Abstract
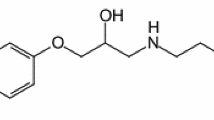
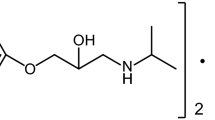
Thermal behavior of β-blocker antihypertensives nadolol and atenolol was investigated using TG/DTG, DTA, and DSC, under dynamic nitrogen and air atmosphere. Nadolol decomposed in a single mass loss step between 196 and 353 °C, after melting at 128.0 °C. In air, an additional event related to the burning of carbonaceous material was observed. Under nitrogen atenolol decomposed also in a single step from 191.8 to 900 °C. However the DTG revealed that this decomposition splits in two successive events resulting in a residue of 17.2 % at the end of the run, with a melting peak at 153.1 °C. In air, an additional step referent to the burning of carbonaceous residue can be seen. Although both presented melting events, only atenolol crystallized on cooling. Evolved gas analysis by TG–FTIR revealed that nadolol decomposes via terc-butylamine releasing, while atenolol loses ammonia in a nonpreviously reported dimerization reaction after melting with a consecutive isopropylamine releasing. A tentative thermal behavior mechanism is presented for both cases.











Similar content being viewed by others
References
Hardman JG, Limbird LE, Gilman AG. The pharmacological basis of therapeutics. 10th ed. New York: McGraw-Hill; 2001 Goodman & Gilman’s.
del Pozo JG, Sevillano ED, de Abajo FJ. Use of antihypertensive drugs in Spain (1995–2001). Rev Esp Cardiol. 2004;57:241–9.
www.sbh.org.br/imprensa/anti_hipertensivos.pdf. Accessed Oct 2011.
Wendlandt WW. Thermal analysis. In: Elving PJ, Winefordner JD, editors. vol 19, 3rd ed., New York: Wiley; 1986.
Lever T, Hains P, Rouquerol J, Charsley EL, Eckeren PV, Burlett DJ. ICTAC nomenclature of thermal analysis (IUPAC) recommendations. Pure Appl Chem. 2014;86:545–53.
Llina`s A, Goodman JM. Polymorph control: past, present and future. Drug Discov Today. 2008;13:198–210.
Nunes RS, Semaan FS, Riga AT, Cavalheiro ETG. Thermal behavior of verapamil and its association with excipients. J Therm Anal Calorm. 2009;97:349–53.
Chieng N, Rades T, Aaltonen J. An overview of recent studies on the analysis of pharmaceutical polymorphs. J Pharm Biomed Anal. 2011;55:618–44.
Ghugare P, Dongre V, Karmuse P, Rana R, Singh D, Kumar A, Filmwala Z. Solid state investigation and characterization of the polymorphic and pseudopolymorphic forms of indapamide. J Pharm Biomed Anal. 2010;51:532–40.
Canotilho J, Castro RAE, Rosado MTS, Nunes SCC, Cruz MSC, Redinha JS. Thermal analysis and crystallization from melts β adrenergic compounds. J Therm Anal Calorim. 2010;100:423–9.
Krill SL, Lau KY, Plachy WZ, Rehfeld SJ. Penetration of dimyristoylphosphatidylcholine monolayers and bilayers by model β-blocker agents of varying lipophilicity. J Pharm Sci. 1998;87:751–6.
Domanska U, Pobdkwska A, Pelczarska A, Winiarska-Tusznio M, Gierycz P. Solubility and pKa of select pharmaceuticals in water, ethanol and 1-octanol. J Chem Thermodyn. 2010;42:1465–72.
Grosvenor MP, Lofroth J. Interaction between bile salts and β-adrenoceptor antagonists. Pharm Res. 1995;12:682–6.
Wesolowski M, Rojek B. Thermogravimetric detection of incompatibilities between atenolol and excipients using multivariate techniques. J Therm Anal Calorim. 2013;113:169–77.
Pereira RN, Valente BR, Cruz AP, Foppa T, Murakami FS, Silva MAS. Thermoanalytical study of atenolol and commercial tablets. Lat Am J Pharm. 2007;26:382–6.
Marini A, Berbenni V, Pegoretti M, Bruni G, Cofrancesco P, Sinistri C, Villa M. Drug-excipient compatibility studies by physico-chemical techniques. The case of atenolol. J Therm Anal Calorim. 2003;73:547–61.
Abou-Sckkina MM, El-Reis MA, Aly FA, Wasse AA. Gamma-promotion of thermal stability and radiolysis mechanism of atenolol beta-blocker. Thermochim Acta. 2002;383:37–44.
Pyramides G, Robinson JW, Zito SW. The combined use of DSC and TGA for the thermal-analysis of atenolol tablets. J Pharm Biomed Anal. 1995;13:103–10.
Ficarra R, Ficarra P, Di Bella MR, Raneri D, Tommasini S, Calabrò ML, Gamberini MC, Rustichelli C. Study of beta-blockers/beta-cyclodextrins inclusion complex by NMR, DSC, X-ray and SEM investigation. J Pharm Biomed Anal. 2000;23:33–40.
Silva ACM, Gálico DA, Guerra RB, Legendre AO, Rinaldo D, Galhiane MS, Bannach G. Study of some volatile compound evolved from the thermal decomposition os atenolol. J Therm Anal Calorim. 2014;115:2517–20.
Jain A, Yalkowsky SH. Estimation of melting points of organic compounds-II. J Pharm Sci. 2006;95:2562–618.
Nordstrom FL, Rasmuson AC. Prediction of solubility curves and melting properties of organics and pharmaceutical compounds. Eur J Pharm Sci. 2009;36:330–44.
Nicolet EPA Vapor Phase database. Omnic 8.0 software. Thermo Scientific.
HR Nicolet Sampler Library. Omnic 8.0 software. Thermo Scientific.
Castro RAE. β-1 antagonists selective adrenergic: structure of atenolol. Thesis (Ph.D. in Pharmaceutical Chemistry), Faculty of Pharmacy, University of Coimbra, Coimbra, 2006.
Acknowledgements
Authors acknowledge the Brazilian Foundations Conselho Nacional de Desenvolvimento Científico (CNPq) e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Apoio à Pesquisa do estado de São Paulo (FAPESP), and Núcleo de Pesquisa em Ciência e Tecnologia de Biorecursos (CiTecBio/NAP’s-PRP/USP) for Research Grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amorim, P.H.O., Ferreira, A.P.G., Machado, L.C.M. et al. Investigation on the thermal behavior of β-blockers antihypertensives atenolol and nadolol using TG/DTG, DTA, DSC, and TG–FTIR. J Therm Anal Calorim 120, 1035–1042 (2015). https://doi.org/10.1007/s10973-014-4357-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-014-4357-3