Abstract
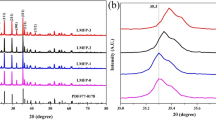
The low electronic conductivity and moderate lithium-ion transfer capability of Li2ZnTi3O8 restrict its application to some degree. In this work, in light of the high affinity of TiO2 surface with carboxylic acids, citric acid was adopted as a carbon source to form uniform carbon coating layer on LZTO particles. When the mixture of LZTO and citric acid with a mass ratio of 1:0.2 was roasted at 700 °C for 5 h, the product demonstrated wonderful rate performance (revealing capacities of 232.6, 202.8, 184.8, 165.6 and 106.7 mAh g−1 at 0.1, 0.2, 0.4, 0.8 and 1.6 A g−1, respectively) as well as outstanding long-term cycling stability (maintaining a capacity of 186.9 mAh g−1 for 1000 cycles at 0.5 A g−1). Combining structure and composition characterizations with electrochemical impedance spectra analysis, the amorphous carbon derived from citric acid was uniformly coated on the LZTO particles by virtue of the intense interaction between LZTO and carboxyl groups. Better particle dispersion and enhanced pseudocapacitive contribution were obtained resulted from the carbon coating, remarkably ameliorating the electronic and ionic conductivities of LZTO and alleviating polarization, thus beneficial to optimizing the electrochemical behavior of LZTO.
Graphic abstract








Similar content being viewed by others
References
Etacheri V, Marom R, Ran E, Salitra G, Aurbach D (2011) Challenges in the development of advanced Li-ion batteries. Energy Environ Sci 4:3243–3262
Nitta N, Wu F, Lee JT, Yushin G (2015) Li-ion battery materials: present and future. Mater Today 18:252–264
Zhang Y, Park S-J (2019) Stabilization of dispersed CuPd bimetallic alloy nanoparticles on ZIF-8 for photoreduction of Cr(VI) in aqueous solution. Chem Eng J 369:353. https://doi.org/10.1016/j.cej.2019.03.083
Zhang Y, Park S-J (2018) Formation of hollow MoO3/SnS2 heterostructured nanotubes for efficient light-driven hydrogen peroxide production. J Mater Chem A 6:20304–20312. https://doi.org/10.1039/c8ta08385a
Hernandez VS, Martinez LMT, Mather GC, West AR (1996) Stoichiometry, structures and polymorphism of spinel-like phases, Li1.33xZn2–2xTi1+0.67xO4. J Mater Chem 6:1533–1536
West A (1998) Crystal chemistry and physical properties of complex lithium spinels Li2MM′3O8 (M = Mg Co, Ni, Zn; M′ = Ti, Ge). J Mater Chem 8:1273–1280
Tang H, Tang Z, Du C, Qie F, Zhu J (2014) Ag-doped Li2ZnTi3O8 as a high rate anode material for rechargeable lithium-ion batteries. Electrochim Acta 120:187–192
Chen W, Zhou Z, Wang R et al (2015) High performance Na-doped lithium zinc titanate as anode material for Li-ion batteries. RSC Adv 5:49890–49898
Tang H, Zhu J, Tang Z, Ma C (2014) Al-doped Li2ZnTi3O8 as an effective anode material for lithium-ion batteries with good rate capabilities. J Electroanal Chem 731:60–66
Chen C, Ai C, Liu X, Wu Y (2017) Advanced electrochemical properties of Ce-modified Li2ZnTi3O8 anode material for lithium-ion batteries. Electrochim Acta 227:285–293
Yi TF, Wu JZ, Yuan J, Zhu Y, Wang P (2015) Rapid lithiation and de-lithiation property of V-doped Li2ZnTi3O8 as anode material for lithium-ion battery. ACS Sustainable Chem Eng 3:3062–3069
Li W, Wu L, Li Z, Lei G, Xiao Q, Ping Z (2011) Synthesis and electrochemical properties of Li2ZnTi3O8 fibers as an anode material for lithium-ion batteries. Electrochim Acta 56:5343–5346
Yang H, Park J, Kim C-S et al (2018) Boosted electrochemical performance of Li2ZnTi3O8 enabled by ion-conductive Li2ZrO3 concomitant with superficial Zr-doping. J Power Sources 379:270–277
Yang H, Wang XH, Qi YX, Lun N, Cao YM, Bai YJ (2017) Improving the electrochemical performance of Li2ZnTi3O8 by surface KCl modification. ACS Sustain Chem Eng 5:6099–6106
Yang H, Park J, Kim CS et al (2017) Uniform surface modification of Li2ZnTi3O8 by liquated Na2MoO4 to boost electrochemical performance. ACS Appl Mater Interfaces 9:43603–43613. https://doi.org/10.1021/acsami.7b12208
Yang H, Lun N, Qi Y-X et al (2019) Li2ZnTi3O8 coated with uniform lithium magnesium silicate layer revealing enhanced rate capability as anode material for Li-ion battery. Electrochim Acta 315:24–32
Tang H, Zan L, Zhu J, Ma Y, Zhao N, Tang Z (2016) High rate capacity nanocomposite lanthanum oxide coated lithium zinc titanate anode for rechargeable lithium-ion battery. J Alloys Compd 667:82–90
Tang H, Zhu J, Ma C, Tang Z (2014) Lithium cobalt oxide coated lithium zinc titanate anode material with an enhanced high rate capability and long lifespan for lithium-ion batteries. Electrochim Acta 144:76–84. https://doi.org/10.1016/j.electacta.2014.08.034
Li ZF, Li H, Cui YH et al (2017) Li2MoO4 modified Li2ZnTi3O8 as a high property anode material for lithium ion battery. J Alloys Compd 692:131–139. https://doi.org/10.1016/j.jallcom.2016.09.042
Tang H, Zan L, Mao W, Tang Z (2015) Improved rate performance of amorphous carbon coated lithium zinc titanate anode material with alginic acid as carbon precursor and particle size controller. J Electroanal Chem 751:57–64
Tang H, Tang Z (2014) Effect of different carbon sources on electrochemical properties of Li2ZnTi3O8/C anode material in lithium-ion batteries. J Alloys Compd 613:267–274
Wang L, Chen B, Meng Z, Luo B, Wang X, Zhao Y (2016) High performance carbon-coated lithium zinc titanate as an anode material for lithium-ion batteries. Electrochim Acta 188:135–144
Xu Y, Hong Z, Xia L, Yang J, Wei M (2013) One step sol–gel synthesis of Li2ZnTi3O8/C nanocomposite with enhanced lithium-ion storage properties. Electrochim Acta 88:74–78
Liu T, Tang H, Liu J et al (2018) Improved electrochemical performance of Li2ZnTi3O8 using carbon materials as loose and porous agent. Electrochim Acta 259:28–35
Li H, Zhou H (2012) Enhancing the performances of Li-ion batteries by carbon-coating: present and future. Chem Commun 48:1201–1217
Lu G, Liu J, Huang W, Wang X, Wang F (2019) Boosting the electrochemical performance of Li4Ti5O12 through nitrogen-doped carbon coating. Appl Organomet Chem 33:e4957. https://doi.org/10.1002/aoc.4957
Ren Y, Lu P, Huang X, Ding J, Wang H (2016) Synthesis and high cycle performance of Li2ZnTi3O8/C anode material promoted by asphalt as a carbon precursor. RSC Adv 6:49298–49306
Stenina I, Nikiforova P, Kulova T, Skundin A, Yaroslavtsev A (2017) Electrochemical properties of Li2ZnTi3O8/C nanomaterials. Nanotechnol Russ 12:605–612
Tang H, Chen C, Liu T, Tang Z (2019) Chitosan and chitosan oligosaccharide: advanced carbon sources are used for preparation of N-doped carbon-coated Li2ZnTi3O8 anode material. J Electroanal Chem 858:113789. https://doi.org/10.1016/j.jelechem.2019.113789
Chen C, Ai C, He Y, Yang S, Wu Y (2017) High performance Li2ZnTi3O8 coated with N-doped carbon as an anode material for lithium-ion batteries. J Alloys Compd 705:438–444
Balajka J, Hines MA, DeBenedetti WJ et al (2018) High-affinity adsorption leads to molecularly ordered interfaces on TiO2 in air and solution. Science 361:786–789
Huang M, Zhu H-L, Qi Y-X, Lun N, Bai Y-J (2019) A uniform few-layered carbon coating derived from self-assembled carboxylate monolayers capable of promoting the rate properties and durability of commercial TiO2. RSC Adv 9:36334–36342
Ren B, Fan M, Liu Q, Wang J, Song D, Bai X (2013) Hollow NiO nanofibers modified by citric acid and the performances as supercapacitor electrode. Electrochim Acta 92:197–204
Umemura K, Ueda T, Munawar SS, Kawai S (2012) Application of citric acid as natural adhesive for wood. J Appl Polym Sci 123:1991–1996
Venkatachalam N, Palanichamy M, Murugesan V (2007) Sol–gel preparation and characterization of alkaline earth metal doped nano TiO2: efficient photocatalytic degradation of 4-chlorophenol. J Mol Catal A: Chem 273:177–185
Holzwarth U, Gibson N (2011) The Scherrer equation versus the ‘Debye–Scherrer equation’. Nat Nanotechnol 6:534. https://doi.org/10.1038/nnano.2011.145
Kim J, Ji YK, Pham-Cong D et al (2016) Individually carbon-coated and electrostatic-force-derived graphene-oxide-wrapped lithium titanium oxide nanofibers as anode material for lithium-ion batteries. Electrochim Acta 199:35–44
Martins Ferreira EH, Moutinho MVO, Stavale F et al (2010) Evolution of the Raman spectra from single-, few-, and many-layer graphene with increasing disorder. Phys Rev B 82:125429. https://doi.org/10.1103/PhysRevB.82.125429
Ng SH, Wang J, Wexler D, Chew SY, Liu HK (2007) Amorphous carbon-coated silicon nanocomposites: A low-temperature synthesis via spray pyrolysis and their application as high-capacity anodes for lithium-ion batteries. J Phys Chem C 111:11131–11138
Rahman MM, Wang JZ, Hassan MF, Wexler D, Liu HK (2011) Amorphous carbon coated high grain boundary density dual phase Li4Ti5O12–TiO2: a nanocomposite anode material for Li-ion batteries. Adv Energy Mater 1:212–220
Wang S, Wang L, Meng Z, Xun R (2019) Design of a three-dimensional-network Li2ZnTi3O8 co-modified with graphene nanosheets and carbon nanotubes as a high performance anode material for lithium-ion batteries. J Alloys Compd 774:581–585
Wang S, Bi Y, Wang L, Meng Z, Luo B (2019) Mo-doped Li2ZnTi3O8@ graphene as a high performance anode material for lithium-ion batteries. Electrochim Acta 301:319–324
Li Z, Cui Y, Wu J, Du C, Zhang X, Tang Z (2016) Synthesis and electrochemical properties of lithium zinc titanate as an anode material for lithium ion batteries via microwave method. RSC Adv 6:39209–39215
Wang L, Meng Z, Wang H, Li X, Zhang G (2016) Effects of TiO2 starting materials on the synthesis of Li2ZnTi3O8 for lithium ion battery anode. Ceram Int 42:16872–16881
Wang G, Bradhurst D, Dou S, Liu H (1999) Spinel Li[Li1/3Ti5/3]O4 as an anode material for lithium ion batteries. J Power Sources 83:156–161
Shu J (2008) Study of the interface between Li4Ti5O12 electrodes and standard electrolyte solutions in 0.0–5.0 V. Electrochem Solid-State Lett 11:A238–A240
Lu W, Belharouak I, Liu J, Amine K (2007) Electrochemical and thermal investigation of Li4/3Ti5/3O4 spinel. J Electrochem Soc 154:A114–A118
Lindström H, Södergren S, Solbrand A et al (1997) Li+ ion insertion in TiO2 (anatase). 2. Voltammetry on nanoporous films. J Phys Chem B 101:7717–7722
Lou S, Cheng X, Zhao Y et al (2017) Superior performance of ordered macroporous TiNb2O7 anodes for lithium ion batteries: understanding from the structural and pseudocapacitive insights on achieving high rate capability. Nano Energy 34:15–25
Lim E, Jo C, Kim MS et al (2016) High-performance sodium-ion hybrid supercapacitor based on Nb2O5@ carbon core-shell nanoparticles and reduced graphene oxide nanocomposites. Adv Funct Mater 26:3711–3719
Shen L, Wang Y, Lv H et al (2018) Ultrathin Ti2Nb2O9 nanosheets with pseudocapacitive properties as superior anode for sodium-ion batteries. Adv Mater 30:1804378. https://doi.org/10.1002/adma.201804378
Wang J, Polleux J, Lim J, Dunn B (2007) Pseudocapacitive contributions to electrochemical energy storage in TiO2 (anatase) nanoparticles. J Phys Chem C 111:14925–14931
Zhang Y, Park S-J (2017) Au–Pd bimetallic alloy nanoparticle-decorated BiPO4 nanorods for enhanced photocatalytic oxidation of trichloroethylene. J Catal 355:1–10. https://doi.org/10.1016/j.jcat.2017.08.007
Zhang Y, Park S-J (2019) Facile construction of MoO3@ZIF-8 core–shell nanorods for efficient photoreduction of aqueous Cr (VI). Appl Catal B 240:92–101. https://doi.org/10.1016/j.apcatb.2018.08.077
Das S, Majumder S, Katiyar R (2005) Kinetic analysis of the Li+ ion intercalation behavior of solution derived nano-crystalline lithium manganate thin films. J Power Sources 139:261–268
Acknowledgements
This work was supported by Project ZR2019MEM029 of Shandong Provincial Natural Science Foundation, PR China, and National Natural Science Foundation of China (51902189).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, H., Zhu, HL., Qi, YX. et al. Optimizing the cycling life and high-rate performance of Li2ZnTi3O8 by forming thin uniform carbon coating derived from citric acid. J Mater Sci 55, 15538–15550 (2020). https://doi.org/10.1007/s10853-020-04980-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-020-04980-1