Abstract
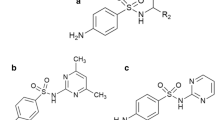
Combating Type-2 diabetes mellitus is a pivotal challenge in front of the present world. Several lines of therapy are in practice for resisting this deadly disease which often culminates with cardiovascular complexities, neuropathy and retinopathy. Among various therapies, administration of alpha glucosidase inhibitors is common and widely practiced. Sulfonylurea category of anti diabetic drug often suffers from cross reactivity with sulfamethoxazole (SMX), a common drug in use to treat a handful of microbial infections. However the specific cellular target generating postprandial hypoglycemia on SMX administration is till date unraveled. The present work has been initiated to elucidate the effects of a group of sulfonamide drugs inclusive of SMX for their amylase inhibitory role. SMX inhibits porcine pancreatic amylase (PPA) in a noncompetitive mode with an average IC50 value 0.94 mM respectively. Interaction of SMX with PPA is manifested with gradual quenching of tryptophan fluorescence with concomitant shift in lambda max value (λmax). Binding is governed by entropy driven factor (24.8 cal mol−1 K−1) with unfavorable contribution from enthalpy change. SMX interferes with the activity of acarbose in a synergistic mode to reduce the effective dose of acarbose as evident from the in vitro PPA inhibition study. In summary, loss of PPA activity in presence of SMX is indicative of structural changes of PPA which is further augmented in the presence of acarbose as explained in the schematic model and docking study.







Similar content being viewed by others
Abbreviations
- PPA:
-
Porcine pancreatic amylase
- SMX:
-
Sulfamethoxazole
- ANS:
-
8-Anilino-1-naphthalenesulfonate
- ITC:
-
Isothermal titration calorimetry
References
World health organization consultation (1999) Definition, diagnoses and classification of diabetes mellitus and its complication. Part 1: diagnosis and classification of diabetes mellitus. Report of a WHO consultation Geneva
Amarican Diabetes Association (2008) Diagnosis and classification of diabetes mellitus. Diabetes Care 31:S55–S60
Kim YM, Jeong YK, Wang MH, Lee WY, Rhee HI (2005) Inhibitory effect of pine extract on alpha-glucosidase activity and postprandial hyperglycemia. Nutrition 21:756–761
De Fronzo R (1999) Pharmacologic therapy for type 2 diabetes mellitus. Ann Intern Med 131:281–303
Aguiar LGK, Villela NR, Bouskela EA (2007) Microcirculação no Diabetes: Implicações nas Complicações Crônicas e Tratamento da Doença. Arq Bras Endocrinol Metab 51:204–211
Cooper ME, Bonnet F, Oldfield M, Jandeleit-Dahm K (2001) Mechanism of diabetic vasculopathy: an overview. Am J Hypertens 14:475–486
Cooper ME, Gilbert RE, Epstein M (1998) Pathophysiology of diabetic nephropathy. Metabolism 47:3–6
Lebowitz HE, Feinglos MN (1978) Sulfonylurea drugs: mechanism of anti-diabetic action and therapeutic usefulness. Diabetes Care 1:189–198
Christensen LK, Mausen JM, Kristensen M (1963) Sulfaphenazole- induced hypoglycemic attacks in tolbutamide-treated diabetics. Lancet 2:1298–1301
Loulatieres A (1957) The hypoglycemic sulfonamides: history and development of the problem from 1942 to 1955. Ann NY Acad Sci 71:4–11
Aren R, Garber AJ, Field JB (1983) Sulfonamide-induced hypoglycemia in chronic renal failure. Arch Intern Med 143:827–829
Mihic M, Mantner LS, Feness JZ, Grant K (1975) Effect of trimethoprim-sulfamethoxazole on blood insulin and glucose concentrations of diabetics. Can Med Assoc J 112:805–825
Goke B, Herrmann-Rinke C (1998) The evolving role of alpha-glucosidase inhibitors. Diabetes Metab Res 14:S31–S38
Lebowitz HE (1998) Alpha-glucosidase inhibitors as agents in the treatment of diabetes. Diabetes Rev 6:132–145
Yoon SH, Robyt JF (2003) Study of the inhibition of four alpha amylases by acarbose and its 4IV-α-maltohexaosyl and 4IV-α-maltododecaosyl analogues. Carbohydr Res 338:1969–1980
Laar FA, Lucassen PLBJ, Akkermans RP, Lisdonk EH, Rutten GEHM, Weel C (2008) Alpha-glucosidase inhibitors for type 2 diabetes mellitus. (Cochrane review). Cochrane Library. John Wiley and Sons, Ltd, p. 182
Pyne S, Gayathri P (2005) Geometric methods in molecular docking. Bioinform India J 3:11–12
Miller GL (1959) Use of dinitrosalisylic acid reagent for determination of reducing sugar. Anal Chem 31:426–429
Burlingham BT, Widlanski TS (2003) An intuitive look at the relationship of Ki and IC50: a more general use for the Dixon plot. J Chem Educ 80:214–218
Zhang XQ, Yang MY, Ma Y, Song JR (2007) Study on biological characteristics and inhibition kinetics of α-amylase inhibitor from white kidney beans. Food Sci 28:29–31
Chakrabarti R, Singh B, Prakrith VN, Vanchhawng L, Thirumurugan K (2014) Screening of nine herbal plants for in vitro a-amylase inhibition. Asian J Pharm Clin Res 7(4):84–89
Lakowitz JR (1983) Principles of fluorescence spectroscopy. Plenum Press, New York, pp 1–44
Dundas J, Ouyang Z, Tseng J, Binkowski A, Turpaz Y, Liang J (2006) CASTp: computed atlas of surface topography of proteins with structural and topographical mapping of functionally annotated resiudes. Nucleic Acid Res 34:W116–W118
Friesner RA, Banks JL, Murphy RB, Halgren TA, Klicic JJ, Mainz DT, Repasky MP, Knoll EH, Shelley M, Perry JK, Shaw DE, Francis P, Shenkin PS (2004) Glide: a new approach for rapid, accurate docking and scoring 1 method and assessment of docking accuracy. J Med Chem 47:1739–1749
Gyémánt G, Kandra L, Nagy V, Somsák L (2003) Inhibition of human salivary α-amylase by glucopyranosylidene-spiro-thiohydantoin. Biochem Biophys Res Commun 312:334–339
Brayer GD, Sidhu G, Maurus R, Rydberg EH, Braun C, Wang Y, Nguyen NT, Overall CM, Withers SG (2000) Subsite mapping of the human pancreatic alpha-amylase active site through structural, kinetic and mutagenesis techniques. Biochem US 39:4778–4791
Prasad AR, Luduena RF, Horowitz PM (1986) Bis (8-anilinonaphthalene-1-sulfonate) as a probe for tubulin decay. Biochemistry 25:739–742
Huang SY, Zho XQ (2010) Advances and challenges in protein-ligand docking. Int J Mol Sci 11:3016–3034
Akkarachiyasit S, Yibchok-Anun S, Wacharasindhu S, Adisakwattana S (2011) In vitro inhibitory effects of cyandin-3-rutinoside on pancreatic α-amylase and its combined effect with acarbose. Molecules 16:2075–2083
Mandel GL, Petri WA (1996) Antimicrobial agents (continued): sulfonamide, trimethoprim-sulfamethoxazole, quinolones and agents for urinary tract infections. In: Hardman JG, Limbird LE (eds) Goodman and Gilman’s the pharmacological basis of therapeutics, 9th edn. McGrow-Hill, New York, pp 1057–1072
Kwon YI, Apostolidis E, Shetty K (2008) In vitro studies of eggplant (Solanum melongena) phrnolics as inhibitors of key enzymes relevant for type 2 diabetes and hypertension. Bioresour Technol 99:2981–2988
Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M (2002) Acarbose for prevention of type 2 diabetes mellitus: the STOP-NIDDM randomized trial. Lancet 359:2072–2077
Acknowledgement
The authors are thankful to the Department of Biotechnology, Haldia Institute of Technology for providing space and infrastructural support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maity, S., Mukherjee, K., Banerjee, A. et al. Inhibition of Porcine Pancreatic Amylase Activity by Sulfamethoxazole: Structural and Functional Aspect. Protein J 35, 237–246 (2016). https://doi.org/10.1007/s10930-016-9668-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-016-9668-8