Abstract
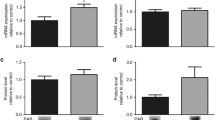
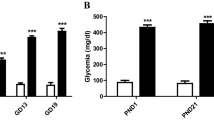
Several studies have examined neonatal diabetes, a rare disease characterized by hyperglycemia and low insulin levels that is usually diagnosed in the first 6 month of life. Recently, the effects of diabetes on the brain have received considerable attention. In addition, hyperglycemia may perturb brain function and might be associated with neuronal death in adult rats. However, few studies have investigated the damaging effects of neonatal hyperglycemia on the rat brain during central nervous system (CNS) development, particularly the mechanisms involved in the disease. Thus, in the present work, we investigated whether neonatal hyperglycemia induced by streptozotocin (STZ) promoted cell death and altered the levels of proteins involved in survival/death pathways in the rat brain. Cell death was assessed using FluoroJade C (FJC) staining and the expression of the p38 mitogen-activated protein kinase (p38), phosphorylated-c-Jun amino-terminal kinase (p-JNK), c-Jun amino-terminal kinase (JNK), protein kinase B (Akt), phosphorylated-protein kinase B (p-Akt), glycogen synthase kinase-3β (Gsk3β), B-cell lymphoma 2 (Bcl2) and Bcl2-associated X protein (Bax) protein were measured by Western blotting. The main results of this study showed that the metabolic alterations observed in diabetic rats (hyperglycemia and hypoinsulinemia) increased p38 expression and decreased p-Akt expression, suggesting that cell survival was altered and cell death was induced, which was confirmed by FJC staining. Therefore, the metabolic conditions observed during neonatal hyperglycemia may contribute to the harmful effect of diabetes on the CNS in a crucial phase of postnatal neuronal development.





Similar content being viewed by others
References
Global report on diabetes
Afrazi S, Esmaeili-Mahani S, Sheibani V, Abbasnejad M (2014) Neurosteroid allopregnanolone attenuates high glucose-induced apoptosis and prevents experimental diabetic neuropathic pain: in vitro and in vivo studies. J Steroid Biochem Mol Biol 139:98–103. https://doi.org/10.1016/j.jsbmb.2013.10.010
Aguilar-Bryan L, Bryan J (2008) Neonatal diabetes mellitus. Endocr Rev 29(3):265–291. https://doi.org/10.1210/er.2007-0029
Alexander MC, Lomanto M, Nasrin N, Ramaika C (1988) Insulin stimulates glyceraldehyde-3-phosphate dehydrogenase gene expression through cis-acting DNA sequences. Proc Natl Acad Sci U S A 85(14):5092–5096. https://doi.org/10.1073/pnas.85.14.5092
Aminzadeh A, Dehpour AR, Safa M, Mirzamohammadi S, Sharifi AM (2014) Investigating the protective effect of lithium against high glucose-induced neurotoxicity in PC12 cells: involvements of ROS, JNK and P38 MAPKs, and apoptotic mitochondria pathway. Cell Mol Neurobiol 34(8):1143–1150. https://doi.org/10.1007/s10571-014-0089-y
Anilkumar U, Prehn JH (2014) Anti-apoptotic BCL-2 family proteins in acute neural injury. Front Cell Neurosci 8:281. https://doi.org/10.3389/fncel.2014.00281
Barber AJ, Nakamura M, Wolpert EB, Reiter CE, Seigel GM, Antonetti DA, Gardner TW (2001) Insulin rescues retinal neurons from apoptosis by a phosphatidylinositol 3-kinase/Akt-mediated mechanism that reduces the activation of caspase-3. J Biol Chem 276(35):32814–32821. https://doi.org/10.1074/jbc.M104738200
Cargnello M, Roux PP (2011) Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev 75(1):50–83. https://doi.org/10.1128/MMBR.00031-10
Cavaletti G, Miloso M, Nicolini G, Scuteri A, Tredici G (2007) Emerging role of mitogen-activated protein kinases in peripheral neuropathies. J Peripher Nerv Syst 12(3):175–194. https://doi.org/10.1111/j.1529-8027.2007.00138.x
Chen J, Guo R, Yan H, Tian L, You Q, Li S, Huang R, Wu K (2014) Naringin inhibits ROS-activated MAPK pathway in high glucose-induced injuries in H9c2 cardiac cells. Basic Clin Pharmacol Toxicol 114(4):293–304. https://doi.org/10.1111/bcpt.12153
Dhanasekaran DN, Reddy EP (2008) JNK signaling in apoptosis. Oncogene 27(48):6245–6251. https://doi.org/10.1038/onc.2008.301
Duarte AI, Santos P, Oliveira CR, Santos MS, Rego AC (2008) Insulin neuroprotection against oxidative stress is mediated by Akt and GSK-3beta signaling pathways and changes in protein expression. Biochim Biophys Acta 1783(6):994–1002. https://doi.org/10.1016/j.bbamcr.2008.02.016
Eldar-Finkelman H, Schreyer SA, Shinohara MM, LeBoeuf RC, Krebs EG (1999) Increased glycogen synthase kinase-3 activity in diabetes- and obesity-prone C57BL/6J mice. Diabetes 48(8):1662–1666. https://doi.org/10.2337/diabetes.48.8.1662
Ellerby LM, Ellerby HM, Park SM, Holleran AL, Murphy AN, Fiskum G, Kane DJ, Testa MP, Kayalar C, Bredesen DE (1996) Shift of the cellular oxidation-reduction potential in neural cells expressing Bcl-2. J Neurochem 67(3):1259–1267
Ertl T, Gyarmati J, Gaál V, Szabó I (2006) Relationship between hyperglycemia and retinopathy of prematurity in very low birth weight infants. Biol Neonate 89(1):56–59. https://doi.org/10.1159/000088199
Esposti MD, Hatzinisiriou I, McLennan H, Ralph S (1999) Bcl-2 and mitochondrial oxygen radicals. New approaches with reactive oxygen species-sensitive probes. J Biol Chem 274(42):29831–29837. https://doi.org/10.1074/jbc.274.42.29831
Evans JL, Goldfine ID, Maddux BA, Grodsky GM (2002) Oxidative stress and stress-activated signaling pathways: a unifying hypothesis of type 2 diabetes. Endocr Rev 23(5):599–622. https://doi.org/10.1210/er.2001-0039
Flanagan SE, Patch AM, Mackay DJ, Edghill EL, Gloyn AL, Robinson D, Shield JP, Temple K, Ellard S, Hattersley AT (2007) Mutations in ATP-sensitive K+ channel genes cause transient neonatal diabetes and permanent diabetes in childhood or adulthood. Diabetes 56(7):1930–1937. https://doi.org/10.2337/db07-0043
Flechtner I, Vaxillaire M, Cavé H, Scharfmann R, Froguel P, Polak M (2008) Neonatal hyperglycaemia and abnormal development of the pancreas. Best Pract Res Clin Endocrinol Metab 22(1):17–40. https://doi.org/10.1016/j.beem.2007.08.003
Garg R, Agthe AG, Donohue PK, Lehmann CU (2003) Hyperglycemia and retinopathy of prematurity in very low birth weight infants. J Perinatol 23(3):186–194. https://doi.org/10.1038/sj.jp.7210879
Greeley SA, Tucker SE, Worrell HI, Skowron KB, Bell GI, Philipson LH (2010) Update in neonatal diabetes. Curr Opin Endocrinol Diabetes Obes 17(1):13–19. https://doi.org/10.1097/MED.0b013e328334f158
Green DR, Reed JC (1998) Mitochondria and apoptosis. Science 281(5381):1309–1312. https://doi.org/10.1126/science.281.5381.1309
Guo YJ, Wang SH, Yuan Y, Li FF, Ye KP, Huang Y, Xia WQ, Zhou Y (2014) Vulnerability for apoptosis in the hippocampal dentate gyrus of STZ-induced diabetic rats with cognitive impairment. J Endocrinol Invest 37(1):87–96. https://doi.org/10.1007/s40618-013-0030-0
Hanada M, Feng J, Hemmings BA (2004) Structure, regulation and function of PKB/AKT--a major therapeutic target. Biochim Biophys Acta 1697(1–2):3–16. https://doi.org/10.1016/j.bbapap.2003.11.009
Hays SP, Smith EO, Sunehag AL (2006) Hyperglycemia is a risk factor for early death and morbidity in extremely low birth-weight infants. Pediatrics 118(5):1811–1818. https://doi.org/10.1542/peds.2006-0628
Heidenreich KA, Kummer JL (1996) Inhibition of p38 mitogen-activated protein kinase by insulin in cultured fetal neurons. J Biol Chem 271(17):9891–9894. https://doi.org/10.1074/jbc.271.17.9891
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407(6805):770–776. https://doi.org/10.1038/35037710.
Ho FM, Liu SH, Liau CS, Huang PJ, Lin-Shiau SY (2000) High glucose-induced apoptosis in human endothelial cells is mediated by sequential activations of c-Jun NH(2)-terminal kinase and caspase-3. Circulation 101(22):2618–2624. https://doi.org/10.1161/01.CIR.101.22.2618
Hockenbery DM, Oltvai ZN, Yin XM, Milliman CL, Korsmeyer SJ (1993) Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 75(2):241–251. https://doi.org/10.1016/0092-8674(93)80066-N
Hommes DW, Peppelenbosch MP, van Deventer SJ (2003) Mitogen activated protein (MAP) kinase signal transduction pathways and novel anti-inflammatory targets. Gut 52(1):144–151. https://doi.org/10.1136/gut.52.1.144
Ichijo H, Nishida E, Irie K, ten Dijke P, Saitoh M, Moriguchi T, Takagi M, Matsumoto K, Miyazono K, Gotoh Y (1997) Induction of apoptosis by ASK1, a mammalian MAPKKK that activates SAPK/JNK and p38 signaling pathways. Science 275(5296):90–94. https://doi.org/10.1126/science.275.5296.90
Juretic N, Santibáñez JF, Hurtado C, Martínez J (2001) ERK 1,2 and p38 pathways are involved in the proliferative stimuli mediated by urokinase in osteoblastic SaOS-2 cell line. J Cell Biochem 83(1):92–98. https://doi.org/10.1002/jcb.1211
Kane DJ, Sarafian TA, Anton R, Hahn H, Gralla EB, Valentine JS, Ord T, Bredesen DE (1993) Bcl-2 inhibition of neural death: decreased generation of reactive oxygen species. Science 262(5137):1274–1277. https://doi.org/10.1126/science.8235659
Kao LS, Morris BH, Lally KP, Stewart CD, Huseby V, Kennedy KA (2006) Hyperglycemia and morbidity and mortality in extremely low birth weight infants. J Perinatol 26(12):730–736. https://doi.org/10.1038/sj.jp.7211593
Kermer P, Klöcker N, Labes M, Bähr M (2000) Insulin-like growth factor-I protects axotomized rat retinal ganglion cells from secondary death via PI3-K-dependent Akt phosphorylation and inhibition of caspase-3 in vivo. J Neurosci 20(2):2–8
Klein D, Kern RM, Sokol RZ (1995) A method for quantification and correction of proteins after transfer to immobilization membranes. Biochem Mol Biol Int 36(1):59–66
Kruse MS, Barutta J, Vega MC, Coirini H (2012) Down regulation of the proliferation and apoptotic pathways in the embryonic brain of diabetic rats. Cell Mol Neurobiol 32(6):1031–1037. https://doi.org/10.1007/s10571-012-9820-8
Kummer JL, Rao PK, Heidenreich KA (1997) Apoptosis induced by withdrawal of trophic factors is mediated by p38 mitogen-activated protein kinase. J Biol Chem 272(33):20490–20494. https://doi.org/10.1074/jbc.272.33.20490
Kyosseva SV (2004) Mitogen-activated protein kinase signaling. Int Rev Neurobiol 59:201–220. https://doi.org/10.1016/S0074-7742(04)59008-6
Kyriakis JM, Avruch J (2012) Mammalian MAPK signal transduction pathways activated by stress and inflammation: a 10-year update. Physiol Rev 92(2):689–737. https://doi.org/10.1152/physrev.00028.2011
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685. https://doi.org/10.1038/227680a0
Li ZG, Sima AA (2004) C-peptide and central nervous system complications in diabetes. Exp Diabesity Res 5(1):79–90. https://doi.org/10.1080/15438600490424550
Liu D, Zhang H, Gu W, Liu Y, Zhang M (2013) Neuroprotective effects of ginsenoside Rb1 on high glucose-induced neurotoxicity in primary cultured rat hippocampal neurons. PLoS One 8(11):e79399. https://doi.org/10.1371/journal.pone.0079399
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
McCubrey JA, Lahair MM, Franklin RA (2006) Reactive oxygen species-induced activation of the MAP kinase signaling pathways. Antioxid Redox Signal 8(9–10):1775–1789. https://doi.org/10.1089/ars.2006.8.1775
McLean WG (1997) The role of axonal cytoskeleton in diabetic neuropathy. Neurochem Res 22(8):951–956. https://doi.org/10.1023/A:1022466624223
Miyashita K, Kawakami K, Nakada M, Mai W, Shakoori A, Fujisawa H, Hayashi Y, Hamada J, Minamoto T (2009) Potential therapeutic effect of glycogen synthase kinase 3beta inhibition against human glioblastoma. Clin Cancer Res 15(3):887–897. https://doi.org/10.1158/1078-0432.CCR-08-0760
Nansseu JR, Ngo-Um SS, Balti EV (2016) Incidence, prevalence and genetic determinants of neonatal diabetes mellitus: a systematic review and meta-analysis protocol. Syst Rev 5(1):188. https://doi.org/10.1186/s13643-016-0369-3
NCD Risk Factor Collaboration (NCD-RisC) (2016) Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4.4 million participants. Lancet 387(10027):1513–1530. https://doi.org/10.1016/S0140-6736(16)00618-8
Nikoulina SE, Ciaraldi TP, Mudaliar S, Mohideen P, Carter L, Henry RR (2000) Potential role of glycogen synthase kinase-3 in skeletal muscle insulin resistance of type 2 diabetes. Diabetes 49(2):263–271. https://doi.org/10.2337/diabetes.49.2.263
Oltvai ZN, Milliman CL, Korsmeyer SJ (1993) Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74(4):609–619. https://doi.org/10.1016/0092-8674(93)90509-O
Politi LE, Rotstein NP, Salvador G, Giusto NM, Insua MF (2001) Insulin-like growth factor-I is a potential trophic factor for amacrine cells. J Neurochem 76(4):1199–1211. https://doi.org/10.1046/j.1471-4159.2001.00128.x
Quinn L (2001) Type 2 diabetes: epidemiology, pathophysiology, and diagnosis. Nurs Clin North Am 36(2):175–192 v
Ramakrishnan R, Kempuraj D, Prabhakaran K, Jayakumar AR, Devi RS, Suthanthirarajan N, Namasivayam A (2005) A short-term diabetes induced changes of catecholamines and p38-MAPK in discrete areas of rat brain. Life Sci 77(15):1825–1835. https://doi.org/10.1016/j.lfs.2004.12.038
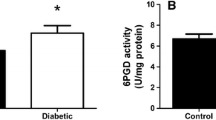
Rosa AP, Jacques CE, de Souza LO, Bitencourt F, Mazzola PN, Coelho JG, Mescka CP, Dutra-Filho CS (2015) Neonatal hyperglycemia induces oxidative stress in the rat brain: the role of pentose phosphate pathway enzymes and NADPH oxidase. Mol Cell Biochem 403(1–2):159–167. https://doi.org/10.1007/s11010-015-2346-x
Sabapathy K (2012) Role of the JNK pathway in human diseases. Prog Mol Biol Transl Sci 106:145–169. https://doi.org/10.1016/B978-0-12-396456-4.00013-4
Sagili H, Kamalanathan S, Sahoo J, Lakshminarayanan S, Rani R, Jayalakshmi D, Kumar KT (2015) Comparison of different criteria for diagnosis of gestational diabetes mellitus. Indian J Endocrinol Metab 19(6):824–828. https://doi.org/10.4103/2230-8210.167550
Scheid MP, Woodgett JR (2001) PKB/AKT: functional insights from genetic models. Nat Rev Mol Cell Biol 2(10):760–768. https://doi.org/10.1038/35096067
Schmued LC, Stowers CC, Scallet AC, Xu L (2005) Fluoro-jade C results in ultra high resolution and contrast labeling of degenerating neurons. Brain Res 1035(1):24–31. https://doi.org/10.1016/j.brainres.2004.11.054
Soetikno V, Sari FR, Sukumaran V, Lakshmanan AP, Mito S, Harima M, Thandavarayan RA, Suzuki K, Nagata M, Takagi R, Watanabe K (2012) Curcumin prevents diabetic cardiomyopathy in streptozotocin-induced diabetic rats: possible involvement of PKC-MAPK signaling pathway. Eur J Pharm Sci 47(3):604–614. https://doi.org/10.1016/j.ejps.2012.04.018
Srinivasan V, Spinella PC, Drott HR, Roth CL, Helfaer MA, Nadkarni V (2004) Association of timing, duration, and intensity of hyperglycemia with intensive care unit mortality in critically ill children. Pediatr Crit Care Med 5(4):329–336. https://doi.org/10.1097/01.PCC.0000128607.68261.7C
Suzuki S, Makita Y, Mukai T, Matsuo K, Ueda O, Fujieda K (2007) Molecular basis of neonatal diabetes in Japanese patients. J Clin Endocrinol Metab 92(10):3979–3985. https://doi.org/10.1210/jc.2007-0486
Thomas J, Garg ML, Smith DW (2013) Altered expression of histone and synaptic plasticity associated genes in the hippocampus of streptozotocin-induced diabetic mice. Metab Brain Dis 28(4):613–618. https://doi.org/10.1007/s11011-013-9418-y
Tomlinson DR, Gardiner NJ (2008) Glucose neurotoxicity. Nat Rev Neurosci 9(1):36–45. https://doi.org/10.1038/nrn2294
Torres M, Forman HJ (2003) Redox signaling and the MAP kinase pathways. Biofactors 17(1–4):287–296. https://doi.org/10.1002/biof.5520170128
Venkatachalam K, Mummidi S, Cortez DM, Prabhu SD, Valente AJ, Chandrasekar B (2008) Resveratrol inhibits high glucose-induced PI3K/Akt/ERK-dependent interleukin-17 expression in primary mouse cardiac fibroblasts. Am J Physiol Heart Circ Physiol 294(5):H2078–H2087. https://doi.org/10.1152/ajpheart.01363.2007
Wild S, Roglic G, Green A, Sicree R, King H (2004) Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 27(5):1047–1053. https://doi.org/10.2337/diacare.27.5.1047
Wu KJ, Chen YF, Tsai HY, Wu CR, Wood WG (2012) Guizhi-Fuling-wan, a traditional Chinese herbal medicine, ameliorates memory deficits and neuronal apoptosis in the Streptozotocin-induced hyperglycemic rodents via the decrease of Bax/Bcl2 ratio and Caspase-3 expression. Evid Based Complement Alternat Med 2012:656150–656111. https://doi.org/10.1155/2012/656150
Xi X, Gao L, Hatala DA, Smith DG, Codispoti MC, Gong B, Kern TS, Zhang JZ (2005) Chronically elevated glucose-induced apoptosis is mediated by inactivation of Akt in cultured Müller cells. Biochem Biophys Res Commun 326(3):548–553. https://doi.org/10.1016/j.bbrc.2004.11.064
Xia Z, Dickens M, Raingeaud J, Davis RJ, Greenberg ME (1995) Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science 270(5240):1326–1331. https://doi.org/10.1126/science.270.5240.1326
Yang RH, Lin J, Hou XH, Cao R, Yu F, Liu HQ, Ji AL, Xu XN, Zhang L, Wang F (2014) Effect of docosahexaenoic acid on hippocampal neurons in high-glucose condition: involvement of PI3K/AKT/nuclear factor-κB-mediated inflammatory pathways. Neuroscience 274:218–228. https://doi.org/10.1016/j.neuroscience.2014.05.042
Yang CM, Lin CC, Hsieh HL (2016) High-glucose-derived oxidative stress-dependent Heme Oxygenase-1 expression from astrocytes contributes to the neuronal apoptosis. Mol Neurobiol 54(1):470–483. https://doi.org/10.1007/s12035-015-9666-4
Ye L, Wang F, Yang RH (2011) Diabetes impairs learning performance and affects the mitochondrial function of hippocampal pyramidal neurons. Brain Res 1411:57–64. https://doi.org/10.1016/j.brainres.2011.07.011
Youle RJ, Strasser A (2008) The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 9(1):47–59. https://doi.org/10.1038/nrm2308
Zhang T, Jia W, Sun X (2010) 3-n-butylphthalide (NBP) reduces apoptosis and enhances vascular endothelial growth factor (VEGF) up-regulation in diabetic rats. Neurol Res 32(4):390–396. https://doi.org/10.1179/016164110X12670144526264
Acknowledgements
This work was supported by research grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Programa de Núcleos de Excelência (PRONEX), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rosa, A.P., Mescka, C.P., Catarino, F.M. et al. Neonatal hyperglycemia induces cell death in the rat brain. Metab Brain Dis 33, 333–342 (2018). https://doi.org/10.1007/s11011-017-0170-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-017-0170-6