Abstract
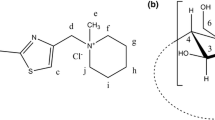
Aqueous-solution complexes of β-cyclodextrin (β-CD) with metoprolol tartrate (MET) have been analysed with 1H NMR and UV–vis spectroscopy. With 1H NMR a [1:1] stoichiometry could be established for the β-CD-MET complex while its stability constant was determined with UV–vis spectroscopy. Powder diffraction data of a polycrystalline sample of the β-CD-MET complex show that a novel product has been formed, likely to be a β-CD-MET [1:1] inclusion complex. Also Hyperchem MM+ molecular-dynamics results suggest an inclusion complex and from 1H NMR data it is inferred that probably the MET is docked in the CD with the formers methoxyethyl-benzene moiety in front.









Similar content being viewed by others
References
Moloney, G., Craik, D.J., Irkouder, M.N., Nero, T.L.: Synthesis, NMR studies and conformational analysis of oxazilidine derivatives of the beta-adrenoreceptor antagonists metoprolol, atenolol and timolol. J.C.S. Perkin Trans. 2, 199–206 (1998).
Szjejtli, J.: Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 98, 1743–1753 (1998).
Ficarra, R., Ficarra, P., Di Bella, M.R., Raneri, D., Tommasini, S., Calabrò, M.L., Gamberini, M.C., Rustichelli, C.: Study of beta-blockers/beta-cyclodextrins inclusion complex by NMR, DSC, X-ray and SEM investigation. J. Pharm. Biomed. Anal. 23, 33–40 (2000).
Park, K.L., Kim, K.H., Jung, S.-H., Lim, H.M., Hong, Ch.H., Kang, J.S.: Enantioselective stabilization of inclusion complexes of metoprolol in carboxymethylated β-cyclodextrin. J. Pharm. Biomed. Anal 27, 569–576 (2002).
Radulovic, D.M., Karljikovic-Rajic, K.D., Lucic, B.M., Vujic, Z.B.: A preliminary study of beta-cyclodextrin/metoprolol tartrate inclusion complex for potential enantiomeric separation. J. Pharm Biomed. Anal. 24, 871–876 (2001).
Bratu, I., Gavira-Vallejo, J.M., Hernanz, A.: 1H-NMR study of the inclusion processes for α- and γ-cyclodextrin with fenbufen. Biopolymers 77, 361–367 (2005).
Ikeda, Y., Hirayama, F., Arima, H., Uekama, K., Yoshitake, Y., Harano, K.: NMR spectroscopic characterization of metoprolol/cyclodextrin complexes in aqueous solution: cavity size dependency. J. Pharm. Sci. 93, 1659–1671 (2004).
Job, P.: Formation and stabitity of inorganic complexes in solution. Ann. Chem. 9, 113–203 (1928).
Bogdan, M., Farcas, S.I., Bojita, M.: Nuclear magnetic resonance study of naproxen and ibuprofen interaction with beta-CD in solution. Studia Univ. Babes-Bolyai, Ser. Phys. Special issue 115–120 (2000).
Bratu, I., Gavira-Vallejo, J.M., Hernanz, A., Bogdan, M., Bora, Gh.: Inclusion complex of fenbufen with β-cyclodextrin. Biopolymers-Biospectroscopy 73, 451–456 (2004).
Saenger, W.: Cyclodextrin inclusion compounds in research and industry. Angew. Chem. Int. Ed. Engl. 92, 343–361 (1980).
Li, S., Purdy, W.C.: Cyclodextrins and their applications in analytical chemistry. Chem. Rev. 92, 1457–1470 (1992).
Junquera, E., Aicart, A.: Potentiometric study of the encapsulation of ketoprophen by hydroxypropyl-β- cyclodextrin. Temperature, solvent, and salt effects. J. Phys. Chem. B 101, 7163–7171 (1997).
Tran, Ch.D., De Paoli Lacerda, S.H.: Determination of binding constants of cyclodextrins in room-temperature ionic liquids by near-infrared spectrometry. Anal. Chem. 74, 5337–5341 (2002).
Scott, R.L.: Some comments on the Benesi-Hildebrand equation. Rec. Trav. Chim. 75, 787–789 (1956).
Liu, L., Guo, Q.-X.: The driving forces in the inclusion complexation of cyclodextrins. J. Inclus. Phenom. Macro. Chem. 42, 1–14 (2002).
HyperChemTM Release 4, Hypercube, Inc. (1994), Waterloo, Ontario N2l 3X2, Canada.
“Computational Chemistry”, Hypercube, Inc. Waterloo, Ontario N2l 3X2, Canada (1994).
Origin® 7.0, OriginLab Corporation, Northhampton, MA 01060, USA.
Zubiaur, M., Jaime, C.: Complexation between tert-butyl ketones and β-cyclodextrin. Structural study by NMR and MD simulations. J. Org. Chem. 65(24), 8139–8145 (2000).
Gavira, J.M., Hernanz, A., Bratu, I.: Dehydration of β-cyclodextrin: An IR ν (OH) band profile analysis. Vib. Spectrosc. 32(2), 137–146 (2003).
Acknowledgments
The authors acknowledge the ESRF (Grenoble, France) for the opportunity to perform the synchrotron diffraction experiments and Dr. H. Emerich for his help at beamline BM01B (Swiss-Norwegian CRG). They also thank Dr. M.M. Pop, Dr. V. Brodski and Dr. D.J.A. De Ridder for their assistance in collecting the data. One of the authors (Borodi) is indebted to the Netherlands Foundation for Scientific research (NWO) for financial support (NB 71-135) to carry out part of the research in Amsterdam. The spectroscopic investigations and molecular modelling were supported by the CEEX no7/2005 VIASAN project. Thanks are due also to Dr. L. Copolovici from the Faculty of Chemistry, “Babes-Bolyai” University for his assistance in the determination of the association constant from the optical spectra.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mihaela Toma is Socrates/Erasmus student at UNED Madrid
Rights and permissions
About this article
Cite this article
Dragan, F., Bratu, I., Borodi, G. et al. Spectroscopic investigation of β-cyclodextrin -metoprolol tartrate inclusion complexes. J Incl Phenom Macrocycl Chem 59, 125–130 (2007). https://doi.org/10.1007/s10847-007-9304-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-007-9304-5