Abstract
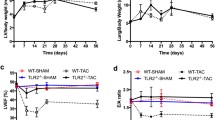
Fibroblast growth factor-2 (FGF-2) is implicated in cardioprotection. However, previously we found that chronic elevation in cardiac FGF-2 levels in transgenic mice was associated with exaggerated, cyclosporine A-preventable, cellular infiltration after isoproterenol-induced injury, suggestive of an adverse outcome, although this was not examined with functional studies. We have now used highly sensitive tissue Doppler imaging (TDI) to evaluate cardiac functional parameters after isoproterenol administration in transgenic mice overexpressing the 18 kDa FGF-2 in the heart in vivo. Cardiac function was assessed in conscious FGF-2 transgenic and non-transgenic mice at 24 h as well as 2 and 4 weeks after isoproterenol administration, and in the absence or presence of either cyclosporine A or anti-CD3ε treatments. Isoproterenol decreased left ventricular endocardial velocity and strain rate by 47–51% at 24 h in non-transgenic mice, but to a significantly lesser extent (by 24%) in transgenic mice. While additional decreases were seen in non-transgenic mice at 2 weeks, there was no further reduction in ventricular endocardial velocity or strain rate up to 4 weeks post-treatment in FGF-2 transgenic mice. Functional improvement at 2 and 4 weeks post-isoproterenol was reduced significantly by treatment with cyclosporine A but not anti-CD3ε; the latter targets T lymphocyte activation more specifically. TDI values in the presence of chronic FGF-2 overexpression are prognostic of an improved cardiac outcome and protection from isoproterenol induced cardiac dysfunction in vivo. Our data also suggest that cyclosporine A-sensitive infiltrating cell population(s) may contribute to the sustained beneficial effect of FGF-2 in vivo.





Similar content being viewed by others
References
Detillieux KA, Sheikh F, Kardami E, Cattini PA (2003) Biological activities of fibroblast growth factor-2 in the adult myocardium. Cardiovasc Res 57:8–19
Jiang ZS, Padua RR, Ju H, Doble BW, Jin Y, Hao J, Cattini PA, Dixon IM, Kardami E (2002) Acute protection of ischemic heart by FGF-2: involvement of FGF-2 receptors and protein kinase C. Am J Physiol Heart Circ Physiol 282:H1071–H1080
Jiang ZS, Srisakuldee W, Soulet F, Bouche G, Kardami E (2004) Non-angiogenic FGF-2 protects the ischemic heart from injury, in the presence or absence of reperfusion. Cardiovasc Res 62:154–166
Padua RR, Merle PL, Doble BW, Yu CH, Zahradka P, Pierce GN, Panagia V, Kardami E (1998) FGF-2-induced negative inotropism and cardioprotection are inhibited by chelerythrine: involvement of sarcolemmal calcium-independent protein kinase C. J Mol Cell Cardiol 30:2695–2709
Padua RR, Sethi R, Dhalla NS, Kardami E (1995) Basic fibroblast growth factor is cardioprotective in ischemia-reperfusion injury. Mol Cell Biochem 143:129–135
Virag JA, Rolle ML, Reece J, Hardouin S, Feigl EO, Murry CE (2007) Fibroblast growth factor-2 regulates myocardial infarct repair: effects on cell proliferation, scar contraction, and ventricular function. Am J Pathol 171:1431–1440
Jiang ZS, Wen GB, Tang ZH, Srisakuldee W, Fandrich RR, Kardami E (2009) High molecular weight FGF-2 promotes postconditioning-like cardioprotection linked to activation of protein kinase C isoforms, as well as Akt and p70 S6 kinases [corrected]. Can J Physiol Pharmacol 87:798–804
Detillieux KA, Cattini PA, Kardami E (2004) Beyond angiogenesis: the cardioprotective potential of fibroblast growth factor-2. Can J Physiol Pharmacol 82:1044–1052
Kardami E, Detillieux K, Ma X, Jiang Z, Santiago JJ, Jimenez SK, Cattini PA (2007) Fibroblast growth factor-2 and cardioprotection. Heart Fail Rev 12:267–277
Clarke MS, Caldwell RW, Chiao H, Miyake K, McNeil PL (1995) Contraction-induced cell wounding and release of fibroblast growth factor in heart. Circ Res 76:927–934
Kaye D, Pimental D, Prasad S, Maki T, Berger HJ, McNeil PL, Smith TW, Kelly RA (1996) Role of transiently altered sarcolemmal membrane permeability and basic fibroblast growth factor release in the hypertrophic response of adult rat ventricular myocytes to increased mechanical activity in vitro. J Clin Invest 97:281–291
Jimenez SK, Sheikh F, Jin Y, Detillieux KA, Dhaliwal J, Kardami E, Cattini PA (2004) Transcriptional regulation of FGF-2 gene expression in cardiac myocytes. Cardiovasc Res 62:548–557
Sheikh F, Sontag DP, Fandrich RR, Kardami E, Cattini PA (2001) Overexpression of FGF-2 increases cardiac myocyte viability after injury in isolated mouse hearts. Am J Physiol Heart Circ Physiol 280:H1039–H1050
House SL, Bolte C, Zhou M, Doetschman T, Klevitsky R, Newman G, Schultz Jel J (2003) Cardiac-specific overexpression of fibroblast growth factor-2 protects against myocardial dysfunction and infarction in a murine model of low-flow ischemia. Circulation 108:3140–3148
Meij JT, Sheikh F, Jimenez SK, Nickerson PW, Kardami E, Cattini PA (2002) Exacerbation of myocardial injury in transgenic mice overexpressing FGF-2 is T cell dependent. Am J Physiol Heart Circ Physiol 282:H547–H555
Campos EC, Romano MM, Prado CM, Rossi MA (2008) Isoproterenol induces primary loss of dystrophin in rat hearts: correlation with myocardial injury. Int J Exp Pathol 89:367–381
Grimm D, Elsner D, Schunkert H, Pfeifer M, Griese D, Bruckschlegel G, Muders F, Riegger GA, Kromer EP (1998) Development of heart failure following isoproterenol administration in the rat: role of the renin-angiotensin system. Cardiovasc Res 37:91–100
Rona G, Chappel CI, Balazs T, Gaudry R (1959) An infarct-like myocardial lesion and other toxic manifestations produced by isoproterenol in the rat. AMA Arch Pathol 67:443–455
Teerlink JR, Pfeffer JM, Pfeffer MA (1994) Progressive ventricular remodeling in response to diffuse isoproterenol-induced myocardial necrosis in rats. Circ Res 75:105–113
Zhou R, Xu Q, Zheng P, Yan L, Zheng J, Dai G (2008) Cardioprotective effect of fluvastatin on isoproterenol-induced myocardial infarction in rat. Eur J Pharmacol 586:244–250
Hess AD (1993) Mechanisms of action of cyclosporine: considerations for the treatment of autoimmune diseases. Clin Immunol Immunopathol 68:220–228
Hirsch R, Bluestone JA, DeNenno L, Gress RE (1990) Anti-CD3 F(ab’)2 fragments are immunosuppressive in vivo without evoking either the strong humoral response or morbidity associated with whole mAb. Transplantation 49:1117–1123
Chetboul V, Escriou C, Tessier D, Richard V, Pouchelon JL, Thibault H, Lallemand F, Thuillez C, Blot S, Derumeaux G (2004) Tissue Doppler imaging detects early asymptomatic myocardial abnormalities in a dog model of Duchenne’s cardiomyopathy. Eur Heart J 25:1934–1939
Neilan TG, Jassal DS, Perez-Sanz TM, Raher MJ, Pradhan AD, Buys ES, Ichinose F, Bayne DB, Halpern EF, Weyman AE, Derumeaux G, Bloch KD, Picard MH, Scherrer-Crosbie M (2006) Tissue Doppler imaging predicts left ventricular dysfunction and mortality in a murine model of cardiac injury. Eur Heart J 27:1868–1875
Sebag IA, Handschumacher MD, Ichinose F, Morgan JG, Hataishi R, Rodrigues AC, Guerrero JL, Steudel W, Raher MJ, Halpern EF, Derumeaux G, Bloch KD, Picard MH, Scherrer-Crosbie M (2005) Quantitative assessment of regional myocardial function in mice by tissue Doppler imaging: comparison with hemodynamics and sonomicrometry. Circulation 111:2611–2616
Srisakuldee W, Nickel BE, Fandrich RR, Jiang ZS, Kardami E (2006) Administration of FGF-2 to the heart stimulates connexin-43 phosphorylation at protein kinase C target sites. Cell Commun Adhes 13:13–19
Colella M, Pozzan T (2008) Cardiac cell hypertrophy in vitro: role of calcineurin/NFAT as Ca2+ signal integrators. Ann N Y Acad Sci 1123:64–68
Halestrap AP, Pasdois P (2009) The role of the mitochondrial permeability transition pore in heart disease. Biochim Biophys Acta 1787:1402–1415
Koulmann N, Novel-Chate V, Peinnequin A, Chapot R, Serrurier B, Simler N, Richard H, Ventura-Clapier R, Bigard X (2006) Cyclosporin A inhibits hypoxia-induced pulmonary hypertension and right ventricle hypertrophy. Am J Respir Crit Care Med 174:699–705
Jiang ZS, Jeyaraman M, Wen GB, Fandrich RR, Dixon IM, Cattini PA, Kardami E (2007) High- but not low-molecular weight FGF-2 causes cardiac hypertrophy in vivo; possible involvement of cardiotrophin-1. J Mol Cell Cardiol 42:222–233
Schultz JE, Witt SA, Nieman ML, Reiser PJ, Engle SJ, Zhou M, Pawlowski SA, Lorenz JN, Kimball TR, Doetschman T (1999) Fibroblast growth factor-2 mediates pressure-induced hypertrophic response. J Clin Invest 104:709–719
Jimenez SK, Jassal DS, Kardami E and Cattini PA (2011) A single bout of exercise promotes sustained left ventricular function improvement after isoproterenol-induced injury in mice. J Physiol Sci. doi:10.1007/s12576-011-0147-x
Acknowledgments
The authors acknowledge the contribution of Ms. Karen Detillieux to the development of this article, and thank Ms. Tielan Fang for technical assistance. This work was supported by a grant from the Canadian Institutes of Health Research (FRN-74733).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jimenez, S.K., Jassal, D.S., Kardami, E. et al. Protection by endogenous FGF-2 against isoproterenol-induced cardiac dysfunction is attenuated by cyclosporine A. Mol Cell Biochem 357, 1–8 (2011). https://doi.org/10.1007/s11010-011-0868-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-0868-4