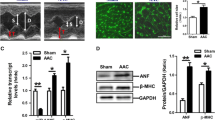
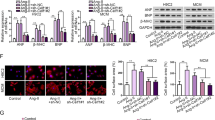
Abstract
Stromal interaction molecule 1 (STIM1) is the key molecule responsible for store-operated Ca2+ entry (SOCE). Numerous studies have demonstrated that STIM1 levels appeared to be enhanced during cardiac hypertrophy. However, the mechanism underlining this process remains to be clarified. In this study, phenylephrine (PE) was employed to establish a model of hypertrophic neonatal rat cardiomyocytes (HNRCs) in vitro, and low expression of primary and mature miR-223 was detected in PE-induced HNRCs. Our results have revealed that downregulation of miR-223 by PE contributed to the increase of STIM1, which in turn induced cardiac hypertrophy. As expected, overexpression of miR-223 could prevent the increase in cell surface and reduce the mRNA levels of ANF and BNP in cardiomyocytes. To address the mechanism triggering downregulation of miR-223 under PE, we demonstrated that PE-induced inhibition of GSK-3β activity led to the activation of β-catenin, which initiates the transcription of SOX2. Increased expression of SOX2 occupied the promoter region of primary miR-223 and suppressed its transcription. Therefore, miR-223 appears to be a promising candidate for inhibiting cardiomyocyte hypertrophy, and miR-223/STIM1 axis might be one of interesting targets for the clinical treatment of hypertrophy.




Similar content being viewed by others
References
Bisping E, Wakula P, Poteser M, Heinzel FR (2014) Targeting cardiac hypertrophy: toward a causal heart failure therapy. J Cardiovasc Pharmacol 64:293–305. doi:10.1097/FJC.0000000000000126
Shimizu I, Minamino T (2016) Physiological and pathological cardiac hypertrophy. J Mol Cell Cardiol 97:245–262. doi:10.1016/j.yjmcc.2016.06.001
Tojyo Y, Morita T, Nezu A, Tanimura A (2014) Key components of store-operated Ca2+ entry in non-excitable cells. J Pharmacol Sci 125:340–346
Majewski L, Kuznicki J (2015) SOCE in neurons: signaling or just refilling? Biochim Biophys Acta 1853:1940–1952. doi:10.1016/j.bbamcr.2015.01.019
Pan Z, Brotto M, Ma J (2014) Store-operated Ca2+ entry in muscle physiology and diseases. BMB Rep 47:69–79
Lopez JJ, Albarran L, Gomez LJ, Smani T, Salido GM, Rosado JA (2016) Molecular modulators of store-operated calcium entry. Biochim Biophys Acta 1863:2037–2043. doi:10.1016/j.bbamcr.2016.04.024
Luo X, Hojayev B, Jiang N, Wang ZV, Tandan S, Rakalin A, Rothermel BA, Gillette TG, Hill JA (2012) STIM1-dependent store-operated Ca(2)(+) entry is required for pathological cardiac hypertrophy. J Mol Cell Cardiol 52:136–147. doi:10.1016/j.yjmcc.2011.11.003
Voelkers M, Salz M, Herzog N, Frank D, Dolatabadi N, Frey N, Gude N, Friedrich O, Koch WJ, Katus HA, Sussman MA, Most P (2010) Orai1 and Stim1 regulate normal and hypertrophic growth in cardiomyocytes. J Mol Cell Cardiol 48:1329–1334. doi:10.1016/j.yjmcc.2010.01.020
Ohba T, Watanabe H, Murakami M, Sato T, Ono K, Ito H (2009) Essential role of STIM1 in the development of cardiomyocyte hypertrophy. Biochem Biophys Res Commun 389:172–176. doi:10.1016/j.bbrc.2009.08.117
Xue J, Chi Y, Chen Y, Huang S, Ye X, Niu J, Wang W, Pfeffer LM, Shao ZM, Wu ZH, Wu J (2016) MiRNA-621 sensitizes breast cancer to chemotherapy by suppressing FBXO11 and enhancing p53 activity. Oncogene 35:448–458. doi:10.1038/onc.2015.96
Ambros V (2001) microRNAs: tiny regulators with great potential. Cell 107:823–826
Gladka MM, da Costa Martins PA, De Windt LJ (2012) Small changes can make a big difference—microRNA regulation of cardiac hypertrophy. J Mol Cell Cardiol 52:74–82. doi:10.1016/j.yjmcc.2011.09.015
Ganesan J, Ramanujam D, Sassi Y, Ahles A, Jentzsch C, Werfel S, Leierseder S, Loyer X, Giacca M, Zentilin L, Thum T, Laggerbauer B, Engelhardt S (2013) MiR-378 controls cardiac hypertrophy by combined repression of mitogen-activated protein kinase pathway factors. Circulation 127:2097–2106. doi:10.1161/CIRCULATIONAHA.112.000882
Care A, Catalucci D, Felicetti F, Bonci D, Addario A, Gallo P, Bang ML, Segnalini P, Gu Y, Dalton ND, Elia L, Latronico MV, Hoydal M, Autore C, Russo MA, Dorn GW 2nd, Ellingsen O, Ruiz-Lozano P, Peterson KL, Croce CM, Peschle C, Condorelli G (2007) MicroRNA-133 controls cardiac hypertrophy. Nat Med 13:613–618. doi:10.1038/nm1582
Wang K, Lin ZQ, Long B, Li JH, Zhou J, Li PF (2012) Cardiac hypertrophy is positively regulated by MicroRNA miR-23a. J Biol Chem 287:589–599. doi:10.1074/jbc.M111.266940
Wang W, Huang X, Xin HB, Fu M, Xue A, Wu ZH (2015) TRAF family member-associated NF-kappaB activator (TANK) inhibits genotoxic nuclear factor kappaB activation by facilitating deubiquitinase USP10-dependent deubiquitination of TRAF6 ligase. J Biol Chem 290:13372–13385. doi:10.1074/jbc.M115.643767
Niu J, Xue A, Chi Y, Xue J, Wang W, Zhao Z, Fan M, Yang CH, Shao ZM, Pfeffer LM, Wu J, Wu ZH (2016) Induction of miRNA-181a by genotoxic treatments promotes chemotherapeutic resistance and metastasis in breast cancer. Oncogene 35:1302–1313. doi:10.1038/onc.2015.189
Zhao J, Cheng F, Wang Y, Arteaga CL, Zhao Z (2016) Systematic prioritization of druggable mutations in approximately 5000 genomes across 16 cancer types using a structural genomics-based approach. Mol Cell Proteomics 15:642–656. doi:10.1074/mcp.M115.053199
Feng HJ, Ouyang W, Liu JH, Sun YG, Hu R, Huang LH, Xian JL, Jing CF, Zhou MJ (2014) Global microRNA profiles and signaling pathways in the development of cardiac hypertrophy. Braz J Med Biol Res 47:361–368
Wasserman WW, Sandelin A (2004) Applied bioinformatics for the identification of regulatory elements. Nat Rev Genet 5:276–287. doi:10.1038/nrg1315
Heallen T, Zhang M, Wang J, Bonilla-Claudio M, Klysik E, Johnson RL, Martin JF (2011) Hippo pathway inhibits Wnt signaling to restrain cardiomyocyte proliferation and heart size. Science 332:458–461. doi:10.1126/science.1199010
Blankesteijn WM, van de Schans VA, ter Horst P, Smits JF (2008) The Wnt/frizzled/GSK-3 beta pathway: a novel therapeutic target for cardiac hypertrophy. Trends Pharmacol Sci 29:175–180. doi:10.1016/j.tips.2008.01.003
Collins HE, Zhu-Mauldin X, Marchase RB, Chatham JC (2013) STIM1/Orai1-mediated SOCE: current perspectives and potential roles in cardiac function and pathology. Am J Physiol Heart Circ Physiol 305:H446–H458. doi:10.1152/ajpheart.00104.2013
Putney JW (2013) Alternative forms of the store-operated calcium entry mediators, STIM1 and Orai1. Curr Top Membr 71:109–123. doi:10.1016/B978-0-12-407870-3.00005-6
Baba Y, Matsumoto M, Kurosaki T (2015) Signals controlling the development and activity of regulatory B-lineage cells. Int Immunol 27:487–493. doi:10.1093/intimm/dxv027
Pozo-Guisado E, Martin-Romero FJ (2013) The regulation of STIM1 by phosphorylation. Commun Integr Biol 6:e26283. doi:10.4161/cib.26283
Lang F, Eylenstein A, Shumilina E (2012) Regulation of Orai1/STIM1 by the kinases SGK1 and AMPK. Cell Calcium 52:347–354. doi:10.1016/j.ceca.2012.05.005
Zhao FY, Han J, Chen XW, Wang J, Wang XD, Sun JG, Chen ZT (2016) miR-223 enhances the sensitivity of non-small cell lung cancer cells to erlotinib by targeting the insulin-like growth factor-1 receptor. Int J Mol Med 38:183–191. doi:10.3892/ijmm.2016.2588
Yang W, Lan X, Li D, Li T, Lu S (2015) MiR-223 targeting MAFB suppresses proliferation and migration of nasopharyngeal carcinoma cells. BMC Cancer 15:461. doi:10.1186/s12885-015-1464-x
Aziz F (2016) The emerging role of miR-223 as novel potential diagnostic and therapeutic target for inflammatory disorders. Cell Immunol 303:1–6. doi:10.1016/j.cellimm.2016.04.003
Taibi F, Metzinger-Le Meuth V, Massy ZA, Metzinger L (2014) miR-223: an inflammatory oncomiR enters the cardiovascular field. Biochim Biophys Acta 1842:1001–1009. doi:10.1016/j.bbadis.2014.03.005
Wang YS, Zhou J, Hong K, Cheng XS, Li YG (2015) MicroRNA-223 displays a protective role against cardiomyocyte hypertrophy by targeting cardiac troponin I-interacting kinase. Cell Physiol Biochem 35:1546–1556. doi:10.1159/000373970
Yang L, Li Y, Wang X, Mu X, Qin D, Huang W, Alshahrani S, Nieman M, Peng J, Essandoh K, Peng T, Wang Y, Lorenz J, Soleimani M, Zhao ZQ, Fan GC (2016) Overexpression of miR-223 tips the balance of pro- and anti-hypertrophic signaling cascades toward physiologic cardiac hypertrophy. J Biol Chem 291:15700–15713. doi:10.1074/jbc.M116.715805
ter Horst P, Smits JF, Blankesteijn WM (2012) The Wnt/Frizzled pathway as a therapeutic target for cardiac hypertrophy: where do we stand? Acta Physiol (Oxf) 204:110–117. doi:10.1111/j.1748-1716.2011.02309.x
De Windt LJ, Lim HW, Haq S, Force T, Molkentin JD (2000) Calcineurin promotes protein kinase C and c-Jun NH2-terminal kinase activation in the heart. Cross-talk between cardiac hypertrophic signaling pathways. J Biol Chem 275:13571–13579
Morisco C, Zebrowski D, Condorelli G, Tsichlis P, Vatner SF, Sadoshima J (2000) The Akt-glycogen synthase kinase 3beta pathway regulates transcription of atrial natriuretic factor induced by beta-adrenergic receptor stimulation in cardiac myocytes. J Biol Chem 275:14466–14475
Haq S, Michael A, Andreucci M, Bhattacharya K, Dotto P, Walters B, Woodgett J, Kilter H, Force T (2003) Stabilization of beta-catenin by a Wnt-independent mechanism regulates cardiomyocyte growth. Proc Natl Acad Sci USA 100:4610–4615. doi:10.1073/pnas.0835895100
Feng R, Wen J (2015) Overview of the roles of Sox2 in stem cell and development. Biol Chem 396:883–891. doi:10.1515/hsz-2014-0317
Fukao T, Fukuda Y, Kiga K, Sharif J, Hino K, Enomoto Y, Kawamura A, Nakamura K, Takeuchi T, Tanabe M (2007) An evolutionarily conserved mechanism for microRNA-223 expression revealed by microRNA gene profiling. Cell 129:617–631. doi:10.1016/j.cell.2007.02.048
Fazi F, Rosa A, Fatica A, Gelmetti V, De Marchis ML, Nervi C, Bozzoni I (2005) A minicircuitry comprised of microRNA-223 and transcription factors NFI-A and C/EBPalpha regulates human granulopoiesis. Cell 123:819–831. doi:10.1016/j.cell.2005.09.023
Acknowledgements
This study was supported by the Chinese Natural Science Foundation Grants (81303132), the Science and Technology Development Special Fund of Shanghai Health and Family Planning Commission (ZK2015A17), the Science and Technology Development Special Fund of Zhoupu Hospital (ZP2014A-01).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11010_2017_3209_MOESM3_ESM.pdf
Supplementary material 3 Supplementary Figure 1. miR-223 expression value in neonatal rat cardiomyocytes from 5-day TCA induction model (GEO/GSE38599). Supplementary Figure 2. Mature miR-223 expression level was examined by qPCR after infection of mir-223. (PDF 921 kb)
Rights and permissions
About this article
Cite this article
Zhao, ZH., Luo, J., Li, Hx. et al. SOX2-mediated inhibition of miR-223 contributes to STIM1 activation in phenylephrine-induced hypertrophic cardiomyocytes. Mol Cell Biochem 443, 47–56 (2018). https://doi.org/10.1007/s11010-017-3209-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-017-3209-4