Abstract
Purpose
To investigate the impacts that a paternal high fat diet (HFD) has on embryology, ovarian/cumulus cell gene expression and COC metabolism from female offspring, using a mouse model.
Methods
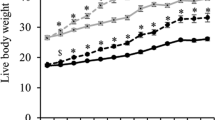
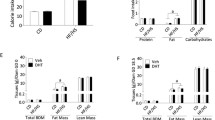
Founder male mice were either fed a control diet (CD) or a HFD for 12 weeks. The HFD induced obesity but not diabetes, and founder males were then mated to normal weight CD fed female mice. Female offspring were maintained on a CD, super-ovulated, mated and the resultant zygotes were cultured to the blastocyst stage for embryo morphology, blastocyst cell number and apoptosis assessment. Ovaries and cumulus cells from offspring were collected for gene expression analysis of selected genes that maintain chromatin remodeling and endoplasmic reticulum (ER), metabolic and inflammatory homeostasis. Cumulus/oocyte complexes were also investigated for glucose uptake and lipid accumulation.
Results
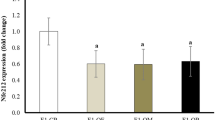
Female offspring sired by obese fathers produced embryos with delayed development and impaired quality, displayed increases in ovarian expression of Glut1, Glut3 and Glut4, and an increase in cumulus cell expression of Glut4. Interestingly their COCs did take up more glucose, but did accumulate more lipid.
Conclusions
A paternal HFD is associated with subfertility in female offspring despite the offspring being fed a CD and this subfertility is concomitant with ovarian/cumulus cell molecular alterations and increased lipid accumulation.





Similar content being viewed by others
References
Ogden CLC, Margaret D; Kit, Brian K.; Flegal, Katherine M. Prevalence of Obesity in the United States, 2009–2010: Centers for Diease Control and Prevention, Statistics NCfH. 2012.
Fejes I, Koloszar S, Szollosi J, Zavaczki Z, Pal A. Is semen quality affected by male body fat distribution? Andrologia. 2005;37(5):155–9.
Hammoud AO, Wilde N, Gibson M, Parks A, Carrell DT, Meikle AW. Male obesity and alteration in sperm parameters. Fertil Steril. 2008;90(6):2222–5.
Aggerholm AS, Thulstrup AM, Toft G, Ramlau-Hansen CH, Bonde JP. Is overweight a risk factor for reduced semen quality and altered serum sex hormone profile? Fertil Steril. 2008;90(3):619–26.
Chavarro JE, Toth TL, Wright DL, Meeker JD, Hauser R. Body mass index in relation to semen quality, sperm DNA integrity, and serum reproductive hormone levels among men attending an infertility clinic. Fertil Steril. 2010;93(7):2222–31.
Jensen TK, Andersson AM, Jorgensen N, Andersen AG, Carlsen E, Petersen JH, et al. Body mass index in relation to semen quality and reproductive hormones among 1,558 Danish men. Fertil Steril. 2004;82(4):863–70.
Kort HI, Massey JB, Elsner CW, Mitchell-Leef D, Shapiro DB, Witt MA, et al. Impact of body mass index values on sperm quantity and quality. J Androl. 2006;27(3):450–2.
Bakos HW, Henshaw RC, Mitchell M, Lane M. Paternal body mass index is associated with decreased blastocyst development and reduced live birth rates following assisted reproductive technology. Fertil Steril. 2011;95(5):1700–4.
Mitchell M, Bakos HW, Lane M. Paternal diet-induced obesity impairs embryo development and implantation in the mouse. Fertil Steril. 2011;95(4):1349–53.
Keltz J, Zapantis A, Jindal SK, Lieman HJ, Santoro N, Polotsky AJ. Overweight men: clinical pregnancy after ART is decreased in IVF but not in ICSI cycles. J Assist Reprod Genet. 2010;27(9–10):539–44.
Binder NK, Mitchell M, Gardner DK. Parental diet-induced obesity leads to retarded early mouse embryo development and altered carbohydrate utilisation by the blastocyst. Reprod Fertil Dev. 2012;24(6):804–12.
Binder NK, Hannan NJ, Gardner DK. Paternal diet-induced obesity retards early mouse embryo development, mitochondrial activity and pregnancy health. PLoS One. 2012;7(12):e52304.
Leibel NI, Baumann EE, Kocherginsky M, Rosenfield RL. Relationship of adolescent polycystic ovary syndrome to parental metabolic syndrome. J Clin Endocrinol Metab. 2006;91(4):1275–83.
Ng S-F, Lin RCY, Laybutt DR, Barres R, Owens JA, Morris MJ. Chronic high-fat diet in fathers programs [bgr]-cell dysfunction in female rat offspring. Nature. 2010;467(7318):963–6. doi:10.1038/nature09491.
Fullston T, Ohlsson Teague EM, Palmer NO, DeBlasio MJ, Mitchell M, Corbett M, et al. Paternal obesity initiates metabolic disturbances in two generations of mice with incomplete penetrance to the F2 generation and alters the transcriptional profile of testis and sperm microRNA content. FASEB J : Off Publ Fed Am Soc Exp Biol. 2013;27(10):4226–43 [Research Support, Non-U.S. Gov’t].
Fullston T, Palmer NO, Owens JA, Mitchell M, Bakos HW, Lane M. Diet-induced paternal obesity in the absence of diabetes diminishes the reproductive health of two subsequent generations of mice. Hum Reprod. 2012;27(5):1391–400 [Research Support, Non-U.S. Gov’t].
Minge CE, Bennett BD, Norman RJ, Robker RL. Peroxisome proliferator-activated receptor-gamma agonist rosiglitazone reverses the adverse effects of diet-induced obesity on oocyte quality. Endocrinology. 2008;149(5):2646–56.
Wu LL, Dunning KR, Yang X, Russell DL, Lane M, Norman RJ, et al. High-fat diet causes lipotoxicity responses in cumulus-oocyte complexes and decreased fertilization rates. Endocrinology. 2010;151(11):5438–45.
Alexander J, Chang GQ, Dourmashkin JT, Leibowitz SF. Distinct phenotypes of obesity-prone AKR/J, DBA2J and C57BL/6J mice compared to control strains. Int J Obes (Lond). 2006;30(1):50–9.
Council NHaMR. Australian Code for the Care and Use of Animals for Scientific Purposes. 2013;8th Edition.
WHO. WHO laboratory manual for the examination and processing of human semen. Fifth Edition ed. 2010.
Nasr-Esfahani MH, Aitken JR, Johnson MH. Hydrogen peroxide levels in mouse oocytes and early cleavage stage embryos developed in vitro or in vivo. Development. 1990;109(2):501–7.
Lane M, Maybach JM, Gardner DK. Addition of ascorbate during cryopreservation stimulates subsequent embryo development. Hum Reprod. 2002;17(10):2686–93.
Gardner DK, Lane M, Watson AJ. In: Gardner DK, Lane M, Watson AJ, editors. A laboratory guide to the mammalian embryo. New York: Oxford University Press; 2004.
Lane M, Gardner DK. Effect of incubation volume and embryo density on the development and viability of mouse embryos in vitro. Hum Reprod. 1992;7(4):558–62 [Research Support, Non-U.S. Gov’t].
Gardner DK, Lane MW, Lane M. EDTA stimulates cleavage stage bovine embryo development in culture but inhibits blastocyst development and differentiation. Mol Reprod Dev. 2000;57(3):256–61.
Zander DL, Thompson JG, Lane M. Perturbations in mouse embryo development and viability caused by ammonium are more severe after exposure at the cleavage stages. Biol Reprod. 2006;74(2):288–94.
Kamjoo M, Brison DR, Kimber SJ. Apoptosis in the preimplantation mouse embryo: effect of strain difference and in vitro culture. Mol Reprod Dev. 2002;61(1):67–77.
Goossens K, van Poucke M, van Soom A, Vandesompele J, van Zeveren A, Peelman LJ. Selection of reference genes for quantitative real-time PCR in bovine preimplantation embryos. BMC Dev Biol. 2005;5:27.
Robert C, McGraw S, Massicotte L, Pravetoni M, Gandolfi F, Sirard MA. Quantification of housekeeping transcript levels during the development of bovine preimplantation embryos. Biol Reprod. 2002;67(5):1465–72 [Research Support, Non-U.S. Gov’t].
Lane M, Gardner DK. Ammonium induces aberrant blastocyst differentiation, metabolism, pH regulation, gene expression and subsequently alters fetal development in the mouse. Biol Reprod. 2003;69(4):1109–17 [Research Support, Non-U.S. Gov’t].
McPherson NO, Bakos HW, Owens JA, Setchell BP, Lane M. Improving metabolic health in obese male mice via diet and exercise restores embryo development and fetal growth. PLoS One. 2013;8(8):e71459 [Research Support, Non-U.S. Gov’t].
van den Berg SW, Dolle ME, Imholz S, van der AD, van ’t Slot R, Wijmenga C, et al. Genetic variations in regulatory pathways of fatty acid and glucose metabolism are associated with obesity phenotypes: a population-based cohort study. Int J Obes (Lond). [Research Support, Non-U.S. Gov’t]. 2009 Oct;33(10):1143–52.
Robker RL, Akison LK, Bennett BD, Thrupp PN, Chura LR, Russell DL, et al. Obese women exhibit differences in ovarian metabolites, hormones, and gene expression compared with moderate-weight women. J Clin Endocrinol Metab. 2009;94(5):1533–40.
Boyle KE, Newsom SA, Janssen RC, Lappas M, Friedman JE. Skeletal muscle MnSOD, mitochondrial complex II, and SIRT3 enzyme activities are decreased in maternal obesity during human pregnancy and gestational diabetes mellitus. J Clin Endocrinol Metab. 2013;98(10):E1601–9 [Research Support, N.I.H., Extramural].
Bakos HW, Mitchell M, Setchell BP, Lane M. The effect of paternal diet-induced obesity on sperm function and fertilization in a mouse model. Int J Androl. 2010 Jul 23.
Palmer NO, Bakos HW, Owens JA, Setchell BP, Lane M. Diet and exercise in an obese mouse fed a high-fat diet improve metabolic health and reverse perturbed sperm function. Am J Physiol Endocrinol Metab. 2012;302(7):E768–80 [In Vitro Research Support, Non-U.S. Gov’t].
Palmer NO, Fullston T, Mitchell M, Setchell BP, Lane M. SIRT6 in mouse spermatogenesis is modulated by diet-induced obesity. Reprod Fertil Dev. 2011;23(7):929–39 [Research Support, Non-U.S. Gov’t].
Mitchell M, Cashman KS, Gardner DK, Thompson JG, Lane M. Disruption of mitochondrial malate-aspartate shuttle activity in mouse blastocysts impairs viability and fetal growth. Biol Reprod. 2009;80(2):295–301 [Research Support, Non-U.S. Gov’t].
Lane M, Gardner DK. Differential regulation of mouse embryo development and viability by amino acids. J Reprod Fertil. 1997;109(1):153–64.
Kwong WY, Wild AE, Roberts P, Willis AC, Fleming TP. Maternal undernutrition during the preimplantation period of rat development causes blastocyst abnormalities and programming of postnatal hypertension. Development. 2000;127(19):4195–202 [Research Support, Non-U.S. Gov’t].
Ghanayem BI, Bai R, Kissling GE, Travlos G, Hoffler U. Diet-induced obesity in male mice is associated with reduced fertility and potentiation of acrylamide-induced reproductive toxicity. Biol Reprod. 2010;82(1):96–104.
Wood IS, Trayhurn P. Glucose transporters (GLUT and SGLT): expanded families of sugar transport proteins. Br J Nutr. 2003;89(1):3–9 [Review].
Garvey WT, Maianu L, Huecksteadt TP, Birnbaum MJ, Molina JM, Ciaraldi TP. Pretranslational suppression of a glucose transporter protein causes insulin resistance in adipocytes from patients with non-insulin-dependent diabetes mellitus and obesity. J Clin Invest. 1991;87(3):1072–81 [Research Support, U.S. Gov’t, Non-P.H.S. Research Support, U.S. Gov’t, P.H.S.].
Sivitz WI, DeSautel SL, Kayano T, Bell GI, Pessin JE. Regulation of glucose transporter messenger RNA in insulin-deficient states. Nature. 1989;340(6228):72–4 [Research Support, Non-U.S. Gov’t Research Support, U.S. Gov’t, P.H.S.].
Berger J, Biswas C, Vicario PP, Strout HV, Saperstein R, Pilch PF. Decreased expression of the insulin-responsive glucose transporter in diabetes and fasting. Nature. 1989;340(6228):70–2 [Research Support, Non-U.S. Gov’t Research Support, U.S. Gov’t, P.H.S.].
Colton SA, Pieper GM, Downs SM. Altered meiotic regulation in oocytes from diabetic mice. Biol Reprod. 2002;67(1):220–31 [Research Support, U.S. Gov’t, P.H.S.].
Wang Q, Ratchford AM, Chi MM, Schoeller E, Frolova A, Schedl T, et al. Maternal diabetes causes mitochondrial dysfunction and meiotic defects in murine oocytes. Mol Endocrinol. 2009;23(10):1603–12 [Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t].
Purcell SH, Moley KH. The impact of obesity on egg quality. J Assist Reprod Genet. 2011;28(6):517–24 [Review].
Adastra KL, Chi MM, Riley JK, Moley KH. A differential autophagic response to hyperglycemia in the developing murine embryo. Reproduction. 2011;141(5):607–15 [Research Support, N.I.H., Extramural].
Wang Q, Chi MM, Schedl T, Moley KH. An intercellular pathway for glucose transport into mouse oocytes. Am J Physiol Endocrinol Metab. 2012;302(12):E1511–8 [Research Support, N.I.H., Extramural].
Roberts R, Stark J, Iatropoulou A, Becker DL, Franks S, Hardy K. Energy substrate metabolism of mouse cumulus-oocyte complexes: response to follicle-stimulating hormone is mediated by the phosphatidylinositol 3-kinase pathway and is associated with oocyte maturation. Biol Reprod. 2004;71(1):199–209 [Research Support, Non-U.S. Gov’t].
Phy JL, Conover CA, Abbott DH, Zschunke MA, Walker DL, Session DR, et al. Insulin and messenger ribonucleic acid expression of insulin receptor isoforms in ovarian follicles from nonhirsute ovulatory women and polycystic ovary syndrome patients. J Clin Endocrinol Metab. 2004;89(7):3561–6 [Research Support, Non-U.S. Gov’t Research Support, U.S. Gov’t, P.H.S.].
Pantasri T, Norman RJ. The effects of being overweight and obese on female reproduction: a review. Gynecol Endocrinol. 2013 Nov 4.
Ekart J, McNatty K, Hutton J, Pitman J. Ranking and selection of MII oocytes in human ICSI cycles using gene expression levels from associated cumulus cells. Hum Reprod. 2013;28(11):2930–42.
Youngson NA, Whitelaw E. The effects of acquired paternal obesity on the next generation. Asian J Androl. 2011;13(2):195–6.
Soubry A, Murphy SK, Wang F, Huang Z, Vidal AC, Fuemmeler BF, et al. Newborns of obese parents have altered DNA methylation patterns at imprinted genes. Int J Obes (Lond). 2013 Oct 25.
Palmer NO, Bakos HW, Fullston T, Lane M. Impact of obesity on male fertility, sperm function and molecular composition. Spermatogenesis. 2012;2(4):253–63.
Sutton-McDowall ML, Gilchrist RB, Thompson JG. The pivotal role of glucose metabolism in determining oocyte developmental competence. Reproduction. 2010;139(4):685–95.
Acknowledgments
The authors thank Vitrolife for their donation of all culture media used in this study.
This work was supported by the Australian National Health and Medical Research Council project grant awarded to ML. ML is a recipient of a NHMRC SRF and TF/NOM are recipients of NHMRC ECFs.
Conflict of interest
T.F., H.S., L.Y.S., W.X.K., L.L.W., R.L.R., N.O.M. and M.L. declare that they have nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Although is has been previously demonstrated that paternal obesity can impede the fertility of female offspring, the development of embryos and the molecular charecterisation of reproductive material had not been investigated. Here we show that as a result of paternal obesity female offspring produce embryos with impaired development/quality, have alterations to the epxression of glucose tranpsorter genes in ovaries/cumulus cells and increased lipid uptake into cumulus-oocyte complexes.
Tod Fullston and Helana Shehadeh contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig. 1
(DOC 160 kb)
Supplemental Table 1
(DOCX 42 kb)
Supplemental Table 2
(DOCX 39 kb)
Supplemental Table 3
(DOCX 39 kb)
Rights and permissions
About this article
Cite this article
Fullston, T., Shehadeh, H., Sandeman, L.Y. et al. Female offspring sired by diet induced obese male mice display impaired blastocyst development with molecular alterations to their ovaries, oocytes and cumulus cells. J Assist Reprod Genet 32, 725–735 (2015). https://doi.org/10.1007/s10815-015-0470-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-015-0470-x