Abstract
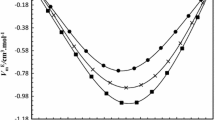
Densities of the ternary system 1,4-dioxane + butyl acrylate + ethyl acrylate and its binary butyl acrylate + ethyl acrylate have been measured in the whole composition range, at 298.15 K and atmospheric pressure, using an Anton Paar DMA 5000 oscillating U-tube densimeter. The calculated excess molar volumes of the binary system are positive and were correlated with the Redlich–Kister equation and with a series of Legendre polynomials. Several models were used to correlate the ternary behavior from the excess molar volume data of their constituent binaries and were found equally good to fit the data. The best fit was based on a direct approach, without information on the component binary systems.
Similar content being viewed by others
References
R. D. Peralta, R. Infante, G. Cortez, R. R. Ramírez, and J. Wisniak, J. Chem. Thermodyn. 35, 239 (2003).
H. C. Van Ness and M. M. Abbott, Classical Thermodynamics of Nonelectrolyte Solutions (McGraw-Hill, New York, 1982).
O. Redlich and A. T. Kister, Ind. Eng. Chem. 40, 345 (1948).
J. Tomiska, CALPHAD 5, 93 (1981).
J. Tomiska, CALPHAD 8, 283 (1984).
J. Wisniak and A. Polishuk, Fluid Phase Equilib. 164, 61 (1999).
I. Cibulka, Collect. Czech. Chem. Commun. 47, 1414 (1982).
P. P. Singh, R. K. Nigam, S. O. Sharma, and S. Aggarwal, Fluid Phase Equilib. 18, 333 (1984).
I. Nagata and K. Tamura, J. Chem. Thermodyn. 22, 279 (1990).
A. Tamir, Chem. Eng. Sci. 36, 1467 (1981).
N. V. Sastry and M. K. Valand, Phys. Chem. Liq. 38, 61 (2000).
I. A. Riddick, W. B. Bunger, and T. K. Sakano, Organic Solvents, 4th Ed., Vol. 2 (Wiley-Interscience, New York, 1986).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wisniak, J., Peralta, R.D., Infante, R. et al. Volumetric Properties of the Ternary System 1,4-Dioxane + Butyl Acrylate + Ethyl Acrylate and Its Binary Butyl Acrylate + Ethyl Acrylate at 298.15 K. J Solution Chem 34, 171–183 (2005). https://doi.org/10.1007/s10953-005-2754-8
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s10953-005-2754-8