
Yanka H, Rinyo R, Das SK et al (2019) A brief cross-cultural ethnobotanical note on the Abotani tribes of Arunachal Pradesh, India. Pleione 13:269–283. https://doi.org/10.26679/Pleione.13.2.2019.269-283
POWO: plants of the world online hosted by Royal Botanic Garden, Kew, UK. http://www.plantsoftheworldonline.org
Li W, Fitzloff JF (2002) HPLC determination of ginsenosides content in ginseng dietary supplements using ultraviolet detection. J Liq Chromatogr Relat Technol 25:2485–2500. https://doi.org/10.1081/JLC-120014269
Sakakibara H, Honda Y, Nakagawa S et al (2003) Simultaneous determination of all polyphenols in vegetables, fruits, and teas. J Agric Food Chem 51:571–581. https://doi.org/10.1021/jf020926l
Roumiguières A, Kinani A, Bouchonnet S, Kinani S (2018) Development and validation of a multiclass method for the determination of organohalogen disinfectant by-products in water samples using solid phase extraction and gas chromatography-tandem mass spectrometry. J Chromatogr A 1579:89–98. https://doi.org/10.1016/j.chroma.2018.10.016
Navarro-Reig M, Jaumot J, García-Reiriz A, Tauler R (2015) Evaluation of changes induced in rice metabolome by Cd and Cu exposure using LC-MS with XCMS and MCR-ALS data analysis strategies. Anal Bioanal Chem 407:8835–8847. https://doi.org/10.1007/s00216-015-9042-2
RCSB-PDB (RCSB-Protein Data Bank). https://www.rcsb.org
Walters WP (2012) Going further than Lipinski’s rule in drug design. Expert Opin Drug Discov 7:99–107. https://doi.org/10.1517/17460441.2012.648612
Parida PK, Paul D, Chakravorty D (2020) The natural way forward: molecular dynamics simulation analysis of phytochemicals from Indian medicinal plants as potential inhibitors of SARS-CoV-2 targets. Phyther Res 34:3420–3433. https://doi.org/10.1002/ptr.6868
Tuckerman M, Berne BJ, Martyna GJ (1992) Reversible multiple time scale molecular dynamics. J Chem Phys 97:1990–2001. https://doi.org/10.1063/1.463137
Daina A, Michielin O, Zoete V (2017) SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep 7:42717. https://doi.org/10.1038/srep42717
Robinson DR, Wu YM, Lin SF (2000) The protein tyrosine kinase family of the human genome. Oncogene 19(49):5548–5557
Zamečnikova A (2014) Novel approaches to the development of tyrosine kinase inhibitors and their role in the fight against cancer. Expert Opin Drug Discov 9(1):77–92
Engel RH, Kaklamani VG (2007) HER2-positive breast cancer. Drugs 67:1329–1341. https://doi.org/10.2165/00003495-200767090-00006
Gravalos C, Jimeno A (2008) HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol 19:1523–1529. https://doi.org/10.1093/annonc/mdn169
Smith NR, Baker D, James NH et al (2010) Vascular endothelial growth factor receptors VEGFR-2 and VEGFR-3 are localized primarily to the vasculature in human primary solid cancers. Clin Cancer Res 16:3548–3561. https://doi.org/10.1158/1078-0432.CCR-09-2797
Wang N, Wang Z-Y, Mo S-L et al (2012) Ellagic acid, a phenolic compound, exerts anti-angiogenesis effects via VEGFR-2 signaling pathway in breast cancer. Breast Cancer Res Treat 134:943–955. https://doi.org/10.1007/s10549-012-1977-9
Rydén L, Linderholm B, Nielsen NH et al (2003) Tumor specific VEGF-A and VEGFR2/KDR protein are co-expressed in breast cancer. Breast Cancer Res Treat 82:147–154. https://doi.org/10.1023/B:BREA.0000004357.92232.cb
Li X, Zhu X, Wang Y et al (2019) Prognostic value and association of Lauren classification with VEGF and VEGFR-2 expression in gastric cancer. Oncol Lett 18:4891–4899. https://doi.org/10.3892/ol.2019.10820
Kopparapu PK, Boorjian SA, Robinson BD et al (2013) Expression of VEGF and its receptors VEGFR1/VEGFR2 is associated with invasiveness of bladder cancer. Anticancer Res 33:2381–2390
Chatterjee S, Heukamp LC, Siobal M et al (2013) Tumor VEGF: VEGFR2 autocrine feed-forward loop triggers angiogenesis in lung cancer. J Clin Invest 123:1732–1740. https://doi.org/10.1172/JCI65385
Al-Batran S-E, Hofheinz RD, Pauligk C et al (2016) Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO. Lancet Oncol 17:1697–1708. https://doi.org/10.1016/S1470-2045(16)30531-9
Dai S-X, Li W-X, Han F-F et al (2016) In silico identification of anti-cancer compounds and plants from traditional Chinese medicine database. Sci Rep 6:1–11. https://doi.org/10.1038/srep25462
Liu L, Xu F-R, Wang Y-Z (2020) Traditional uses, chemical diversity and biological activities of Panax L.(Araliaceae): a review. J Ethnopharmacol 112792. https://doi.org/10.1016/j.jep.2020.112792
Kim D-H (2012) Chemical diversity of Panax ginseng, Panax quinquifolium, and Panax notoginseng. J Ginseng Res 36:1. https://doi.org/10.5142/jgr.2012.36.1.1
Lin S, Hu J, Zhou X, Cheung PCK (2017) Inhibition of vascular endothelial growth factor-induced angiogenesis by chlorogenic acid via targeting the vascular endothelial growth factor receptor 2-mediatignallinging pathway. J Funct Foods 32:285–295. https://doi.org/10.1016/j.jff.2017.03.009
Luthria DL, Mukhopadhyay S (2006) Influence of sample preparation on assay of phenolic acids from eggplant. J Agric Food Chem 54:41–47. https://doi.org/10.1021/jf0522457
Ushio-Fukai M, Nakamura Y (2008) Reactive oxygen species and angiogenesis: NADPH oxidase as target for cancer therapy. Cancer Lett 266:37–52. https://doi.org/10.1016/j.canlet.2008.02.044
Lee J-S, Park BC, Ko YJ et al (2008) Grifola frondosa (maitake mushroom) water extract inhibits vascular endothelial growth factor-induced angiogenesis through inhibition of reactive oxygen species and extracellular signal-regulated kinase phosphorylation. J Med Food 11:643–651. https://doi.org/10.1089/jmf.2007.0629
Baskar AA, Ignacimuthu S, Michael GP, Al Numair KS (2011) Cancer chemopreventive potential of luteolin-7-O-glucoside isolated from Ophiorrhiza mungos Linn. Nutr Cancer 63:130–138. https://doi.org/10.1080/01635581.2010.516869
Goodarzi S, Tabatabaei MJ, Mohammad Jafari R et al (2020) Cuminum cyminum fruits as source of luteolin-7-O-glucoside, potent cytotoxic flavonoid against breast cancer cell lines. Nat Prod Res 34:1602–1606. https://doi.org/10.1080/14786419.2018.1519824
Acknowledgements
The corresponding author (PKH) is deeply thankful to Department of Biotechnology (DBT), Government of India, for the financial support through DBT Twinning project (Grant No. BT/PR16813/NER/95/300/2015, dated 27 January 2017). The co-corresponding author (HT) expresses thankfulness to Board of Research in Nuclear Science (BRNS), Bhabha Atomic Research Centre (BARC), Trombay, India, under Department of Atomic Energy, Government of India, for funding Support (Grant No. 35/14/16/2016-BRNS/35055, dated 4 June 2016). All the authors are also thankful to Rajiv Gandhi University, Rono Hills, Doimukh, Arunachal Pradesh, India, and National Institute of Technology (NIT), Arunachal Pradesh, India, for laboratory and logistic support. The authors (SM and BT) express thankfulness to National Institute of Electronics and Information Technology, Guwahati, Assam, India, and Guwahati University, Jalukbari, Guwahati, Assam, India, for aiding software-based and computational support to successfully complete these experiments.
Funding
This study was funded by Department of Biotechnology (DBT), Government of India, through DBT Twinning project (Grant No. BT/PR16813/NER/95/300/2015, dated 27 January 2017) and Board of Research in Nuclear Science (BRNS), Bhabha Atomic Research Centre (BARC), Trombay, and Department of Atomic Energy, Government of India (Grant No. 35/14/16/2016-BRNS/35055, dated 4 June 2016).
Author information
Authors and Affiliations
Contributions
HT, PKH, RT, RD, SJ, SM and BT are PIs and research mentors who played a key role in experimental designing, critically verified the data and written this manuscript after several rounds of consultation with all the co-authors involved in the paper. SM, DP, SKD, PKH and BT have significantly contributed in in silico identification of the anti-cancer phytocompounds using molecular docking and molecular dynamics simulation analysis, while HT, PKH, DP, DDG and SKD significantly contributed in phytochemistry data generation and analysis.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Paul, D., Mahanta, S., Tag, H. et al. Identification of tyrosine kinase inhibitors from Panax bipinnatifidus and Panax pseudoginseng for RTK—HER2 and VEGFR2 receptors, by in silico approach. Mol Divers 26, 1933–1955 (2022). https://doi.org/10.1007/s11030-021-10304-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-021-10304-5