Abstract
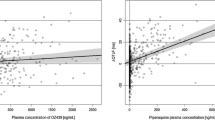
Drug-induced cardiotoxicity, including tachycardia and QT prolongation, remains a major safety concern that needs to be identified and its risk mitigated in early stages of drug development. In the present study, an integrated toxicokinetic–toxicodynamic (TK–TD) modeling approach within a nonlinear mixed-effect modeling framework is applied to investigate concurrent abnormal heart rate and QT changes in three beagle dogs, using a Novartis internal compound (NVS001) as the case example. By accounting for saturable drug absorption, circadian rhythms, drug-effect tolerance, and nonlinear rate-dependency of QT interval, the dynamic TK–TD model captures the experimentally observed drug effects on heart rate and QT interval across a wide dosing range of NVS001 in beagle dogs. Further analyses reveal that the NVS001-induced QT prolongation observed in the low-dose groups is potentially caused by direct drug inhibition on the hERG channel, while the apparent QT shortening in the high-dose groups may be due to strong rate-dependency of QT at high heart rates. This study also suggests that the TK–TD model can be used to identify direct drug effects on the non-rate-dependent QT component by dissociating QT changes from tachycardia and deriving a new QT correction method. The integrated TK–TD model presented here may serve as a novel quantitative framework for evaluating drug-induced concurrent changes in heart rate and QT to potentially facilitate preclinical and clinical safety studies.










Similar content being viewed by others
References
Akaike H (1981) Citation classic—a new look at the statistical-model identification. Cc/Eng Tech Appl Sci 51:22
Bonate PL (2006) Pharmacokinetic–pharmacodynamic modeling and simulation. Springer, New York
Braunwald E (1982) Mechanism of action of calcium-channel-blocking agents. New Engl J Med 307:1618–1627
Brynne L, Paalzow LK, Karlsson MO (1999) Mechanism-based modeling of rebound tachycardia after chronic I-propranolol infusion in spontaneous hypertensive rats. J Pharmacol Exp Ther 290:664–671
Burn JH, Rand MJ (1958) Action of nicotine on the heart. Br Med J 1:137–139
Cavero I (2007) Using pharmacokinetic/pharmacodynamic modelling in safety pharmacology to better define safety margins: a regional workshop of the Safety Pharmacology Society. Expert Opin Drug Saf 6:465–471
Chan A, Isbister GK, Kirkpatrick CM et al (2007) Drug-induced QT prolongation and torsades de pointes: evaluation of a QT nomogram. QJM 100:609–615
Chow MJ, Ambre JJ, Ruo TI et al (1985) Kinetics of cocaine distribution, elimination, and chronotropic effects. Clin Pharmacol Ther 38:318–324
Cryer PE (1980) Physiology and pathophysiology of the human sympathoadrenal neuroendocrine system. N Engl J Med 303:436–444
Darpo B (2010) The thorough QT/QTc study 4 years after the implementation of the ICH E14 guidance. Br J Pharmacol 159:49–57
De Daly MB, Scott MJ (1958) The effects of stimulation of the carotid body chemoreceptors on heart rate in the dog. J Physiol 144:148–166
Degaute JP, Van De Borne P, Linkowski P et al (1991) Quantitative analysis of the 24-hour blood pressure and heart rate patterns in young men. Hypertension 18:199–210
Elkayam U, Kulick D, Mcintosh N et al (1987) Incidence of early tolerance to hemodynamic-effects of continuous infusion of nitroglycerin in patients with coronary-artery disease and heart-failure. Circulation 76:577–584
Fijorek K, Patel N, Klima L et al (2013) Age and gender dependent heart rate circadian model development and performance verification on the proarrhythmic drug case study. Theor Biol Med Model 10:1–11
Fischman MW, Schuster CR, Javaid J et al (1985) Acute tolerance development to the cardiovascular and subjective effects of cocaine. J Pharmacol Exp Ther 235:677–682
Gabrielsson J, Weiner D (2006) Pharmacokinetic & pharmacodynamic data analysis: concepts and applications. Swedish Pharmaceutical Press, Stockholm
Guyton AC, Hall JE (2000) Textbook of medical physiology. W.B. Saunders Company, Philadelphia
Holzgrefe H, Ferber G, Champeroux P et al (2014) Preclinical QT safety assessment: cross-species comparisons and human translation from an industry consortium. J Pharmacol Toxicol Methods 69:61–101
ICh (2005) Guidance for industry: E14 clinical evaluation of QT/QTc interval prolongation and proarrhythmic potential for non-antiarrhythmic Drugs. FDA, Maryland
ICh (2001) Guidance for industry: S7A safety pharmacology studies for human pharmaceuticals. FDA, Maryland
ICh (2005) Guidance for industry: S7B nonclinical evaluation of the potential for delayed ventricular repolarization (QT interval prolongation) by human pharmaceuticals. FDA, Maryland
Kleinbloesem CH, Vanbrummelen P, Danhof M et al (1987) Rate of increase in the plasma-concentration of nifedipine as a major determinant of its hemodynamic-effects in humans. Clin Pharmacol Ther 41:26–30
Koroboki E, Manios E, Psaltopoulou T et al (2012) Circadian variation of blood pressure and heart rate in normotensives, white-coat, masked, treated and untreated hypertensives. Hellenic J Cardiol 53:432–438
Li L, Desai M, Desta Z et al (2004) QT analysis: a complex answer to a ‘simple’ problem. Stat Med 23:2625–2643
Mager DE, Wyska E, Jusko WJ (2003) Diversity of mechanism-based pharmacodynamic models. Drug Metab Dispos 31:510–518
Malfatto G, Facchini M, Zaza A (2003) Characterization of the non-linear rate-dependency of QT interval in humans. Europace 5:163–170
Malfatto G, Zaza A, Facchini M (2007) Different effects of antiarrhythmic drugs on the rate-dependency of QT interval: a study with amiodarone and flecainide. J Cardiovasc Pharmacol 50:535–540
Miyazaki H, Yoshida M, Samura K et al (2002) Ranges of diurnal variation and the pattern of body temperature, blood pressure and heart rate in laboratory beagle dogs. Exp Anim Tokyo 51:95–98
Moltzen EK, Bang-Andersen B (2006) Serotonin reuptake inhibitors: the corner stone in treatment of depression for half a century—a medicinal chemistry survey. Curr Top Med Chem 6:1801–1823
Mould DR, Upton RN (2013) Basic concepts in population modeling, simulation, and model-based drug development-part 2: introduction to pharmacokinetic modeling methods. CPT Pharmacomet Syst Pharmacol 2:e38
Packer DL, Bardy GH, Worley SJ et al (1986) Tachycardia-induced cardiomyopathy: a reversible form of left ventricular dysfunction. Am J Cardiol 57:563–570
Piotrovsky V (2005) Pharmacokinetic-pharmacodynamic modeling in the data analysis and interpretation of drug-induced QT/QTc prolongation. AAPS J 7:E609–E624
Piotrovsky V (2005) Pharmacokinetic–pharmacodynamic modeling in the data analysis and interpretation of drug-induced QT/QTc prolongation.pdf. AAPS J 7:E611–E624
Polak S, Pugsley MK, Stockbridge N et al (2015) Early drug discovery prediction of proarrhythmia potential and its covariates. AAPS J 17:1025–1032
Porchet HC, Benowitz NL, Sheiner LB (1988) Pharmacodynamic model of tolerance—application to nicotine. J Pharmacol Exp Ther 244:231–236
Porchet HC, Benowitz NL, Sheiner LB (1988) Pharmacodynamic model of tolerance: application to nicotine. J Pharmacol Exp Ther 244:231–236
Quan KJ, Carlson MD, Thames MD (1997) Mechanisms of heart rate and arterial blood pressure control: implications for the pathophysiology of neurocardiogenic syncope. Pace 20:764–774
Redfern WS, Carlsson L, Davis AS et al (2003) Relationships between preclinical cardiac electrophysiology, clinical QT interval prolongation and torsade de pointes for a broad range of drugs: evidence for a provisional safety margin in drug development. Cardiovasc Res 58:32–45
Robertson D, Wade D, Workman R et al (1981) Tolerance to the humoral and hemodynamic-effects of caffeine in man. J Clin Investig 67:1111–1117
Sager PT, Gintant G, Turner JR et al (2014) Rechanneling the cardiac proarrhythmia safety paradigm: a meeting report from the Cardiac Safety Research Consortium. Am Heart J 167:292–300
Spence S, Soper K, Hoe CM et al (1998) The heart rate-corrected QT interval of conscious beagle dogs: a formula based on analysis of covariance. Toxicol Sci 45:247–258
Stauss HM (2003) Heart rate variability. Am J Physiol 285:R927–R931
Sugimoto T, Sagawa K, Guyton AC (1966) Effect of tachycardia on cardiac output during normal and increased venous return. Am J Physiol 211:288–292
Tsume Y, Mudie DM, Langguth P et al (2014) The biopharmaceutics classification system: subclasses for in vivo predictive dissolution (IPD) methodology and IVIVC. Eur J Pharm Sci 57:152–163
Upton RN, Mould DR (2014) Basic concepts in population modeling, simulation, and model-based drug development: part 3-introduction to pharmacodynamic modeling methods. CPT Pharmacomet Syst Pharmacol 3:e88
Van Steeg TJ, Freijer J, Danhof M et al (2007) Pharmacokinetic-pharmacodynamic modelling of S(−)-atenolol in rats: reduction of isoprenalineinduced tachycardia as a continuous pharmacodynamic endpoint. Br J Pharmacol 151:356–366
Vinik HR, Kissin I (1998) Rapid development of tolerance to analgesia during remifentanil infusion in humans. Anesth Analg 86:1307–1311
Acknowledgements
The authors are grateful to generous support from Kaci Phizackerley in formulation preparation and from David Tully in project management.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors were full-time employees of Novartis and potentially owned stock in Novartis when work presented in this manuscript was conducted.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, F., Heimbach, T., Hatsis, P. et al. Integrated TK–TD modeling for drug-induced concurrent tachycardia and QT changes in beagle dogs. J Pharmacokinet Pharmacodyn 44, 449–462 (2017). https://doi.org/10.1007/s10928-017-9532-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-017-9532-2