Abstract
Purpose
Autosomal recessive dedicator of cytokinesis 8 (DOCK8−/−) and autosomal dominant signal transducer and activator of transcription 3 (STAT3−/+) deficiencies are inborn errors of immunity (IEI) disorders present with the classic features of eczema and create a dilemma during differentiation from atopic dermatitis (AD). Therefore, an appropriate approach is required for eczema to diagnose DOCK8−/− and STAT3−/+ early. Here, we described a set of clinical and immunological variables, including atypical AD localizations and lymphocyte subsets, to differentiate DOCK8−/− or STAT3−/+ from AD.
Methods
This multicenter study involved 100 patients with DOCK8−/− and STAT3−/+ and moderate/severe AD. We recruited disease manifestations, including detailed localizations of eczema, infections, and allergy. Principle component analysis (PCA) was used to discriminate DOCK8−/− or STAT3−/+ from AD.
Results
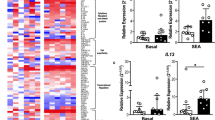
There were 43 patients with DOCK8−/−, 23 with STAT3−/+, and 34 with AD. Pneumonia, severe infections, mucocutaneous candidiasis, and skin abscesses were commonly observed in DOCK8 and STAT3 deficiencies. Atypical skin involvement with neonatal rash, retro auricular, axillary, sacral, and genital eczema discriminate DOCK8−/− and STAT3−/+ from AD with high specificity ranges between 73.5 and 94.1% and positive predictive index ranges between 55 and 93.1%. Together with using absolute numbers of CD3+, CD4+, and CD8+ T cells, the combined clinical and laboratory features showed perfect differentiation between DOCK8−/− or STAT3−/+ and AD via PCA.
Conclusions
The described features can be easily implemented by physicians providing early diagnosis of DOCK8 and STAT3 deficiencies.




Similar content being viewed by others
Data Availability
The data generated during the study are included in this published article and its supplementary file.
References
Woellner C, Gertz EM, Schaffer AA, Lagos M, Perro M, Glocker EO, et al. Mutations in STAT3 and diagnostic guidelines for hyper-IgE syndrome. J Allergy Clin Immunol. 2010;125(2):424-32 e8.
Holland SM, DeLeo FR, Elloumi HZ, Hsu AP, Uzel G, Brodsky N, et al. STAT3 mutations in the hyper-IgE syndrome. N Engl J Med. 2007;357(16):1608–19.
Minegishi Y, Saito M, Tsuchiya S, Tsuge I, Takada H, Hara T, et al. Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature. 2007;448(7157):1058–62.
Renner ED, Puck JM, Holland SM, Schmitt M, Weiss M, Frosch M, et al. Autosomal recessive hyperimmunoglobulin E syndrome: a distinct disease entity. J Pediatr. 2004;144(1):93–9.
Tangye SG, Al-Herz W, Bousfiha A, Cunningham-Rundles C, Franco JL, Holland SM, et al. Human Inborn Errors of Immunity: 2022 Update on the Classification from the International Union of Immunological Societies Expert Committee. J Clin Immunol. 2022;42(7):1473–507.
Engelhardt KR, McGhee S, Winkler S, Sassi A, Woellner C, Lopez-Herrera G, et al. Large deletions and point mutations involving the dedicator of cytokinesis 8 (DOCK8) in the autosomal-recessive form of hyper-IgE syndrome. J Allergy Clin Immunol. 2009;124(6):1289-302 e4.
Buckley RH, Wray BB, Belmaker EZ. Extreme hyperimmunoglobulinemia E and undue susceptibility to infection. Pediatrics. 1972;49(1):59–70.
Zhang Q, Davis JC, Lamborn IT, Freeman AF, Jing H, Favreau AJ, et al. Combined immunodeficiency associated with DOCK8 mutations. N Engl J Med. 2009;361(21):2046–55.
Aydin SE, Kilic SS, Aytekin C, Kumar A, Porras O, Kainulainen L, et al. DOCK8 deficiency: clinical and immunological phenotype and treatment options - a review of 136 patients. J Clin Immunol. 2015;35(2):189–98.
Shah NN, Freeman AF, Su H, Cole K, Parta M, Moutsopoulos NM, et al. Haploidentical Related Donor Hematopoietic Stem Cell Transplantation for Dedicator-of-Cytokinesis 8 Deficiency Using Post-Transplantation Cyclophosphamide. Biol Blood Marrow Transplant. 2017;23(6):980–90.
Gernez Y, Freeman AF, Holland SM, Garabedian E, Patel NC, Puck JM, et al. Autosomal Dominant Hyper-IgE Syndrome in the USIDNET Registry. J Allergy Clin Immunol Pract. 2018;6(3):996–1001.
Grimbacher B, Holland SM, Gallin JI, Greenberg F, Hill SC, Malech HL, et al. Hyper-IgE syndrome with recurrent infections–an autosomal dominant multisystem disorder. N Engl J Med. 1999;340(9):692–702.
Engelhardt KR, Gertz ME, Keles S, Schaffer AA, Sigmund EC, Glocker C, et al. The extended clinical phenotype of 64 patients with dedicator of cytokinesis 8 deficiency. J Allergy Clin Immunol. 2015;136(2):402–12.
Lehman H, Gordon C. The Skin as a Window into Primary Immune Deficiency Diseases: Atopic Dermatitis and Chronic Mucocutaneous Candidiasis. J Allergy Clin Immunol Pract. 2019;7(3):788–98.
Eberting CL, Davis J, Puck JM, Holland SM, Turner ML. Dermatitis and the newborn rash of hyper-IgE syndrome. Arch Dermatol. 2004;140(9):1119–25.
Nihal A, Comstock JR, Holland KE, Singh AM, Seroogy CM, Arkin LM. Clearance of atypical cutaneous manifestations of hyper-IgE syndrome with dupilumab. Pediatr Dermatol. 2022;39(6):940–2.
Chu EY, Freeman AF, Jing H, Cowen EW, Davis J, Su HC, et al. Cutaneous manifestations of DOCK8 deficiency syndrome. Arch Dermatol. 2012;148(1):79–84.
Olaiwan A, Chandesris MO, Fraitag S, Lortholary O, Hermine O, Fischer A, et al. Cutaneous findings in sporadic and familial autosomal dominant hyper-IgE syndrome: a retrospective, single-center study of 21 patients diagnosed using molecular analysis. J Am Acad Dermatol. 2011;65(6):1167–72.
de Wit J, Brada RJK, van Veldhuizen J, Dalm V, Pasmans S. Skin disorders are prominent features in primary immunodeficiency diseases: A systematic overview of current data. Allergy. 2019;74(3):464–82.
Stadler PC, Renner ED, Milner J, Wollenberg A. Inborn Error of Immunity or Atopic Dermatitis: When to be Concerned and How to Investigate. J Allergy Clin Immunol Pract. 2021;9(4):1501–7.
Kasap N, Celik V, Isik S, Cennetoglu P, Kiykim A, Eltan SB, et al. A set of clinical and laboratory markers differentiates hyper-IgE syndrome from severe atopic dermatitis. Clin Immunol. 2021;223:108645.
Eken A, Cansever M, Okus FZ, Erdem S, Nain E, Azizoglu ZB, et al. ILC3 deficiency and generalized ILC abnormalities in DOCK8-deficient patients. Allergy. 2020;75(4):921–32.
Ma CS, Chew GY, Simpson N, Priyadarshi A, Wong M, Grimbacher B, et al. Deficiency of Th17 cells in hyper IgE syndrome due to mutations in STAT3. J Exp Med. 2008;205(7):1551–7.
Kunz B, Oranje AP, Labreze L, Stalder JF, Ring J, Taieb A. Clinical validation and guidelines for the SCORAD index: consensus report of the European Task Force on Atopic Dermatitis. Dermatology. 1997;195(1):10–9.
Kiykim A, Ogulur I, Dursun E, Charbonnier LM, Nain E, Cekic S, et al. Abatacept as a Long-Term Targeted Therapy for LRBA Deficiency. J Allergy Clin Immunol Pract. 2019;7(8):2790-800 e15.
Catak MC, Akcam B, BilgicEltan S, Babayeva R, Karakus IS, Akgun G, et al. Comparing the levels of CTLA-4-dependent biological defects in patients with LRBA deficiency and CTLA-4 insufficiency. Allergy. 2022;77(10):3108–23.
Sefer AP, Abolhassani H, Ober F, Kayaoglu B, BilgicEltan S, Kara A, et al. Expanding the Clinical and Immunological Phenotypes and Natural History of MALT1 Deficiency. J Clin Immunol. 2022;42(3):634–52.
Baris S, Benamar M, Chen Q, Catak MC, Martinez-Blanco M, Wang M, et al. Severe allergic dysregulation due to a gain of function mutation in the transcription factor STAT6. J Allergy Clin Immunol. 2023.
Kennedy K, Heimall J, Spergel JM. Advances in atopic dermatitis in 2017. J Allergy Clin Immunol. 2018;142(6):1740–7.
Schimke LF, Sawalle-Belohradsky J, Roesler J, Wollenberg A, Rack A, Borte M, et al. Diagnostic approach to the hyper-IgE syndromes: immunologic and clinical key findings to differentiate hyper-IgE syndromes from atopic dermatitis. J Allergy Clin Immunol. 2010;126(3):611-7 e1.
Wu J, Chen J, Tian ZQ, Zhang H, Gong RL, Chen TX, et al. Clinical Manifestations and Genetic Analysis of 17 Patients with Autosomal Dominant Hyper-IgE Syndrome in Mainland China: New Reports and a Literature Review. J Clin Immunol. 2017;37(2):166–79.
Biggs CM, Keles S, Chatila TA. DOCK8 deficiency: Insights into pathophysiology, clinical features and management. Clin Immunol. 2017;181:75–82.
Sanal O, Jing H, Ozgur T, Ayvaz D, Strauss-Albee DM, Ersoy-Evans S, et al. Additional diverse findings expand the clinical presentation of DOCK8 deficiency. J Clin Immunol. 2012;32(4):698–708.
Funding
This work was supported by the Scientific and Technological Research Council of Turkey to S.B. (318S202).
Author information
Authors and Affiliations
Contributions
N.K. and S.B. conceptualized the study. N.K., A.K., and S.B. wrote the manuscript. A.K., V.C., K.S.A., and S.B. performed the statistical analysis. N.K., V.C., S.B.E., I.A.H., H.K., A.A., E.A., N.Y., S.N.G., I.R., S.K., S.C., S.S.K., N.E.K., N.G., F.G., A.O., A.D.Y., E.K.A., and S.B. cared for patients and provided samples intellectually contributed to the manuscript and discussions. All authors read the manuscript and contributed to the revision and discussions.
Corresponding author
Ethics declarations
Ethical Approval
The Ethics Committee of Marmara University School of Medicine approved the study.
Consent to Participate
Informed consent for participation was obtained from the family.
Consent for Publication
Informed publication consent was obtained from the family.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kasap, N., Kara, A., Celik, V. et al. Atypical Localization of Eczema Discriminates DOCK8 or STAT3 Deficiencies from Atopic Dermatitis. J Clin Immunol 43, 1882–1890 (2023). https://doi.org/10.1007/s10875-023-01554-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-023-01554-z