Abstract
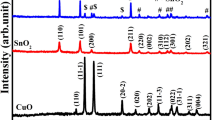
This paper reports the preparation of Pt–SnO2 composite thin film gas sensors for the detection of H2 gas having lower concentration (150 ppm) at relatively lower operating temperature of 85 °C. Nano crystalline tin oxide (SnO2) thin films were prepared by simple spin coating technique. Subsequently platinum thin film was sputtered over the SnO2 thin film to get the Pt–SnO2 composite thin films. The structural, nano structural and gas sensing properties of both SnO2 and Pt–SnO2 thin film sensor were studied. XRD indicate the formation of tetragonal SnO2 where as in case of Pt–SnO2 the existence of cubic SnPt3 was observed. A comparative study of the hydrogen sensing properties viz. operating temperature, response and recovery time and selectivity was carried out. Due to Pt loading, the sensor showed reduction in the operating temperature to 85 °C with good modulation and response time of 0.5 s to H2 gas. The response to other gases such as carbon monoxide (CO) and LPG (at 150 ppm) of the Pt–SnO2 composite thin film sensor were also investigated. The sensor showed highest response to H2 gas where as lowest response was noted for CO gas.











Similar content being viewed by others
References
T. Hubert, L. Boon Brett, G. Black, U. Banach, Hydrogen sensors—a review. Sens. Actuators B: Chem, 157, 329 (2011)
B.H. Chu, J. Nicolosi, C.F. Lo, W. Strupinski, S.J. Pearton, F. Ren, Effect of coated platinum thickness on hydrogen detection sensitivity of graphene-based sensors. Electrochem. Solid.-State Lett. 14, K43 (2011)
L.C. Tien, P.W. Sadik, D.P. Norton, L.F. Voss, S.J. Pearton, H.T. Wang, B.S. Kang, F. Ren, J. Jun, J. Lin, Hydrogen sensing at room temperature with Pt-coated ZnO thin films and nanorods. Appl. Phys. Lett. 87, 222106 (2005)
I. Sayago, E. Terrado, E. Lafuente, M.C. Horillo, W.K. Maser, A.M. Benito, R. Navarro, E.P. Urriolabeita, M.T. Martinez, J. Gutierrez, Hydrogen sensors based on carbon nanotubes thin films. Synth. Met. 148, 15 (2005)
C. Agashe, R.C. Aiyer, High-yield synthesis of nano crystalline tin dioxide by thermal decomposition for use in gas sensors. Int. J. Appl. Ceram. Technol. 5, 181 (2008)
S.F. Bamsaoud, S.B. Rane, R.C. Aiyer, SnO2 film with bimodal distribution of nano-particles for low concentration hydrogen sensor: effect of firing temperature on sensing properties. Mater. Chem. Phys. 133, 681 (2012)
S.F. Bamsaoud, S.B. Rane, R.N. Karekar, R.C. Aiyer, Nano particulate SnO2 based resistive films as a hydrogen and acetone vapour sensor. Sens. Actuators, B 153, 382 (2011)
S.K. Arya, K. Subramanian, H. Silva, S. Jean, S. Bhansali, Advances in materials for room temperature hydrogen sensors. Analyst 137, 2743 (2012)
S. Trocino, A. Donato, M. Latino, N. Donato, S. Gianluca Leonardi, Giovanni Neri, Pt–TiO2/MWCNTs hybrid composites for monitoring low hydrogen concentrations in air. Sensors, 12, 12361 (2012)
G.W. Hunter, P.G. Neudeck, R.S. Okojie, G.M. Beheim, V. Thomas, L. Chen, D. Lukco, C.C. Liu, B. Ward, D. Makel, Development of SiC gas sensor systems. Electrochem. Soc. Proc. 93, 2002 (2002)
L. Tien, H.T. Wang, B.S. Kang, F. Ren, P.W. Sadik, D.P. Norton, S.J. Pearton, J. Lin, Room-temperature hydrogen-selective sensing using single Pt-coated ZnO nanowires at microwatt power levels. Electrochem. Solid-State Lett. 8, G230 (2005)
Y. Lu, J. Li, H.T. Ng, C. Binder, C. Partridge, M. Meyyapan, Room temperature methane detection using palladium loaded single-walled carbon nanotube sensors. Chem. Phys. Lett. 391, 344 (2004)
H.T. Wang, B.S. Kang, F. Ren, L.C. Tien, P.W. Sadik, D.P. Notron, S.J. Pearton, Hydrogen-selective sensing at room temperature with ZnO nanorods. J. Lin. Appl. Phys. Lett. 86, 243503 (2005)
O. Kryliouk, H.J. Park, H.T. Wang, B.S. Kang, T.J. Anderson, F. Ren, S.J. Pearton, Pt-coated InN nanorods for selective detection of hydrogen at room temperature. J. Vac. Sci. Technol., B 23, 1891 (2005)
H.T. Wang, B.S. Kang, F. Ren, L.C. Tien, P.W. Sadik, D.P. Norton, S.J. Pearton, J. Lin, Detection of hydrogen at room temperature with catalyst-coated multiple ZnO nanorods. Appl. Phys. A 81, 1117 (2005)
C.H. Lin, W.C. Chang, X. Qi, Growth and Characterization of pure and doped SnO2 films for H2 gas detection. Proc. Eng. 36, 476 (2012)
ASTM, Powder Diffraction Files Inorganic Compounds (ASTM, Philadelphia, PA, 1979)
B. Wang, Y.H. Yang, C.X. Wang, N.S. Xu, G.W. Yang, Field emission and photoluminescence of SnO 2 nanograss. J. Appl. Phys. 98, 124303 (2005)
M.Y. Smirnov, A.V. Kalinkin, A.V. Pashis, I.P. Prosvirin, V.I. Bukhtiyarov, Interaction of SO2 with Pt model supported catalysts studied by XPS. J. Phys. Chem. C 118, 22120 (2014)
M. Sauvan, C. Pijolat, Selectivity improvement of SnO2 films by superficial metallic films. Sens. Actuators B. Chemical 58, 295 (1999)
A. Cabot, J. Arbiol, J.R. Morante, U. Weimar, N. Barsan, W. Gopel, Analysis of noble metal catalytic additives introduced by impregnation of as obtained SnO2 sol-gel nanocrystals for gas sensors. Sens. Actuators B. Chemical 70, 87 (2000)
L. De Luca, A. Donato, S. Santangelo, G. Faggio, G. Messina, N. Donato, G. Neri, Hydrogen sensing characteristics of Pt/TiO2/MWCNTs composites. Int. J. Hydr. Energy 37, 1842 (2012)
S. Volkening, K. Bedurftig, K. Jacobi, J. Wintterlin, G. Ertl, Dual-path mechanism for catalytic oxidation of hydrogen on platinum surfaces. Phys. Rev. Lett. 83, 2672 (1999)
Acknowledgments
One of the authors Mrs. Sapana Rane is grateful to Late Prof. R. N. Karekar for his valuable inputs and timely suggestions during this work. She also acknowledges Dr. K. R. Patil, NCL, Pune for XPS analysis and Mr. Saquib for the necessary experimental help.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rane, S., Arbuj, S., Rane, S. et al. Hydrogen sensing characteristics of Pt–SnO2 nano-structured composite thin films. J Mater Sci: Mater Electron 26, 3707–3716 (2015). https://doi.org/10.1007/s10854-015-2889-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-015-2889-3