Abstract
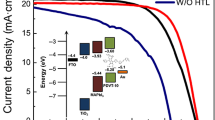
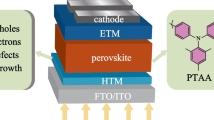
Designing organic hole transporting materials (HTMs) for stable perovskite photovoltaic devices remains a great challenge. Herein, we have prepared two small carbazole-based HTMs bearing a polymerizable double bond and one of the corresponding polymers. All compounds can be easily synthetized by a short procedure from largely available commercial products. These compounds have thermal, morphological, optical and electrochemical properties suitable for an application in perovskite solar cells. The side chains at the N-position on the carbazole have a negligible influence on the opto-electrochemical properties. However, the thermal properties differ largely between the monomer and its corresponding polymer. While these materials have the same photovoltaic performance, the polymeric HTM led to more stable devices. This illustrates the higher thermal stability of polymeric HTM versus its corresponding monomer.










Similar content being viewed by others
References
Calió L, Momblona C, Gil-Escrig L, Kazim S, Sessolo M, Sastre-Santos Á, Bolink HJ, Ahmad S (2017) Vacuum deposited perovskite solar cells employing dopant-free triazatruxene as the hole transport material. Sol Energy Mater Sol Cells 163:237–241. https://doi.org/10.1016/j.solmat.2017.01.037
Bui T-T, Shah SKSK, Abbas M, Sallenave X, Sini G, Hirsch L, Goubard F (2015) Carbazole-based molecular glasses as hole-transporting materials in solid state dye-sensitized solar cells. ChemNanoMat 1(3):203–210. https://doi.org/10.1002/cnma.201500014
Bui T-T, Goubard F (2013) Recent advances in small molecular, non-polymeric organic hole transporting materials for solid-state DSSC. EPJ Photovolt 4:40402. https://doi.org/10.1051/epjpv/2013024
Bui TT, Beouch L, Sallenave X, Goubard F (2013) Carbazol-N-Yl and diphenylamino end-capped triphenylamine-based molecular glasses: synthesis, thermal, and optical properties. Tetrahedron Lett 54(32):4277–4280
Xue Y, Wu Y, Li Y (2017) Readily synthesized dopant-free hole transport materials with phenol core for stabilized mixed perovskite solar cells. J Power Sources 344:160–169. https://doi.org/10.1016/j.jpowsour.2017.01.121
Al Mousawi A, Lara DM, Noirbent G, Dumur F, Toufaily J, Hamieh T, Bui T-T, Goubard F, Graff B, Gigmes D et al (2017) Carbazole derivatives with thermally activated delayed fluorescence property as photoinitiators/photoredox catalysts for LED 3D printing technology. Macromolecules 50(13):4913–4926. https://doi.org/10.1021/acs.macromol.7b01114
Al Mousawi A, Dumur F, Garra P, Toufaily J, Hamieh T, Goubard F, Bui T-T, Graff B, Gigmes D, Pierre Fouassier J et al (2017) Azahelicenes as visible light photoinitiators for cationic and radical polymerization: preparation of photoluminescent polymers and use in high performance LED projector 3D printing resins. J Polym Sci Part A Polym Chem 55(7):1189–1199. https://doi.org/10.1002/pola.28476
Bui T-T, Goubard F, Ibrahim-Ouali M, Gigmes D, Dumur F (2018) Thermally activated delayed fluorescence emitters for deep blue organic light emitting diodes: a review of recent advances. Appl Sci 8(4):494. https://doi.org/10.3390/app8040494
Bui T-T, Goubard F, Ibrahim-Ouali M, Gigmes D, Dumur F (2018) Recent advances on organic blue thermally activated delayed fluorescence (TADF) emitters for organic light-emitting diodes (OLEDs). Beilstein J Org Chem 14(1):282–308. https://doi.org/10.3762/bjoc.14.18
Sathiyan G, Sivakumar EKT, Ganesamoorthy R, Thangamuthu R, Sakthivel P (2016) Review of carbazole based conjugated molecules for highly efficient organic solar cell application. Tetrahedron Lett 57(3):243–252. https://doi.org/10.1016/j.tetlet.2015.12.057
Di Giacomo F, Zardetto V, D’Epifanio A, Pescetelli S, Matteocci F, Razza S, Di Carlo A, Licoccia S, Kessels WMM, Creatore M et al (2015) Flexible perovskite photovoltaic modules and solar cells based on atomic layer deposited compact layers and UV-irradiated TiO2 scaffolds on plastic substrates. Adv Energy Mater 5(8):1401808. https://doi.org/10.1002/aenm.201401808
Kim HS, Jang IH, Ahn N, Choi M, Guerrero A, Bisquert J, Park NG (2015) Control of I–V hysteresis in CH3NH3PbI3 perovskite solar cell. J Phys Chem Lett 6(22):4633–4639. https://doi.org/10.1021/acs.jpclett.5b02273
Ulfa M, Zhu T, Goubard F, Pauporté T (2018) Molecular versus polymeric hole transporting materials for perovskite solar cell application. J Mater Chem A 6(27):13350–13358. https://doi.org/10.1039/C8TA03875A
Hsu C-Y, Chen Y-C, Lin RY-Y (2012) Solid-state dye-sensitized solar cells based on spirofluorene (Spiro-OMeTAD) and arylamines as hole transporting materials. Phys Chem Chem Phys 14(41):14099–14109. https://doi.org/10.1039/c2cp41326d
Holliday S, Donaghey JE, McCulloch I (2014) Advances in charge carrier mobilities of semiconducting polymers used in organic transistors. Chem Mater 26(1):647–663. https://doi.org/10.1021/cm402421p
Arias AC, MacKenzie JD, McCulloch I, Rivnay J, Salleo A (2010) Materials and applications for large area electronics: solution-based approaches. Chem Rev 110(1):3–24. https://doi.org/10.1021/cr900150b
Zaumseil J, Sirringhaus H (2007) Electron and ambipolar transport in organic field-effect transistors. Chem Rev 107(4):1296–1323. https://doi.org/10.1021/CR0501543
Bui T-T, Shah SK, Sallenave X, Abbas M, Sini G, Hirsch L, Goubard F (2015) Di(p-methoxyphenyl)amine end-capped tri(p-thiophenylphenyl)amine based molecular glasses as hole transporting materials for solid-state dye-sensitized solar cells. RSC Adv 5(61):49590–49597. https://doi.org/10.1039/c5ra07226c
Bui T-T, Goubard F, Troughton J, Watson T (2017) Simple 3,6-bis(diphenylaminyl)carbazole molecular glasses as hole transporting materials for hybrid perovskite solar cells. J Mater Sci Mater Electron 28(23):17551–17556. https://doi.org/10.1007/s10854-017-7691-y
Cho A-N, Kim H-S, Bui T-T, Sallenave X, Goubard F, Park N-G (2016) Role of LiTFSI in High Tg triphenylamine-based hole transporting material in perovskite solar cell. RSC Adv 6(72):68553–68559. https://doi.org/10.1039/c6ra12574c
Daskeviciene M, Paek S, Wang Z, Malinauskas T, Jokubauskaite G, Rakstys K, Cho KT, Magomedov A, Jankauskas V, Ahmad S et al (2017) Carbazole-based enamine: low-cost and efficient hole transporting material for perovskite solar cells. Nano Energy 32:551–557. https://doi.org/10.1016/j.nanoen.2017.01.015
Rakstys K, Paek S, Gao P, Gratia P, Marszalek T, Grancini G, Cho KT, Genevicius K, Jankauskas V, Pisula W et al (2017) Molecular engineering of face-on oriented dopant-free hole transporting material for perovskite solar cells with 19% PCE. J Mater Chem A 5(17):7811–7815. https://doi.org/10.1039/C7TA01718A
Zhang M-D, Zheng B-H, Zhuang Q-F, Huang C-Y, Cao H, Chen M-D, Wang B (2017) Two dimethoxyphenylamine-substituted carbazole derivatives as hole-transporting materials for efficient inorganic-organic hybrid perovskite solar cells. Dye Pigment 146:589–595. https://doi.org/10.1016/J.DYEPIG.2017.07.037
Zhao X, Wang M (2018) Organic hole-transporting materials for efficient perovskite solar cells. Mater Today Energy 7:208–220. https://doi.org/10.1016/j.mtener.2017.09.011
Berton N, Nakar R, Schmaltz B (2019) DMPA-containing carbazole-based hole transporting materials for perovskite solar cells: recent advances and perspectives. Synth Metals. https://doi.org/10.1016/j.synthmet.2019.04.004
Torres A, Rego LGC (2014) Surface effects and adsorption of methoxy anchors on hybrid lead iodide perovskites: insights for Spiro-MeOTAD attachment. J Phys Chem C 118(46):26947–26954. https://doi.org/10.1021/jp510595s
Bui T-T, Ulfa M, Maschietto F, Ottochian A, Nghiêm M-P, Ciofini I, Goubard F, Pauporté T (2018) Design of dendritic core carbazole-based hole transporting materials for efficient and stable hybrid perovskite solar cells. Org Electron 60:22–30. https://doi.org/10.1016/j.orgel.2018.05.024
Ulfa M, Pauporte T, Bui T-T, Goubard F (2018) Impact of organic hole transporting material and doping on the electrical response of perovskite solar cells. J Phys Chem C 122(22):11651–11658. https://doi.org/10.1021/acs.jpcc.8b02141
Abdallah M, Magaldi D, Hijazi A, Graff B, Dumur F, Fouassier J, Bui T, Goubard F, Lalevée J (2019) Development of new high-performance visible light photoinitiators based on carbazole scaffold and their applications in 3D printing and photocomposite synthesis. J Polym Sci Part A Polym Chem. https://doi.org/10.1002/pola.29471
Konishi A, Yoda N, Marvel CS (1965) Base-catalyzed polymerization of p-styrenesulfonamides. II. N-methyl and N-phenyl derivatives. J Polym Sci Part A Gen Pap 3(11):3833–3842. https://doi.org/10.1002/pol.1965.100031114
Jenkins AD, Jones RG, Moad G (2009) Terminology for reversible-deactivation radical polymerization previously called “controlled” radical or “living” radical polymerization (IUPAC Recommendations 2010). Pure Appl Chem 82(2):483–491. https://doi.org/10.1351/PAC-REP-08-04-03
Mayo FR (1943) Chain transfer in the polymerization of styrene: the reaction of solvents with free radicals1. J Am Chem Soc 65(12):2324–2329. https://doi.org/10.1021/ja01252a021
Flory PJ (1937) The mechanism of vinyl polymerizations. J Am Chem Soc 59(2):241–253. https://doi.org/10.1021/ja01281a007
Furuncuoǧlu T, Uǧur I, Degirmenci I, Aviyente V (2010) Role of chain transfer agents in free radical polymerization kinetics. Macromolecules 43(4):1823–1835. https://doi.org/10.1021/ma902803p
Matyjaszewski K, Davis TP (2002) Handbook of radical polymerization, vol 125. John Wiley & Sons, Inc., USA. https://doi.org/10.1002/0471220450
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA et al (2009) Gaussian, Inc., Wallingford CT. Gaussian, Inc., Wallingford, p 2009
Chai JD, Head-Gordon M (2008) Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections. Phys Chem Chem Phys 10(44):6615–6620. https://doi.org/10.1039/b810189b
Pommerehne J, Vestweber H, Guss W, Mahrt RF, Bässler H, Porsch M, Daub J (1995) Efficient two layer leds on a polymer blend basis. Adv Mater 7(6):551–554. https://doi.org/10.1002/adma.19950070608
Cardona CM, Li W, Kaifer AE, Stockdale D, Bazan GC (2011) Electrochemical considerations for determining absolute frontier orbital energy levels of conjugated polymers for solar cell applications. Adv Mater 23(20):2367–2371. https://doi.org/10.1002/adma.201004554
Wang P, Ulfa M, Pauporté T (2018) Effects of perovskite monovalent cation composition on the high and low frequency impedance response of efficient solar cells. J Phys Chem C 122(4):1973–1981. https://doi.org/10.1021/acs.jpcc.7b11010
Zhao X, Kim HS, Seo JY, Park NG (2017) Effect of selective contacts on the thermal stability of perovskite solar cells. ACS Appl Mater Interfaces 9(8):7148–7153. https://doi.org/10.1021/acsami.6b15673
Acknowledgements
M.U. and D.M. acknowledge the Indonesia Endowment Fund for Education (LPDP) and the Mexican government for their Ph.D. scholarship, respectively.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Magaldi, D., Ulfa, M., Nghiêm, MP. et al. Hole transporting materials for perovskite solar cells: molecular versus polymeric carbazole-based derivatives. J Mater Sci 55, 4820–4829 (2020). https://doi.org/10.1007/s10853-019-04342-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-019-04342-6