Abstract
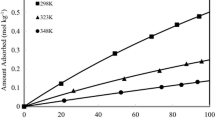
Adsorption of CO2 and CO at 25 °C has been conducted using commercially-available (Y, ZSM-5) and laboratory-synthesized (SSZ-13, SAPO-34) H-zeolites with different framework topologies and chemical compositions, and their textual and surface properties have been characterized by N2 sorption and NH3 adsorption techniques. All the zeolites were microporous, although ZSM-5 and SSZ-13 apparently showed a mesoporous sorption behavior due to the interparticle spaces. The zeolites had Si/Al values in the order of SSZ-13 (16.44) > ZSM-5 (16.08) ≫ Y (2.82) ≫ SAPO-34 (0.19). Regardless, high CO2 adsorption capacity was obtained for SSZ-13 and SAPO-34 with a CHA framework. The FAU zeolite Y with the highest micropore volume showed less CO2 adsorption than the CHA zeolites and the MFI-type ZSM-5 yielded the poorest performance. Probing acid sites in the H-form zeolites using NH3 disclosed that these all contain both weak and strong acid sites with significant dependence of their strengths and amounts on the topology. The acid strength of the weak acid sites in the CHA zeolites was the weakest, which might allow a stronger interaction with CO2. The H-zeolites gave CO2/CO selectivity factors that were in the range of 4.61–11.0, depending on the framework topology.







Similar content being viewed by others
References
S. Sircar, Ind. Eng. Chem. Res. 45, 5435 (2006)
R.T. Yang, Adsorbents: Fundamental and Applications (Wiley, Hoboken, 2003), pp. 8–190
P.K. Prabhakaran, J. Deschamps, J. Porous Mater. 22, 1073 (2015)
J.R. Li, Y. Ma, M.C. McCarthy, J. Sculley, J. Yu, H.K. Jeong, P.B. Balbuena, H.C. Zhou, Coord. Chem. Rev. 255, 1791 (2011)
J.R. Li, R.J. Kuppler, H.C. Zhou, Chem. Soc. Rev. 38, 1477 (2009)
P.L. Llewellyn, S. Bourrelly, C. Serre, A. Vimont, M. Daturi, L. Hamon, G. de Weireld, J.S. Chang, D.Y. Hong, Y.K. Hwang, S.H. Jhung, G. Ferey, Langmuir 24, 7245 (2008)
H. Furukawa, N. Ko, Y.B. Go, N. Aratani, S.B. Choi, E. Choi, A.O. Yazaydin, R.Q. Snurr, M. O’Keeffe, J. Kim, O.M. Yaghi, Science 329, 424 (2010)
D. Yuan, D. Zhao, D. Sun, H.C. Zhou, Angew. Chem. Int. Ed. 49, 5357 (2010)
A.R. Millward, O.M. Yaghi, J. Am. Chem. Soc. 127, 17998 (2005)
M.H. Kim, S.O. Choi, S.T. Choo, Clean Technol. 19, 370 (2013)
M. Palomino, A. Corma, J.L. Jorda, F. Rey, S. Valencia, Chem. Commun. 48, 215 (2012)
K.S. Walton, M.B. Abney, M.D. LeVan, Microporous Mesoporous Mater. 91, 78 (2006)
D. Barthomeuf, Microporous Mesoporous Mater. 66, 1 (2003)
S.K. Wirawan, D. Creaser, Sep. Purif. Technol. 52, 224 (2006)
G. Maurin, Ph Llewellyn, Th Poyet, B. Kuchta, J. Phys. Chem. B 109, 125 (2005)
R.A. Schoonheydt, P. Geerlings, E.A. Pidko, R.A. van Santen, J. Mater. Chem. 22, 18705 (2012)
G. Vitale, L.M. Bull, B.M. Powell, A.K. Cheetham, J. Chem. Soc. Chem. Commun. 2253 (1995)
J.A. Delgado, M.A. Uguina, J.M. Gomez, L. Ortega, Sep. Purif. Technol. 48, 223 (2006)
S.U. Rege, R.T. Yang, AIChE J. 46, 734 (2000)
G.T. Kokotailo, S.L. Lawton, D.H. Olson, W.M. Meier, Nature 272, 437 (1978)
L.J. Smith, A. Davidson, A.K. Cheetham, Catal. Lett. 49, 143 (1997)
I. Daems, R. Singh, G. Baron, J. Denayer, Chem. Commun. 1316 (2007)
M.R. Hudson, W.L. Queen, J.A. Mason, D.W. Fickel, R.F. Lobo, C.M. Brown, J. Am. Chem. Soc. 134, 1970 (2012)
M. Itakura, I. Goto, A. Takahashi, T. Fujitani, Y. Ide, M. Sadakane, T. Sano, Microporous Mesoporous Mater. 144, 91 (2011)
A.M. Prakash, S. Unnikrishnan, J. Chem. Soc. Faraday Trans. 90, 2291 (1994)
A. Saito, H.C. Foley, AIChE J. 37, 429 (1991)
W.H. Yang, M.H. Kim, S.W. Ham, Catal. Today 123, 94 (2007)
A.S. Kovo, O. Hernandez, S.M. Holmes, J. Mater. Chem. 19, 6207 (2009)
Q. Zhu, J.N. Kondo, T. Tatsumi, S. Inagaki, R. Ohnuma, Y. Kubota, Y. Shimodaira, H. Kobayashi, K. Domen, J. Phys. Chem. 111, 5409 (2007)
G. Liu, P. Tian, J. Li, D. Zhang, F. Zhou, Z. Liu, Microporous Mesoporous Mater. 111, 143 (2008)
N. Salman, C.H. Ruscher, J.C. Buhl, W. Lutz, H. Toufar, M. Stocker, Microporous Mesoporous Mater. 90, 339 (2006)
M. Krol, W. Mozgawa, W. Jastrzebski, K. Barczyk, Microporous Mesoporous Mater. 156, 181 (2012)
P.A. Jacobs, H.K. Beyer, J. Valyon, Zeolites 1, 161 (1981)
K.S. Triantafyllidis, L. Nalbandian, P.N. Trikalitis, A.K. Ladavos, T. Mavromoustakos, C.P. Nicolaides, Microporous Mesoporous Mater. 75, 89 (2004)
F. Pechar, D. Rykl, Zeolites 3, 333 (1983)
A.J.M. de Man, R.A. van Santen, Zeolites 12, 269 (1992)
F.D.P. Mees, L.R.M. Martens, M.J.G. Janssen, A.A. Verberckmoes, E.F. Vansant, Chem. Commun. 44 (2003)
K.S.W. Sing, D.H. Everett, R.A.W. Haul, L. Moscou, R.A. Pierotti, J. Rouquerol, T. Siemieniewska, Pure Appl. Chem. 57, 603 (1985)
A.J.J. Koekkoek, H. Xin, Q. Yang, C. Li, E.J.M. Hensen, Microporous Mesoporous Mater. 145, 172 (2011)
J.W. Park, J.Y. Lee, K.S. Kim, S.B. Hong, G. Seo, Appl. Catal. A 339, 36 (2008)
D.M. D’Alessandro, B. Smit, J.R. Long, Angew. Chem. Int. Ed. 49, 6058 (2010)
T.D. Pham, M.R. Hudson, C.M. Brown, R.F. Lobo, ChemSusChem 7, 3031 (2014)
R.F. Lobo, AIChE J. 54, 1402 (2008)
S. Choi, J.H. Drese, C.W. Jones, ChemSusChem 2, 796 (2009)
J. McEwen, J.D. Hayman, A.O. Yazaydin, Chem. Phys. 412, 72 (2013)
R.V. Siriwardane, M.S. Shen, E.P. Fisher, J.A. Poston, Energy Fuels 15, 279 (2001)
T.D. Pham, Q. Liu, R.F. Lobo, Langmuir 29, 832 (2013)
N. Katada, H. Igi, J.H. Kim, M. Niwa, J. Phys. Chem. B 101, 5969 (1997)
H.Y. Jeon, C.H. Shin, H.J. Jung, S.B. Hong, Appl. Catal. A 305, 70 (2006)
B.M. Lok, C.A. Messina, R.L. Patton, R.T. Gajek, T.R. Cannan, E.M. Flanigen, J. Am. Chem. Soc. 106, 6092 (1984)
N. Katada, K. Nouno, J.K. Lee, J. Shin, S.B. Hong, M. Niwa, J. Phys. Chem. C 115, 22505 (2011)
G.I. Kapustin, T.R. Brueva, A.L. Klyachko, S. Beran, B. Wichterlova, Appl. Catal. 42, 239 (1988)
L. Smith, A.K. Cheetham, L. Marchese, J.M. Thomas, P.A. Wright, J. Chen, Catal. Lett. 41, 13 (1996)
A. Zukal, J. Jagiello, J. Mayerova, J. Cejka, Phys. Chem. Chem. Phys. 13, 15468 (2011)
Acknowledgments
A partial grant-in-aid for this study was provided by the New & Renewable Energy of the Korea Institute of Energy Technology Evaluation and Planning (KETEP) via Grant # 20123010100010.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, M.H., Cho, I.H., Park, J.H. et al. Adsorption of CO2 and CO on H-zeolites with different framework topologies and chemical compositions and a correlation to probing protonic sites using NH3 adsorption. J Porous Mater 23, 291–299 (2016). https://doi.org/10.1007/s10934-015-0081-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-015-0081-z