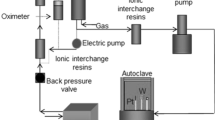
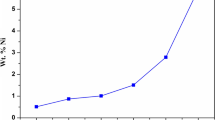
Abstract
A novel cyanide-free zinc deposition bath was developed in which sorbitol was added at various concentrations. Voltammetric studies indicated that the reduction process is influenced thermodynamically and kinetically by the sorbitol concentration. Also, two cathodic processes were observed, one (wave) associated with the hydrogen evolution reaction (HER) on 1010 steel, the other (peak) with zinc bulk reduction, simultaneous to the HER. Furthermore, the plating-process kinetics was controlled by mass transport. The presence of sorbitol in the bath led to formation of light-grey zinc films, even during the HER, without cracks and dendrites. Plating current efficiency decreased from ∼ ∼62% to 43% on increasing the sorbitol concentration in the plating bath. In the presence of 0.1 M \([\hbox{Zn(OH)}_{4}]^{2-}\) and/or sorbitol concentrations higher than 0.2 M, Zn electrode dissolution was inhibited. However, a small dissolution of zinc electrode was observed with 0.05 M sorbitol in alkaline solution without zincate. SEM micrographs showed that the 1010 steel substrate was fully covered by Zn film and that the sorbitol affected the morphology of zinc films, acting as a grain refiner.
Similar content being viewed by others
References
F.A. Lowenheim (1974) Modern Electroplating. John Wiley & Sons, New York, pp. 436
M. Pushpavanam (2000) Bull. Electrochem. 16:559–566
N.V. Partasaradhy (1989) Practical Electroplating. Prentice Hall, New Jersey
H.S. (1977) Schneider Plating Surf. Finish. 64: 52
Bapu G.N.K.R., Devaraj G., Ayyapparaj J. (1998) J. Solid State Electrochem. 3: 48
Ramachandran A., Mayanna S.M. (1992) Met Finish. 90: 61
Cachet C., Stroder U., Wiart R. (1982) Electrochem. Acta. 27: 903
Diggle J.W., Damjanovic A. (1972) J. Electrochem. Soc. 119: 1649
Blinov V.M., Kuprit A.V., Gnedenkov L.Yu., Loshkarev Yu.M., Trofimenko V.V. (1988) Élektrokhimiya 24: 461
Blinov V.M., Burov L.M., Trofimenko V.V., Gnedenkov L.Yu., and Loshkarev Yu.M. (1989) Élektrokhimiya. 25: 930
Litovka G.P., Loshkarev Yu.M., Trofimenko V.V., Kazarinov V.E., Grigorev N.B. (1979) Élektrokhimiya 15: 1229
Ravindran V., Muralidharan V.S. (2000) Bull. Electrochem. 16: 60
Nagarajan K.S., Rajendran S., Vembu K., Rengarajan S. (1999) Bull. Electrochem. 15: 199
Naik Y.A., Venkatesha T.V. (2003) Indian Journal of Engineering and Materials Sciences. 10: 318
Zuniga V., Ortega R., Meas Y., Trejo G. (2004) Plat Surf. Finish. 91: 46
M. S. Pereira, Estudo da influência do sorbitol no processo de deposição de zinco: caracterização eletroanalítica e espectrofotométrica do banho de deposição e morfologia dos de zinco, Dissertation (Universidade Federal de São Carlos, São Carlos, 2003).
Galvani F., Carlos I.A. (1997) Metal Finish. 2: 70
L.L. Barbosa, G.M. Oliveira and I.A. Carlos, submitted to Surf. Coat. Technol. 2005
Carlos I.A., de Almeida M.R.H. (2004) J. Electroanal. Chem. 562: 153
Barbosa L.L., de Almeida M.R.H., Carlos R.M., Ionashiro M., Oliveira G.M., Carlos I.A. (2005) Surf. Coat. Technol. 192: 145
Fletcher S., Halliday C.S., Gates D., Wescott M., Lwin T., Nelson G. (1983) J. Electroanal. Chem.159: 267
P. C. Túlio, Estudo dos mecanismos de eletrodeposição de prata sobre platina a partir de soluções de nitrato de prata, Dissertation (Universidade Federal de São Carlos, São Carlos 1996).
T.M.C. Nogueira, PhD Thesis, (Universidade Federal de São Carlos, São Carlos 1996).
Acknowledgements
Financial support from the Brazilian research foundations: Capes and FAPESP (Proc. no 01/13740-5) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pereira, M.S., Barbosa, L.L., Souza, C.A.C. et al. The influence of sorbitol on zinc film deposition, zinc dissolution process and morphology of deposits obtained from alkaline bath. J Appl Electrochem 36, 727–732 (2006). https://doi.org/10.1007/s10800-006-9133-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-006-9133-z