Abstract
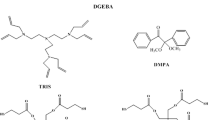
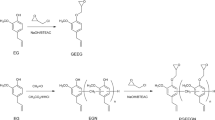
In this study is reported a new method to photocure epoxy resins rapid and quantitatively. It was combined the thiol-ene photopolymerization with the anionic polymerization of an epoxy resin. A tetrallylic tertiary diamine was used as the curing agent. The basic character of the diamine initiated the anionic polymerization of the epoxy resin and at the same time, the allylic groups of the curing agent reacted with a multifunctional thiol. It was found that increasing the concentration of the thiol-ene system in the epoxy formulation resulted in a higher photopolymerization rate and conversion of the epoxy groups. The moduli of the polymers with increasing concentrations of the thiol-ene system were higher than the modulus of a polymer cured with diethylenetriamine which is a conventional curing agent. A study with model compounds revealed that tertiary amine and multifunctional thiol, as well as the polythioethers formed in the thiol-ene reaction induced the anionic polymerization of the epoxy resin.










Similar content being viewed by others
References
Acosta Ortiz R, Garcia Valdéz AE, Berlanga Duarte ML, Guerrero Santos R, Ovando Flores LR, Soucek MD (2008) Development and Study of a Coupling Agent for Photocurable Hybrid Thiol-Ene/cationic Formulations. Macromol Chem Phys 209:2157–2168
Acosta Ortiz R, Puente Urbina BA, Cabello Valdez LV, Berlanga Duarte ML, Guerrero Santos R, García Valdez AE, Soucek MD (2007) The effect of introducing a cationic system into a Thiol-Ene Photopolymerizable Formulation. J Polym Sci A Polym Chem 45:4829–4843
Acosta Ortiz R, Puente Urbina BA, Guerrero Santos R, Berlanga Duarte ML, Garcia Valdez AE, Soucek MD (2008) Development of hybrid polymeric materials based on Photocurable Thiol-Ene/cationic formulations. Macromol Mater Eng 293:731–739
Ashcroft WR (1993) Curing Agents for epoxy resins. Blackie Academic & Professional, Bishonpbriggs
Beyazkilic Z, Kahveci MU, Aydogan B, Kiskan B, Yagci Y (2012) Synthesis of polybenzoxazine precursors, using thiols: simultaneous thiol-ene and ring-opening reactions. J Polym Sci: Part A: Polym Chem 40:4029–4036
Bor-sem C, Khan SA (1997) Real-time FTIR and in situ rheological studies on the UV curing kinetics of thiol-ene polymers Real-time FTIR and in situ rheological studies on the UV curing kinetics of thiol-ene polymers. Macromolecules 30:7322–7328
Carioscia JA, Liu H, Stansbury JW, Bowman CN (2005) Thiol-ene oligomers as dental restorative materials. Dent Mater 21:1137–1143
Carioscia JA, Stansbury JW, Bowman CN (2007) Evaluation and control of thiol-ene/thiol-epoxy hybrid networks. Polymer 48:1526–1532
Cho JD, Hong JW (2004) UV-initiated free radical and cationic photopolymerizations of Acrylate/Epoxide and Acrylate/Vinyl Ether Hybrid systems with and without Photosensitizers. J Appl Polym Sci 93:1473–1483
Cramer NB, Bowman CN (2001) Kinetics of Thiol-ene and Thiol-Acrylate Photopolymerizations with Real-Time Fourier Transform Infrared. J Polym Scie: Part A: Polym Chem 39:3311–3318
Flores M, Tomuta AM, Fernández-Francos X, Ramis X, Sangermano M, Serra A (2013) A new two-stage curing system: Thiol-ene/epoxy homopolymerization using an allyl terminated hyperbranched polyester as reactive modifier. Polymer 54:5473–5481
Henkel, Ag & Co Kgaa 2013 US patent application 20130146223, 13 junio
Hodgkin, J.H.; Demerac, S. (1980) Ions in polymers, Cyclopolymerization of N, N-disubstituted diallyl amines, In Advances in Chemistry, ed. Adi Eisenberg, vol 187, chap14, p.211-233, ACS publications
Hoyle CE, Bowman CN (2010) Thiol-Ene Click Chemistry Angew. Chem, Inter Ed 49(9):1540–1573
Hoyle CE, Lee TY, Roper TT-e (2004) Chemistry of the past with prommise for the future. J Polym Sci: Part A: Polym Chem 42:5301–5338
Jacobine, A.F. (1993); In Radiation curing in polymer scienceand technology, III, polymerization mechanism, Fouassier, J.D. Rabek, J.F. eds. Elsevier Applied Polymer Science, London,, Vol.3
Kim SK, Baguenard C, Guymon CA (2013) Influence of Photopolymerization Characteristics onThermo-Mechanical Properties of Nanocomposites Utilizing Polymerizable Organoclays in Thiol-Acrylate Systems. Macromol Symp 329:173–192
Lee TY, Roper TM, Jonsson ES, Guymon CA, Hoyle CE (2004) Thiol-Ene Photopolymerization of vinyl acrylate/Multifunctional Thiol mixtures. Macromolecules 37:3606–3613
Lee TY, Smith Z, Reddy SK, Cramer NB, Bowman CN (2007) Thiol-Allyl Ether-Methacrylate ternary systems. Polymerization mechanism, Macromolecules 40:1466–1472
Liow SS, Lipik VT, Widjaja LK, Abadie MJM (2012) Synthesis, characterization and photopolymerization of vinyl ether and acrylate functionalized hybrid oligo-caprolactone. J Polym Res 19:9748–9756
Lowe A (2010) Thiol-Ene Click reactions and recent applications in polymer and material synthesis. Polym Chem 1:17–36
Rozenberg BA (1986) Advances in Polymer Sciences 75:113–165
Tanaka, Y.M., (1973) Epoxide-curig reactions, In: Epoxy resins, chemistry and technology, ed. C.T., May, New York, Marcel Dekker
Van Assche G, Swier S, Van Mele B (2002) Modeling and experimental verification of the kinetics of reacting polymer systems. Thermochim Acta 388:327–314
Xiao M, He Y, Nie J (2008) Novel Bisphenol A epoxide-acrylate hybrid oligomer and its photopolymerization. Designed Monomer and Polymers 11(4):383–394
Ying C, Jessop JLP (2006) Decreased Oxygen Inhibition in photopolymerized Acrylate/Epoxide Hybrid polymer coatings as demostrated by Raman Spectroscopy. Polymer 47(19):6560–6566
Acknowledgments
The authors thank the Mexican National Council of Science and Technology (CONACYT) for funding this project 151489. We also gratefully acknowledged the assistance in analysis of samples by Guadalupe Mendez, Jose Luis Saucedo, Sergio Zertuche and Rodrigo Cedillo,
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Acosta Ortiz, R., García Valdez, A.E., Navarro Tovar, A.G. et al. The development of an Epoxy-amine/Thiol-ene photocurable system. J Polym Res 21, 504 (2014). https://doi.org/10.1007/s10965-014-0504-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-014-0504-6