Abstract
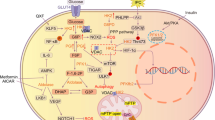
Exogenous insulin therapy improves endothelial function in insulin resistant patients, indirectly indicating that nitric oxide synthase activity and NO production may be impaired. Insulin stimulates production of NO by activating a signaling pathway including insulin receptor substrate-1, phosphatidylinositol-3-kinase and protein kinase B (PKB/Akt). Angiotensin II type I (AT1) receptor-evoked oxidative stress is implicated in the inactivation of NO, impairing endothelium-dependent vasodilatation. Blocking the actions of Angiotensin II with an AT1 receptor antagonist (Losartan), has beneficial effects in patients with insulin resistance or type 2 diabetes mellitus. This study investigated whether elevated Angiotensin II influences myocardial insulin resistance, insulin signaling and NO production in a rat model of diet-induced obesity (DIO) by antagonizing the actions of the AT1 receptor with Losartan. Isolated, perfused hearts, Western blotting and flow-cytometric methods were utilized to determine myocardial function, expression and phosphorylation of key proteins and NO production, respectively. Results showed that hearts from DIO rats are insulin resistant (higher serine phosphorylation of IRS-1, lower insulin-stimulated phosphorylation of PKB/Akt and eNOS, lower NO production) and had poorer functional recovery and larger infarct development after ischaemia/reperfusion. Losartan improved the impaired functional recovery, and NO production and enhanced eNOS expression and phosphorylation and reduced infarct size in hearts from the DIO animals. Data obtained from Losartan treatment also revealed that Angiotensin II signaling modulates myocardial PKB/Akt expression. We conclude that Angiotensin II signaling exacerbates inhibition of NO production in insulin resistance and that this can be improved by AT1 antagonism.







Similar content being viewed by others
References
Sharma AM, Engeli S (2006) The role of rennin-angiotensin system blockade in the management of hypertension associated with the cardiometabolic syndrome. J Cardiometab Syndr 1:29–35
Ruige JB, Assendelft WJ, Dekker JM, Kostense PJ, Heine RJ, Bouter LM (1998) Insulin and risk of cardiovascular disease: a meta-analysis. Circulation 97:996–1001
Du XL, Edelstein D, Dimmeler S, Ju Q, Sui C, Brownlee M (2002) Hyperglycemia inhibits endothelial nitric oxide synthase activity by posttranslational modification at the Akt site. J Clin Invest 108:1341–1348
Cai H, Harrison DG (2002) Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ Res 87:840–844
Landmesser U, Dikalov S, Price SR et al (2003) Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J Clin Invest 111:1201–1209
Li J, Zhao X, Li X, Lerea KM, Olson SC (2007) Angiotensin II type 2 receptor-dependent increases in nitric oxide synthase expression in the pulmonary endothelium is mediated via a G alpha i3.Ras/Raf.MAPK pathway. Am J Physiol Cell Physiol 292:C2185–C2196
Wei Y, Sowers JR, Nistala R et al (2006) Angiotensin II-induced NADPH oxidase activation impairs insulin signaling in skeletal muscle cells. J Biol Chem 281:35137–35146
Vehkavaara S, Mäkimattila S, Schlenzka A, Vakkilainen J, Westerbacka J, Yki-Järvinen H (2006) Insulin therapy improves endothelial function in type 2 diabetes. Arterioscler Thromb Vasc Biol 20:545–550
Zeng G, Nystrom FH, Ravichandran LV et al (2000) Roles for insulin receptor, PI3-kinase, and Akt in insulin-signaling pathways related to production of nitric oxide in human vascular endothelial cells. Circulation 101:1539–1545
Sowers JR (2004) Insulin resistance and hypertension. Am J Physiol Heart Circ Physiol 286:H1597–H1602
Montagnani M, Chen H, Barr VA, Quon MJ (2001) Insulin-stimulated activation of eNOS is independent of Ca2+ but requires phosphorylation by Akt and Ser1179. J Biol Chem 276:30392–30398
Metha PD, Griendling KK (2007) Angiotensin II cell signaling: physiological and pathological effects in the cardiovascular system. Am J Physiol Cell Physiol 292:C82–C97
Werner N, Nickenig G (2003) AT1 receptors in atherosclerosis: biological effects including growth, angiogenesis and apoptosis. Eur Heart J Supp 5:9–13
Brede M, Roell WS, Ritter O et al (2003) Cardiac hypertrophy is associated with decreased eNOS expression in angiotensin AT2 receptor-deficient mice. Hypertension 42:1177–1182
Velloso LA, Folli F, Sun XJ, White MF, Saad MJA, Kahn CR (1996) Cross-talk between the inhsulin and angiotensin signaling systems. Proc Natl Acad Sci USA 93:12490–12495
Andreozzi F, Laratta E, Sciacqua A, Perticone F, Sesti G (2004) Angiotensin II impairs the insulin signaling pathway promoting production of nitric oxide by inducing phosphorylation of insulin receptor substrate-1 on Ser312 and Ser616 in human umbilical vein endothelial cells. Circ Res 94:1211–1218
Shinozaki K, Ayajiki K, Nishio Y, Sungaya T, Kashiwagi A, Okamura T (2004) Evidence for a causal role of the renin-angiotensin system in vascular dysfunction associated with insulin resistance. Hypertension 43:255–262
Dahlöf B, Devereux R, Kjeldsen SE et al (2002) LIFE Study Group. Losartan Intervention for Endpoint reduction in hypertension. Lancet 359:995–1003
Pickavance LC, Tadayyon M, Widdowson PS, Buckinham RE, Wilding JPH (1999) Therapeutic index for rosilitazone in dietary obese rats: separation of efficacy and haemodilution. Br J Pharmacol 128:1570–1576
Marais E, Genade S, Salie R, Huisamen B, Maritz S, Moolman JA, Lochner A (2005) The temporal relationship between p38 MAPK and HSP27 activation in ischaemic and pharmacological preconditioning. Basic Res Cardiol 100:35–47
Huisamen B, Donthi RV, Lochner A (2001) Insulin in combination with vanadate stimulates glucose transport in isolated cardiomyocytes from obese Zucker rats. Cardiovasc Drugs Ther 15:445–452
Strijdom H, Jacobs S, Hattingh S, Page C, Lochner A (2006) Nitric oxide production is higher in rat cardiac microvessel endothelial cells than ventricular cardiomyocytes in baseline and hypoxic conditions: a comparative study. FASEB J 20:314–316
Strijdom H, Muller C, Lochner A (2004) Direct intracellular nitric oxide detection in isolated adult cardiomyocytes: flow cytometric analysis using the fluorescent probe, diaminofluorescein. J Mol Cell Cardiol 37:897–902
Du Toit EF, Nabben M, Lochner A (2005) A potential role for angiotensin II in obesity induced cardiac hypertrophy and ischaemic/reperfusion injury. Basic Res Cardiol 100:346–354
Landmesser U, Hornig B, Drexler H (2004) Endothelial function: a critical determinant in atherosclerosis? Circulation 109:27–33
Galili O, Versari D, Sattler KJ et al (2006) Early experimental obesity is associated with coronary endothelial dysfunction and oxidative stress. Am J Physiol Heart Circ Physiol 292:H904–H911
Jaap AJ, Shore AC, Tooke JE (1997) Relationship of insulin resistance to microvascular dysfunction in subjects with fasting hyperglycaemia. Diabetologia 40:238–243
Naderali EK, Pickavance LC, Wilding John PH, Williams G (2001) Diet-induced endothelial dysfunction in the rat is independent of the degree of increase in total body weight. Clin Sci 100:635–641
Navarro-Cid J, Maeso R, Perez-Vizcaino F et al (1995) Effects of Losartan on blood pressure, metabolic alterations and vascular reactivity in the fructose-induced hypertensive rat. Hypertension 26:1074–1078
Van den Meiracker AH, Admiraal PJJ, Janssen JA et al (1995) Hemodynamic and biochemical effects of the AT1 receptor antagonist Irbesartan in hypertension. Hypertension 25:22–29
Folli F, Kahn CR, Hansen H, Bouchie JL, Feener EP (1997) Angiotensin II inhibits insulin signalling in aortic smooth muscle cells at multiple levels. A potential role of serine phosphorylation in insulin/angiotensin II crosstalk. J Clin Invest 100:2158–2169
Hotta H, Miura T, Miki T et al (2010) Angiotensin II type 1 receptor mediated upregulation of calcineurin activity underlies impairment of cardioprotective signaling in diabetic hearts. Circ Res 106:129–132
Seshiah PN, Weber DS, Tocic P, Valppu L, Taniyama Y, Griendling KK (2002) Angiotensin II stimulation of NAD(P)H oxidase activity—upstream mediators. Circ Res 91:406–413
Henriksen EJ (2007) Improvement of insulin sensitivity by antagonism of the rennin-angiotensin system. Am J Physiol Regul Integr Comp Physiol 293:R974–R980
Unger T, Chung O, Csikos T et al (1996) Angiotensin receptors. J Hypertens 14:3–9
Griendling KK, Lasseque B, Alexander RW (1996) Angiotensin receptors and their therapeutic implications. Annu Rev Pharmacol Toxicol 36:281–306
Kawahara S, Umemoto S, Tanaka M, Umeji K, Matsuda S, Kubo M, Matsuzaki M (2005) Up-regulation of Akt and eNOS induces vascular smooth muscle cell differentiation in hypertension in vivo. J Cardiovasc Pharmacol 45:367–374
Lochner A, Genade S, Moolman JA (2003) Ischemic preconditioning: infarct size is a more reliable endpoint than functional recovery. Basic Res Cardiol 98:337–346
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huisamen, B., Pêrel, S.J.C., Friedrich, S.O. et al. ANG II type I receptor antagonism improved nitric oxide production and enhanced eNOS and PKB/Akt expression in hearts from a rat model of insulin resistance. Mol Cell Biochem 349, 21–31 (2011). https://doi.org/10.1007/s11010-010-0656-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0656-6