Abstract
Purpose
The aim of this study was to use digital holographic microscopy (DHM) in human sperm imaging and compare quantitative phase contrast of sperm heads in normozoospermia (NZ) and oligoasthenozoospermia (OAT).
Methods
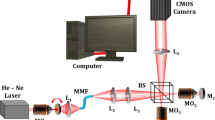
DHM spermatozoa imaging and repeated quantitative phase shift evaluation were used. Five NZ and 5 OAT samples were examined. Semen samples were examined by semen analysis and processed for DHM. Main outcome measures were maximum phase shift value of the sperm heads. Differences of the phase shift and in NZ and OAT samples were statistically tested.
Results
In NZ samples median phase shifts were in the range 2.72–3.21 rad and 2.00–2.15 in OAT samples. Differences among individual samples were statistically significant (p < 0.001) in both groups. Median phase shift according to sperm count was 2.90 rad in NZ samples and 2.00 rad in OAT samples. This difference was statistically significant (p < 0.001).
Conclusion
Quantitative evaluation of the phase shift by DHM could provide new information on the exact structure and composition of the sperm head. At present, this technique is not established for clinical utility.

Similar content being viewed by others
References
Marquet P, Rappaz B, Magistretti PJ, Cuche E, Emery Y, Colomb T, et al. Digital holographic microscopy: a noninvasive contrast imaging technique allowing quantitative visualization of living cells with subwavelength axial accuracy. Opt Lett. 2005;30:468–70.
Vesely P, Blase C, Matouskova E, Bereiter-Hahn J. Arising podosomal structures are associated with neoplastic cell morphological phenotype induced by the microenvironment. Anticancer Res. 2006;26:967–72.
Depeursinge C, Colomb T, Emery Y, Kuhn J, Charriere F, Rappaz B, et al. Digital holographic microscopy applied to life sciences. Conf Proc IEEE Eng Med Biol Soc. 2007;2007:6244–7.
Janeckova H, Vesely P, Chmelik R. Proving tumour cells by acute nutritional/energy deprivation as a survival threat: a task for microscopy. Anticancer Res. 2009;29:2339–45.
Kolman P, Chmelik R. Coherence-controlled holographic microscope. Opt Express. 2010;18:21990–2003.
Chmelik R, Harna Z. Parallel-mode confocal microscope. Opt Eng. 1999;38:1635–9.
Chmelik R. Three-dimensional scalar imaging in high-aperture low-coherence interference and holographic microscopes. J Mod Opt. 2006;53:2673–89.
Dubois F, Joannes L, Legros JC. Improved three-dimensional imaging with a digital holography microscope with a source of partial spatial coherence. Appl Opt. 1999;38:7085–94.
Janeckova H, Kolman P, Vesely P, Chmelik R. Digital holographic microscope with low-spatial and temporal coherence of illumination. In: Optical and Digital Image Processing. Proc. of SPIE 2008; 7000:1–8.
World Health Organization Laboratory Manual for the Examination of Human Semen and Sperm-Cervical Mucus Interaction. 4th ed, 1999. Cambridge University Press.
Crha I, Kralikova M, Melounova J, Ventruba P, Zakova J, Beharka R, et al. Seminal plasma homocysteine, folate and cobalamin in men with obstructive and non-obstructive azoospermia. J Assist Reprod Genet. 2010;27:533–8.
Oliva R. Protamines and male infertility. Hum Reprod Update. 2006;12:417–35.
Lefièvre L, Bedu-Addo K, Conner SJ, Machado-Oliveira GS, Chen Y, Kirkman-Brown JC, et al. Counting sperm does not add up any more: time for a new equation? Reproduction. 2007;133:675–84.
Zini A, Phillips S, Courchesne A, Boman JM, Baazeem A, Bissonnette F, et al. Sperm head morphology is related to high deoxyribonucleic acid stainability assessed by sperm chromatin structure assay. Fertil Steril. 2009;91:2495–500.
Mudrak O, Tomilin N, Zalensky A. Chromosome architecture in the decondensing human sperm nucleus. J Cell Sci. 2005;118:4541–50.
Ward WS. Function of sperm chromatin structural elements in fertilization and development. Mol Hum Reprod. 2010;16:30–6.
Moskovtsev SI, Willis J, Azad A, Mullen JB. Sperm DNA integrity: correlation with sperm plasma membrane integrity in semen evaluated for male infertility. Arch Androl. 2005;51:33–40.
Akgul M, Ozkinay F, Ercal D, Cogulu O, Dogan O, Altay B, et al. Cytogenetic abnormalities in 179 cases with male infertility in Western Region of Turkey: Report and review. J Assist Reprod Genet. 2009;26:119–22.
Winkle T, Rosenbusch B, Gagsteiger F, Paiss T, Zoller N. The correlation between male age, sperm quality and sperm DNA fragmentation in 320 men attending a fertility center. J Assist Reprod Genet. 2009;26:41–6.
García-Herrero S, Garrido N, Martínez-Conejero JA, Remohí J, Pellicer A, Meseguer M. Ontological evaluation of transcriptional differences between sperm of infertile males and fertile donors using microarray analysis. J Assist Reprod Genet. 2010;27:111–20.
Gianaroli L, Magli MC, Ferraretti AP, Crippa A, Lappi M, Capitani S, et al. Birefringence characteristics in sperm heads allow for the selection of reacted spermatozoa for intracytoplasmic sperm injection. Fertil Steril. 2010;93:807–13.
Petersen CG, Vagnini LD, Mauri AL, Massaro FC, Cavagna M, Baruffi RL, Oliveira JB, Franco JG Jr. Relationship between DNA damage and sperm head birefringence. Reprod Biomed Online. 2011 Mar 21. [Epub ahead of print] doi:10.1016/j.rbmo.2011.03.017
Hammadeh ME, Bernardi A, Zeginiadou T, Amer A, Schmidt W. Relationship between nuclear chromatin decondensation (NCD) in vitro and other sperm parameters and their predictive value on fertilization rate in IVF program. J Assist Reprod Genet. 2005;22:301–5.
Kolle S, Reese S, Kummer W. New aspects of gamete transport, fertilization, and embryonic development in the oviduct gained by means of live cell imaging. Theriogenology. 2010;73:786–95.
Ierardi V, Niccolini A, Alderighi M, Gazzano A, Martelli F, Solaro R. AFM characterization of rabbit spermatozoa. Microsc Res Tech. 2008;71:529–35.
Acknowledgements
Authors wish to thank Hana Uhlirova (born Janeckova), M.Sc., Ph.D. from the Institute of Physical Engineering, Faculty of Mechanical Engineering, Brno University of Technology for her kind and skillful technical assistance in DHM.
This work was supported by Internal Grant Agency (IGA) of the Ministry of Health of the Czech Republic-No. NS/9661-4.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule
Quantitative evaluation of the phase shift by digital holographic microscopy could provide new information on the exact structure and composition of the sperm head.
Rights and permissions
About this article
Cite this article
Crha, I., Zakova, J., Huser, M. et al. Digital holographic microscopy in human sperm imaging. J Assist Reprod Genet 28, 725–729 (2011). https://doi.org/10.1007/s10815-011-9584-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-011-9584-y