Abstract
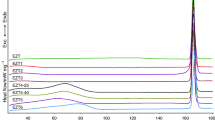
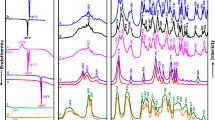
The amorphous complex of 2-hydroxypropyl-β-cyclodextrin (HP-β-CyD) with an oral hypoglycemic agent, chlorpropamide (CPM), in a molar ratio of 1:1 was prepared by the spray-drying method. The effects of storage (temperature and humidity) and moulding pressure on the polymorphic transition of CPM in HP-β-CyD matrix were investigated, in comparison with those of the CPM polymorphs, Form A (stable form) and Form C (metastable form). The formation of an amorphous complex of CPM with HP-β-CyD was confirmed by powder X-ray diffractometry and differential scanning calorimetry. During storage at various temperature and humidity conditions, the metastable Form C of CPM converted to the stable Form A, where the conversion proceeded according to the Jander equation with an activation energy of 51 kJ/mol (25–60–°C) and a reaction-order of 1.55 with respect to water content (relative humidity (RH) 20–75%). No polymorphic transition of Form A crystals was observed under the experimental conditions. In the case of the amorphous HP-β-CyD complex, Form C crystals were slowly produced, but the further conversion of the resulting Form C to Form A was markedly suppressed in HP-β-CyD matrix. Upon compression (2000kg/cm2), Forms A and C were converted to amorphous CPM in a major portion and Forms C and A, respectively, in a minor portion. The polymorphic transition behavior was clearly reflected in the dissolution rate of CPM, i.e., (1) the dissolution rate was in the order of HP-β-CyD complex (Form C) Form A, and (2) the dissolution rate of Forms A and C after the compression increased because of the conversion to amorphous state, while the complex maintained the fast dissolving property even after the compression. The results indicated that HP-β-CyD is useful not only for converting crystalline CPM to an amorphous substance, but also for maintaining the metastable form with fast dissolution rate, Form C, over a long period.
Similar content being viewed by others
References
J. Haleblian W. McCrone (1969) ArticleTitle␣ J. Pharm. Sci. 58 911
H.G. Brittain S.J. Bogdanowich D.E. Bugay J.D. Vincentis G. Lewen A.W. Newman (1991) Pharm. Res. 8 963
K. Uekama F. Hirayama T. Irie (1999) Chem. Rev. 98 2045
4.K. Uekama (Theme Editor):Adv. Drug Deliv. Rev. 36: 1 (1999)
E. Albers B.W. Müller (1996) CRC Crit. Rev. Ther. Drug. Carrier. Syst. 12 311
J. Horsky J. Pitha (1996) J. Pharm. Sci. 85 96
T. Loftsson M.E. Brewster (1996) J. Pharm. Sci. 85 1017
K. Kimura F. Hirayama H. Arima K. Uekama (1996) Pharm. Res. 16 1729
K. Kimura F. Hirayama H. Arima K. Uekama (1999) Chem. Pharm. Bull 48 646
H. Ueda N. Nambu T. Nagai (1989) Chem. Pharm. Bull. 32 244
M. Otsuka T. Matsumoto N. Kaneniwa (1983) J. Pharm. Pharmacol. 41 665
H . Hyqvisty (1983) Int. J. Pharm. Technol. Prod. Mfr. 4 47
H. Nogami T. Nagai Y. Yotsuyanagi (1969) Chem. Pharm. Bull 17 499
J.D. Hancock J.H. Sharp (1972) J. Am. Ceram. Soc 55 74
K. Kimura F. Hirayama K. Uekama (1999) J. Pharm. Sci. 88 385
J.T. Carstensen E.S. Aron D.C. Spera J.J. Vance (1966) J. Pharm. Sci. 55 561
Z. Ostwald (1897) Physik. Chem. 22 209
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sonoda, Y., Hirayama, F., Arima, H. et al. Effects of 2-Hydroxypropyl-b-cyclodextrin on Polymorphic Transition of Chlorpropamide in Various Conditions: Temperature, Humidity and Moulding Pressure. J Incl Phenom Macrocycl Chem 50, 73–77 (2004). https://doi.org/10.1007/s10847-003-8842-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10847-003-8842-8