Abstract
Purpose
To compare E-selectin, resistin and reactive oxygen species (ROS) levels in serum and follicular fluid (FF) of subfertile women undergoing Controlled Ovarian Hyperstimulation (COH) during IVF/ICSI cycles, using GnRH-agonist and -antagonist protocols.
Methods
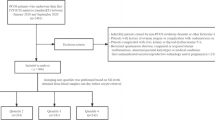
In this prospective cohort study, 85 subfertile women undergoing IVF/ICSI were included. Participants underwent the GnRH-agonist and -antagonist protocols; and blood samples were collected at three time points: basic (at start of COH), on the day of hCG and at oocyte retrieval (OR); and from the FF from the first follicle aspirate. Clinical and IVF cycle characteristics, were compared between groups, together with the levels of E-selectin, resistin and ROS in serum and FF, through ELISA. Their prognostic value on pregnancy outcomes was examined.
Result(s)
Examining molecules levels are increasing in serum, from start of COH until OR, irrespectively of the protocol used; FF levels at OR were similar to those in serum at that day. Resistin FF levels were lower in GnRH agonists, compared with the antagonist protocol. Resistin levels at start of COH were associated with clinical pregnancy rates, and this remained significant following adjustment for age, BMI and IVF protocol used, while values of >13.5 ng/ml were associated with a six times greater odd of a pregnancy.
Conclusion
E-selectin, resistin and ROS levels are increasing during COH, reaching their highest values at OR, with comparable values measured in the FF at that time. Resistin values >13.5 ng/ml are linked with a 6-fold increase on the odds of a pregnancy.


Similar content being viewed by others
References
Abramov Y, Schenker JG, Lewin A, Kafka I, Jaffe H, Barak V. Soluble ICAM-1 and E-selectin levels correlate with clinical and biological aspects of severe ovarian hyperstimulation syndrome. Fertil Steril. 2001;76:51–7.
Wang B, Sheng J-Z, He R-H, Qian Y-L, Jin F, Huang H-F. High expression of L selectin ligand in secretory endometrium is associated with better endometrial receptivity and facilitates embryo implantation in human being. Am J Reprod Immunol. 2008;60:127–34.
Orvieto R, Ben-Rafael Z, Abir R, Bar-Hava I, Fisch B, Molad Y. Controlled ovarian hyperstimulation: a state of neutrophil activation. Am J Reprod Immunol. 1999;42:288–91.
Lai TH, Zhao Y, Shih IM, Ho CL, Bankowski B, Vlahos N. Expression of L-selectin ligands in human endometrium during the implantation window after controlled ovarian stimulation for oocyte donation. Fertil Steril. 2006;85:761–3.
Orvieto R, Badir W, Bar J, La Marca A, Ashkenazi J, Avrech OM, et al. Serum P-selectin level during controlled ovarian hyperstimulation–a preliminary report. Am J Reprod Immunol. 2004;52:139–42.
Kitaya K, Yasuo T. Aberrant expression of selectin E, CXCL1, and CXCL13 in chronic endometritis. Mod Pathol. 2010;23:1136–46.
Vigano P, Fusi F, Gaffuri B, Bonzi V, Ferrari A, Vignali M. Soluble intercellular adhesion molecule-1 in ovarian follicles: production by granulosa luteal cells and levels in follicular fluid. Fertil Steril. 1998;69:774–9.
Ratcliffe KE, Anthony FW, Richardson MC, Stones RW. Morphology and functional characteristics of human ovarian microvascular endothelium. Hum Reprod. 1999;14:1549–54.
Daniel Y, Geva E, Amit A, Eshed-Englender T, Bar-Am A, Lessing JB. Soluble endothelial and platelet selectins in serum and ascitic fluid of women with ovarian hyperstimulation syndrome. Am J Reprod Immunol. 2001;45:154–60.
Matsui S, Yasui T, Uemura H, Yamamoto S, Matsuzaki T, Tsuchiya N, et al. Induction of circulating monocyte chemoattractant protein-1 in women with gonadotropin-releasing hormone agonist. J Reprod Immunol. 2011;90:227–34.
Yasui T, Saijo A, Uemura H, Matsuzaki T, Tsuchiya N, Yuzurihara M, et al. Effects of oral and transdermal estrogen therapies on circulating cytokines and chemokines in postmenopausal women with hysterectomy. Eur J Endocrinol. 2009;161:267–73.
Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM, et al. The hormone resistin links obesity to diabetes. Nature. 2001;409:307–12.
Nagaev I, Bokarewa M, Tarkowski A, Smith U. Human resistin is a systemic immune-derived proinflammatory cytokine targeting both leukocytes and adipocytes. PLoS One. 2006;1:e31.
Comninos AN, Jayasena CN, Dhillo WS. The relationship between gut and adipose hormones, and reproduction. Hum Reprod Update. 2013;20:153–74.
Robertson SA, Rae CJ, Graham A. Induction of angiogenesis by murine resistin: putative role of PI3-kinase and NO-dependent pathways. Regul Pept. 2009;152:41–7.
Patel L, Buckels AC, Kinghorn IJ, Murdock PR, Holbrook JD, Plumpton C, et al. Resistin is expressed in human macrophages and directly regulated by PPAR gamma activators. Biochem Biophys Res Commun. 2003;300:472–6.
Munir I, Yen H-W, Baruth T, Tarkowski R, Azziz R, Magoffin DA, et al. Resistin stimulation of 17alpha-hydroxylase activity in ovarian theca cells in vitro: relevance to polycystic ovary syndrome. J Clin Endocrinol Metab. 2005;90:4852–7.
Maillard V, Froment P, Ramé C, Uzbekova S, Elis S, Dupont J. Expression and effect of resistin on bovine and rat granulosa cell steroidogenesis and proliferation. Reproduction. 2011;141:467–79.
Way JM, Gorgun CZ, Tong Q, Uysal KT, Brown KK, Harrington WW, et al. Adipose tissue resistin expression is severely suppressed in obesity and stimulated by peroxisome proliferator-activated receptor gamma agonists. J Biol Chem. 2001;276:25651–3.
Yamauchi T, Waki H, Kamon J, Murakami K, Motojima K, Komeda K, et al. Inhibition of RXR and PPARgamma ameliorates diet-induced obesity and type 2 diabetes. J Clin Invest. 2001;108:1001–13.
Panidis D, Koliakos G, Kourtis A, Farmakiotis D, Mouslech T, Rousso D. Serum resistin levels in women with polycystic ovary syndrome. Fertil Steril. 2004;81:361–6.
Yilmaz M, Bukan N, Demirci H, Ozturk C, Kan E, Ayvaz G, et al. Serum resistin and adiponectin levels in women with polycystic ovary syndrome. Gynecol Endocrinol. 2009;25:246–52.
Seow KM, Juan CC, Wu LY, Hsu YP, Yang WM, Tsai YL, et al. Serum and adipocyte resistin in polycystic ovary syndrome with insulin resistance. Hum Reprod. 2004;19:48–53.
Chen YC, Tsai EM, Chen HS, Liu YH, Lee CH, Chou FH, et al. Serum resistin level is a predictor of ovarian response in in vitro fertilisation cycle. Acta Obstet Gynecol Scand. 2007;86:963–7.
Seow KM, Juan CC, Hsu YP, Ho LT, Wang YY, Hwang JL. Serum and follicular resistin levels in women with polycystic ovarian syndrome during IVF-stimulated cycles. Hum Reprod. 2005;20:117–21.
Lu XE, Huang HF, Li MG, Zhu YM, Qiang YL, Dong MY. Resistin levels of serum and follicular fluid in non-obese patients with polycystic ovary syndrome during IVF cycles. J Zhejiang Univ Sci B. 2005;6:897–902.
Niles LP, Lobb DK, Kang NH, Armstrong KJ. Resistin expression in human granulosa cells. Endocrine. 2012;42:742–5.
Reverchon M, Cornuau M, Ramé C, Guerif F, Royère D, Dupont J. Resistin decreases insulin-like growth factor I-induced steroid production and insulin-like growth factor I receptor signaling in human granulosa cells. Fertil Steril. 2013;100:247–55.
Agarwal A, Gupta S, Sikka S. The role of free radicals and antioxidants in reproduction. Curr Opin Obstet Gynecol. 2006;18:325–32.
Riley J, Behrman H. Oxygen radicals and reactive oxygen species in reproduction. Proc Soc Exp Biol Med. 1991;198:781–91.
Askoxylaki M, Siristatidis C, Chrelias C, Vogiatzi P, Creatsa M, Salamalekis G, et al. Reactive oxygen species in the follicular fluid of subfertile women undergoing in vitro fertilization: a short narrative review. J Endocrinol Invest. 2013;36:1117–20.
Orvieto R. Controlled ovarian hyperstimulation—an inflammatory state. C J Soc Gynecol Investig. 2004;11:424–6.
Attaran M, Pasqualotto E, Falcone T, Goldberg JM, Miller KF, Agarwal A, et al. The effect of follicular reactive oxygen species on the outcome of in vitro fertilization. Int J Fertil. 2000;45:314–20.
Das R, Chattopadhyay R, Ghosh S, Goswami SK, Chakravarty BN, Chaudhury K. Reactive oxygen species level in follicular fluid-embryo quality marker in IVF. Human Reprod. 2006;21:2403–7.
Jana S, Babu NK, Chattopadhyay R, Chakravarty B, Chaudhury K. Upper control limit of reactive oxygen species in follicular fluid beyond which viable embryo formation is not favorable. Reprod Toxicol. 2010;29:447–51.
Wienerr-Megnazi Z, Vardi L, Lissak A, Shnizer S, Reznick AZ, Ishai D, et al. Oxidative stress indices in follicular fluid as measured by the thermochemiluminescence assay correlate with outcome parameters in in vitro fertilization. Fertil Steril. 2004;82:1171–6.
Elizur SE, Lebovitz O, Orvieto R, Dor J, Zan-Bar T. Reactive oxygen species in follicular fluid may serve as biochemical markers to determine ovarian aging and follicular metabolic age. Gynecol Endocrinol. 2014;30:705–7.
Al-Inany HG, Youssef MAFM, Aboulghar M, Broekmans FJ, Sterrenburg MD, Smit JG, et al. Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. Cochrane Database Syst Rev. 2011, 5: CD001750. doi: 10.1002/14651858.CD001750.pub3.
Higgins JPT, Green S. 2011. Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration. <http://handbook.cochrane.org/>. Accessed on December 31, 2014.
Carpintero NL, Suárez OA, Mangas CC, Varea CG, Rioja RG. Follicular steroid hormones as markers of oocyte quality and oocyte development potential. J Hum Reprod Sci. 2014;7:187–93.
Rosen MP, Shen S, Dobson AT, Rinaudo PF, McCulloch CE, Cedars MI. A quantitative assessment of follicle size on oocyte developmental competence. Fertil Steril. 2008;90:684–90.
Acknowledgments
The authors would like to thank the staff of the IVF Unit or the 3rd Department of Obstetrics and Gynecology of University of Athens.
Conflict of interest
All authors declare no conflict of interest.
Funding
There was no finding for the current work. The study is a part of the PhD thesis of the second author.
Authors’ roles
CS: Study design, interpretation of findings and manuscript drafting.
MA: Study design, data collection and interpretation of findings.
CV: Study design, data analysis and manuscript preparation.
DK: Data acquisition and interpretation of results.
CC: Data acquisition, data collection and interpretation of results.
All authors critically reviewed and approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule
This prospective cohort study showed that E-selectin, resistin and ROS levels are increasing during controlled ovarian hyperstimulation for IVF/ICSI, reaching their highest values at the day of oocyte recovery, with comparable values measured in the follicular fluid at that time. Resistin values >13.5 ng/ml are linked with a 6-fold increase on the odds of a pregnancy.
Rights and permissions
About this article
Cite this article
Siristatidis, C., Askoxylaki, M., Varounis, C. et al. E-selectin, resistin and reactive oxygen species levels in GnRH -agonist and -antagonist protocols in IVF/ICSI: a prospective cohort study. J Assist Reprod Genet 32, 959–967 (2015). https://doi.org/10.1007/s10815-015-0489-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-015-0489-z