Abstract
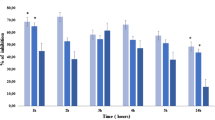
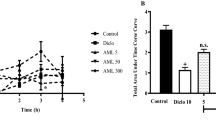
The aqueous and organic extracts of Acacia visco Lor. Ap Griseb (Fabaceae) were tested for anti-inflammatory activity in experimental models in rat. Besides, the free-radical scavenging capacity of extracts from A. visco was determined. The extracts revealed anti-inflammatory effect against carrageenan-induced oedema, phospholipase A2-induced oedema, cotton pellet-induced granuloma and they did not show acute toxic effect. Among the class of compounds characterized from A. visco leaves, the triterpenoid 20(29)-lupen-3β-ol (lupeol), 12-ursen-3β-ol (α-amyrin) and 12-oleanen-3β-ol (β-amyrin) may be mainly responsible for the pharmacological activities.





Similar content being viewed by others
Abbreviations
- Phospholipase A2 :
-
PLA2
- AvInL:
-
Acacia visco leaves infusion
- AvInB:
-
Acacia visco bark infusion
- AvMeL:
-
Acacia visco leaves methanolic extract
- AvMeB:
-
Acacia visco bark methanolic extract
- AvHeL:
-
Acacia visco leaves hexane extract
- AvChL:
-
Acacia visco leaves chloroform extract
- DPPH:
-
1,1-Diphenyl-2-picrylhydrazyl
- IC50 :
-
Inhibitory concentration 50
References
Agarwal RB, Rangari VD (2003) Antiinflammatory and arthritic activities of lupeol and 19 alpha-H-lupeol isolated from Strobilanthus callosus and Strobilanthus ixiocephala roots. Indian J Pharmacol 35:384–387
Backhouse N, Delporte C, Givernau M et al (1994) Anti-inflammatory and antipyretic effects of boldine. Agents Actions. 42:117–144
Bailey PJ, Sturm A, Lopez-Ramos B (1982) A biochemical study of the cotton pellet granuloma in the rat. Effects of dexamethasone and indomethacin. Biochem Pharmacol 31:121–128
Billingham MEJ (1983) Models of arthritis and the search for antirheumatic drugs. Pharmacol Ther 21:389
Budzikiewicz H, Djerassi C, Williams DH (1964) Structure elucidation of natural products by mass spectrometry, vol Vol II. Holden Day, San Francisco, p 203
Calhoun WJ, Yu J, Sung A et al (1989) Pharmacological modulation of D-49 phospholipase A2-induced paw edema in the mouse. Agents Actions 27:418
Chang ST, Wu JH, Wang SY et al (2001) Antioxidant activity of extracts from Acacia confusa bark and heartwood. J Agric Food Chem 49:3420
Cotelle N, Bernier JL, Catteau JP et al (1996) Antioxidant properties of hydroxy-flavones. Free Radic Biol Med 20:35–43
Dafallah AA, Al-Mustafa Z (1996) Investigation of the anti-inflammatory activity of Acacia nilotica and Hibiscus sabdariffa. Am J Chin Med 24(3–4):263
Di Rosa M, Giround JP, Willougbby DA (1971) Studies of the mediators of the acute inflammatory response induced in rats in different sites by carrageenan and turpentine. J Pathol 104:15–29
Dongmo AB, Nguelefack T, Lacaille-Dubois MA (2005) Antinociceptive and anti-inflammatory activities of Acacia pennata wild (Mimosaceae). J Ethnopharmacol 98:201–206
Ellis JS, Chain BM, Coke A et al (1992) Adjuvant composition determines the induction of type II collagen-induced arthritis. Scand J Immunol 36:49–56
Escrig V, Ubeda A, Ferrandiz ML et al (1997) Variabilin: a dual inhibitor of human secretory and cytosolic phospholipase A2 with anti-inflammatory activity. J Pharmacol Exp Ther 282(1):123–131
Farmacopea Nacional Argentina, 6th edn (1978), pp 581
Garai S, Mahato SB (1997) Isolation and structure elucidation of three triterpenoid saponins from Acacia auriculiformis. Phytochemistry 44(1):137–140
Geetha T, Varalakshmi P, Mary LR (1998) Effect of triterpenes from Crataeva nurvata stem bark on lipid peroxidation in adjuvant induced arthritis in rats. Pharmacol Res 37(3):191–195
Hamilton TA, Adams DO (1987) Molecular mechanism of signal transduction in macrophages. Inmunol Today 8:151
Haridas V, Arutzen CJ, Gutterman JU (2001) Avicins, a family of triterpenoid saponins from Acacia victoriae (Bentham), inhibit activation of nuclear factor-kappaB by inhibiting both its nuclear localization and ability to bind DNA. Proc Natl Acad Sci USA 98(20):11557
Hwang SB, Lam MH, Li CL et al (1986) Release of platelet activating factor and its involvement in the first phase of carrageenin-induced rat foot edema. Eur J Pharmacol 120(1):33–41
Kumpulainen JT, Salonen JT (1999) Natural antioxidants and anticarcinogens. In: Nutrition, health and disease. The Royal Society of Chemistry, UK, pp 178
Li RW, Myers SP, Leach DN et al (2003) A cross-cultural study anti-inflammatory activity of Australian and Chinese plants. J Ethnopharmacol 85(1):25–32
Meier R, Schuler W, Desaulles P (1950) Zur frage dex mechanismus der Hemmung des Bindegewebswachstums durch Cortisone. Experientia 6(12):468
Mujoo K, Haridas V, Hoffmann JJ et al (2001) Triterpenoid saponins from Acacia victorae (Bentham) decrease tumor cell proliferation and induce apoptosis. Cancer Res 61(14):5486
Mustafa FB, Ng FS, Nguyen TH et al (2008) Honeybee venom secretory phospholipase A2 induces leukotriene production but not histamine release from human basophils. Clin Exp Immunol 151(1):94–100
Nguemfo EL, Dimo T, Dongmo AB et al (2008) Anti-oxidative and anti-inflammatory activities of some isolated constituents from the stem bark of Allanblackia monticola Staner L.C. (Guttiferae). Inflammopharmacol 17:37–41
OECD Guidelines (2001) OECD guidelines for testing of chemicals. Gce No 423. CTA class method. URL:http://www.oecd.org
Ogunkoya L (1981) Application of mass spectrometry in structural problems in triterpenes. Phytochemistry 20:121–126
Olajide OA, Makinde JM, Okpako DT (2003) Evaluation of the anti-inflammatory property of the extract of Combretum micranthum G. Don (Combretaceae). Inflammopharmacol 11:293–298
Parvataneni R, Pragada RR, Jorige A et al (2005) Anti-inflammatory activity of a new sphingosine derivative and cembrenoid diterpene (Lobohedleolide) isolated from marine soft corals of Sinularia crassa TIXIER-DURIVAULT and Lobophytum species of the Andaman and Nicobar Islands. Biol Pharm Bull 28:1311–1313
Ratsch C (2005) The encyclopedia of psychoactive plants ethnopharmacology and its applications. Inner Traditions, 51 p
Saleem A, Ahotupa M, Pihlaja KZ (2001) Total phenolics concentration and antioxidant potential of extracts of medicinal plants of Pakistan. Naturforschung 56(11–12):973–978
Sarkar D, Dutta A, Das M et al (2005) Effect of Aloe vera on nitric oxide production by macrophages during inflammation. Indian J Pharmacol 36(60):371–375
Searle PA, Molinski TF (1995) 4-Dehydroecdysterone, a new ecdysteroid from the zoanthid Parazoanthus sp. J Nat Prod 58(2):264–268
Theisen-Popp P, Müller-Peddinghaus R (1994) Antirheumatic drug profiles evaluated in adjuvant rats by multiparameter analysis. Agents Actions 42:50–55
Uniyal SK, Badoni V, Sati OP (1992) A new triterpenoidal saponin from Acacia auriculiformis. J Nat Prod 55(4):500–502
Vinegar R, Truax JF, Selph JL, Vowelker FA (1982) Pathway of onset development and decay of carrageenan pleurisy in the rat. Fed. Proc. 41:2588–2595
Winter CA, Risley EA, Nuss GW (1962) Carrageenan-induced edema in hind paw of the rat as an assay for antiinflammatory drugs. Proc Soc Exp Biol Med 111:544–547
Acknowledgments
This work was supported by National University of San Luis, Science and Technique (CYT), Project No. 22/Q604. The authors thank Dr. Carlos E. Ardanaz for his assistance in the discussion of mass spectral data and valuable comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pedernera, A.M., Guardia, T., Guardia Calderón, C.E. et al. Anti-inflammatory effect of Acacia visco extracts in animal models. Inflammopharmacol 18, 253–260 (2010). https://doi.org/10.1007/s10787-009-0028-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-009-0028-6