Abstract
Purpose
To examine associations between objectively measured sleep duration and sleep timing with odds of completion of an in vitro fertilization (IVF) cycle.
Methods
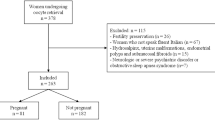
This prospective cohort study enrolled 48 women undergoing IVF at a large tertiary medical center between 2015 and 2017. Sleep was assessed by wrist-worn actigraphy, 1–2 weeks prior to initiation of the IVF cycle. Reproductive and IVF cycle data and demographic and health information were obtained from medical charts. Sleep duration, midpoint, and bedtime were examined in relation to IVF cycle completion using logistic regression models, adjusted for age and anti-Müllerian hormone levels. A sub-analysis excluded women who worked non-day shifts to control for circadian misalignment.
Results
The median age of all participants was 33 years, with 29% of women >35 years. Ten women had an IVF cycle cancelation prior to embryo transfer. These women had shorter sleep duration, more nocturnal awakenings, lower sleep efficiency, and later sleep timing relative to those who completed their cycle. Longer sleep duration was associated with lower odds of uncompleted IVF cycle (OR = 0.88; 95%CI 0.78, 1.00, per 20-min increment of increased sleep duration). Women with later sleep midpoint and later bedtime had higher odds of uncompleted cycle relative to those with earlier midpoint and earlier bedtime; OR = 1.24; 95%CI 1.09, 1.40 and OR = 1.33; 95%CI 1.17, 1.53 respectively, for 20-min increments. These results were independent of age, anti-Müllerian hormone levels, or sleep duration, and remained significant after exclusion of shift-working women.
Conclusions
Shorter sleep duration and later sleep timing increase the odds of uncompleted cycles prior to embryo transfer.


Similar content being viewed by others
References
Thoma ME, McLain AC, Louis JF, King RB, Trumble AC, Sundaram R, et al. Prevalence of infertility in the United States as estimated by the current duration approach and a traditional constructed approach. Fertil Steril. 2013;99:1324–31.e1.
Louis JF, Thoma ME, Sørensen DN, McLain AC, King RB, Sundaram R, et al. The prevalence of couple infertility in the United States from a male perspective: evidence from a nationally representative sample. Andrology. 2013;1:741–8.
Boivin J, Bunting L, Collins JA, Nygren KG. International estimates of infertility prevalence and treatment-seeking: potential need and demand for infertility medical care. Hum Reprod. 2007;22:1506–12.
De Geyter C, Calhaz-Jorge C, Kupka MS, Wyns C, Mocanu E, Motrenko T, et al. ART in Europe, 2014: results generated from European registries by ESHRE: the European IVF-monitoring Consortium (EIM) for the European Society of Human Reproduction and Embryology (ESHRE). Hum Reprod. 2018;33:1586–601.
SART. Society of Assisted Reproductive Technology national summary report 2017. In, 2019.
Matthiesen S, Frederiksen Y, Ingerslev HJ, Zachariae RJHr. Stress, distress and outcome of assisted reproductive technology (ART): a meta-analysis 2011;26:2763-76.
Kalmbach DA, Anderson JR, Drake CLJJosr. The impact of stress on sleep: pathogenic sleep reactivity as a vulnerability to insomnia and circadian disorders 2018;27:e12710.
Kloss JD, Perlis ML, Zamzow JA, Culnan EJ, Gracia CR. Sleep, sleep disturbance, and fertility in women. Sleep Med Rev. 2015;22:78–87.
Willis SK, Hatch EE, Wesselink AK, Rothman KJ, Mikkelsen EM, Wise LAJF et al. Female sleep patterns, shift work, and fecundability in a North American preconception cohort study 2019;111:1201-10. e1.
Lin YH, Chueh KH, Lin JLJJocn. Somatic symptoms, sleep disturbance and psychological distress among women undergoing oocyte pick-up and in vitro fertilisation–embryo transfer 2016;25:1748-56.
Huang L-H, Kuo C-P, Lu Y-C, Lee M-S, Lee S-HJTJO. Gynecology. Association of emotional distress and quality of sleep among women receiving in-vitro fertilization treatment. 2019;58:168–72.
Goldstein CA, Lanham MS, Smith YR, O’Brien LM. Sleep in women undergoing in vitro fertilization: a pilot study. Sleep Med. 2017;32:105–13.
Mills J, Kuohung W. Impact of circadian rhythms on female reproduction and infertility treatment success. Curr Opin Endocrinol Diabetes Obes. 2019;26:317–21.
Willis SK, Hatch EE, Wise LA. Sleep and female reproduction. Curr Opin Obstet Gynecol. 2019;31:222–7.
Penzias A, Bendikson K, Butts S, Coutifaris C, Falcone T, Fossum G, et al. ASRM standard embryo transfer protocol template: a committee opinion. Fertil Steril. 2017;107:897–900.
Ancoli-Israel S, Cole R, Alessi C, Chambers M, Moorcroft W, Pollak CP. The role of actigraphy in the study of sleep and circadian rhythms. Sleep. 2003;26:342–92.
Smith MT, McCrae CS, Cheung J, Martin JL, Harrod CG, Heald JL, et al. Use of actigraphy for the evaluation of sleep disorders and circadian rhythm sleep-wake disorders: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2018;14:1231–7.
Patel SR, Weng J, Rueschman M, Dudley KA, Loredo JS, Mossavar-Rahmani Y, et al. Reproducibility of a standardized actigraphy scoring algorithm for sleep in a US Hispanic/Latino population. Sleep. 2015;38:1497–503.
Jirge PR. Poor ovarian reserve. Journal of human reproductive sciences. 2016;9:63–9.
Stocker LJ, Macklon NS, Cheong YC, Bewley SJJO. Influence of shift work on early reproductive outcomes: a systematic review and meta-analysis. Gynecology. 2014;124:99–110.
NSF. National Sleep Foundation recommends new sleep times. In, 2015.
Amyx M, Xiong X, Xie Y, Buekens P. Racial/ethnic differences in sleep disorders and reporting of trouble sleeping among women of childbearing age in the United States. Matern Child Health J. 2017;21:306–14.
Touzet S, Rabilloud M, Boehringer H, Barranco E, RJF E. Relationship between sleep and secretion of gonadotropin and ovarian hormones in women with normal cycles. Sterility. 2002;77:738–44.
Michels KA, Mendola P, Schliep KC, Yeung EH, Ye A, Dunietz GL et al. The influences of sleep duration, chronotype, and nightwork on the ovarian cycle 2020;37:260-71.
Willis SK, Hatch EE, Wesselink AK, Rothman KJ, Mikkelsen EM, Wise LA. Female sleep patterns, shift work, and fecundability in a North American preconception cohort study. Fertil Steril. 2019;111:1201–10. e1.
Grajewski B, Whelan EA, Lawson CC, Hein MJ, Waters MA, Anderson JL, et al. Miscarriage among flight attendants. Epidemiology. 2015;26:192–203.
Fernandez RC, Marino JL, Varcoe TJ, Davis S, Moran LJ, Rumbold AR, et al. Fixed or rotating night shift work undertaken by women: implications for fertility and miscarriage. Semin Reprod Med. 2016;34:74–82.
Begtrup LM, Specht IO, Hammer PEC, Flachs EM, Garde AH, Hansen J, et al. Night work and miscarriage: a Danish nationwide register-based cohort study. Occup Environ Med. 2019;76:302–8.
Labyak S, Lava S, Turek F, Zee P. Effects of shiftwork on sleep and menstrual function in nurses. Health care for women international. 2002;23:703–14.
Bisanti L, Olsen J, Basso O, Thonneau P, Karmaus W. Shift work and subfecundity: a European multicenter study. European Study Group on Infertility and Subfecundity. J Occup Environ Med. 1996;38:352–8.
Aspholm R, Lindbohm ML, Paakkulainen H, Taskinen H, Nurminen T, Tiitinen A. Spontaneous abortions among Finnish flight attendants. J Occup Environ Med. 1999;41:486–91.
Knutsson A. Health disorders of shift workers. Occupational medicine (Oxford, England) 2003;53:103-8.
Liu C, Politch JA, Cullerton E, Go K, Pang S, Kuohung W. Impact of daylight savings time on spontaneous pregnancy loss in in vitro fertilization patients. Chronobiol Int. 2017;34:571–7.
Kirshenbaum M, Ben-David A, Zilberberg E, Elkan-Miller T, Haas J, Orvieto R. Influence of seasonal variation on in vitro fertilization success. PLoS One. 2018;13:e0199210.
Mínguez-Alarcón L, Souter I, Williams PL, Ford JB, Hauser R, Chavarro JE et al. Occupational factors and markers of ovarian reserve and response among women at a fertility centre 2017;74:426-31.
Sen A, Hoffmann HM. Role of core circadian clock genes in hormone release and target tissue sensitivity in the reproductive axis. Mol Cell Endocrinol. 2020;501:110655.
Lateef OM, Akintubosun MO. Sleep and reproductive health. J Circadian Rhythms. 2020;18:1.
Smeenk JMJ, Verhaak CM, Vingerhoets AJJM, Sweep CGJ, Merkus JMWM, Willemsen SJ, et al. Stress and outcome success in IVF: the role of self-reports and endocrine variables. Hum Reprod. 2005;20:991–6.
du Plessis SS, Makker K, Desai NR, Agarwal A. Impact of oxidative stress on IVF. Expert Review of Obstetrics & Gynecology. 2008;3:539–54.
Nikolettos N, Asimakopoulos B, Nicolettos N, Efthimiadou A, Mourvati E, Demirel C. Evaluation of leptin, interleukin-1β, tumor necrosis factor-α and vascular endothelial growth factor in serum and follicular fluids of women undergoing controlled ovarian hyperstimulation as prognostic markers of ICSI outcome. In vivo (Athens, Greece) 2004;18:667-74.
Singh AK, Chattopadhyay R, Chakravarty B, Chaudhury K. Markers of oxidative stress in follicular fluid of women with endometriosis and tubal infertility undergoing IVF. Reprod Toxicol. 2013;42:116–24.
Williams WP 3rd, Kriegsfeld LJ. Circadian control of neuroendocrine circuits regulating female reproductive function. Front Endocrinol (Lausanne). 2012;3:60.
Cahill GM, Hurd MW, Batchelor MM. Circadian rhythmicity in the locomotor activity of larval zebrafish. Neuroreport. 1998;9:3445–9.
Khattab AF, Mustafa FA, Taylor PJ. The use of urine LH detection kits to time intrauterine insemination with donor sperm. Hum Reprod. 2005;20:2542–5.
Rossmanith WG. Ultradian and circadian patterns in luteinizing hormone secretion during reproductive life in women. Hum Reprod. 1993;8(Suppl 2):77–83.
Kennaway DJ, Boden MJ, Varcoe TJ. Circadian rhythms and fertility. Mol Cell Endocrinol. 2012;349:56–61.
Sakai N, Endo A. Effects of delayed mating on preimplantation embryos in spontaneously ovulated mice. Gamete Res. 1988;19:381–5.
Endo A, Watanabe T. Effects of non-24-hour days on reproductive efficacy and embryonic development in mice. Gamete Res. 1989;22:435–41.
Summa KC, Vitaterna MH, Turek FW. Environmental perturbation of the circadian clock disrupts pregnancy in the mouse. PLoS One. 2012;7:e37668.
Boden MJ, Varcoe TJ, Voultsios A, Kennaway DJ. Reproductive biology of female Bmal1 null mice. Reproduction. 2010;139:1077–90.
Fischer D, Lombardi DA, Marucci-Wellman H, Roenneberg T. Chronotypes in the US – influence of age and sex. PLoS One. 2017;12:e0178782.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pimolsri, C., Lyu, X., Goldstein, C. et al. Objective sleep duration and timing predicts completion of in vitro fertilization cycle. J Assist Reprod Genet 38, 2687–2696 (2021). https://doi.org/10.1007/s10815-021-02260-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-021-02260-8