Abstract
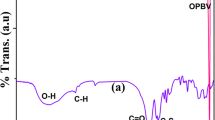
The kinetics of crystallization of poly(ε-caprolactone), PCL, have been measured by FTIR spectroscopy using the absorbance of the crystalline and amorphous phase carbonyl bands at 1725 and 1735 cm−1, respectively, to determine the fractional crystallinity as a function of time and over the temperature range 43–47 °C. A comparison was also made with DSC which was found to have limited sensitivity such that it could only measure the primary stage of the crystallization and not the secondary. Both primary and secondary crystallization could be measured by FTIR spectroscopy with sufficient accuracy to measure the kinetics of each and limited only by the length of time over which the measurements were made.















Similar content being viewed by others
References
Chen Z, Hay JN, Jenkins MJ. FTIR spectroscopic analysis of poly(ethylene terephthalate) on crystallization. Eur Polym J. 2012;48:1586–610.
Chen Z, Hay JN, Jenkins MJ. The kinetics of crystallization of poly(ethylene terephthalate) measured by FTIR spectroscopy. Eur Polym J. 2013;49:1722–30.
Henricks J, Boyum M, Zheng W. Crystallization kinetics and structure evolution of a polylactic acid during melt and cold crystallization. J Therm Anal Calorim. 2015;120:1765–74.
Liu GJ. Isothermal crystallization kinetics of AB2 hyper-branched polymer (HBP)-filled polypropylene (PP). J Therm Anal Calorim. 2014;118:1401–6.
Schawe JEK. Influence of processing conditions on polymer crystallization measured by fast scanning DSC. J Therm Anal Calorim. 2014;116:1165–74.
Avrami MJ. Kinetics of phase change. I. General theory. J Chem Phys. 1939;7:1103–12.
Cheng SZD. Wunderlich B. Modification of the Avrami treatment of crystallization to account for nucleus and interface. Macromolecules. 1988;21:3327–8.
Hillier IH. Modified Avrami equation for the bulk crystallization kinetics of spherulitic polymers. J Polym Sci A. 1965;3:3067–78.
Hay JN, Booth A. The effect of a secondary process on the course of polymer crystallisation. Br Polym J. 1972;4:19–26.
Chen Z, Hay JN, Jenkins MJ. The effect of secondary crystallization on melting. Eur Polym J. 2013;49:2697–703.
Chen Z, Hay JN, Jenkins MJ. Annealing of poly (ethylene terephthalate). Eur Polym J. 2014;50:235–447.
Hoffman JD, Weekes JJ. Rate of spherulitic crystallization with chain folds in polychlorotrifluoroethylene. J Chem Phys. 1962;37:1723–39.
Chan TW, Isayev AI. Quiescent polymer crystallization: modelling and measurements. Polym Eng Sci. 1994;34:461–71.
Bittiger H, Marchessault RH, Niegisch WD. Crystal structure of poly (e-caprolactone). Acta Cryst. 1970;B26:1923–7.
Hoffman JD, Frolen LJ, Ross GS, Lauritzen JI Jr. On the growth rates of spherulites and axialites from the melt in polyethylene fractions: Regime l and ll crystallization. J Res NBS. 1975;79A(6):671–700.
De Gennes PG. Reptation of a polymer chain in the presence of fixed obstacles. J Chem Phys. 1971;55:572–9.
Doi M, Edwards SF. The theory of polymer dynamics. Oxford: Oxford University Press; 1988.
Doi M, Edwards SF. Dynamics of concentrated polymer systems. Part 1, 2 and 3. J Chem Soc Faraday Trans 2. 1978;74:1789–1832.
Kim K, Sperling LH, Klein A, Hammouda B. Reptation time, temperature and cosurfactant effects on the molecular interdiffusion rate between polystyrene latex film formation. Macromolecules. 1994;27:6841–50.
Klein J, Briscoe BJ. The diffusion of long-chain molecules through bulk polyethelene. Proc R Soc Lond. 1979;A365:53–73.
Baumgärtner A, Ebert U, Schafer L. Segment motion in the reptation model of polymer dynamics. II. Simulations. J Stat Phys. 1998;90:1376–400.
El Maaty MIA, Bassett DC. Evidence for isothermal lamellar thickening at and behind the growth front as polyethylene crystallizes from the melt. Polymer. 2005;46:8682–8.
Acknowledgements
We would like to thank Mr. Frank Biddlestone for technical support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Phillipson, K., Jenkins, M.J. & Hay, J.N. The kinetics of crystallization of poly(ε-caprolactone) measured by FTIR spectroscopy. J Therm Anal Calorim 123, 1491–1500 (2016). https://doi.org/10.1007/s10973-015-5047-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-5047-5