Abstract
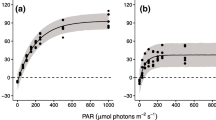
In winter of 2009/2010, Aphanizomenon flos-aquae bloomed in the ice and snow covered oligo-mesotrophic Lake Stechlin, Germany. The photosynthesis of the natural population was measured at eight temperatures in the range of 2–35°C, at nine different irradiance levels in the range of 0–1,320 μmol m−2 s−1 PAR at each applied temperature. The photoadaptation parameter (I k) and the maximum photosynthetic rate (P max) correlated positively with the temperature between 2 and 30°C, and there was a remarkable drop in both parameters at 35°C. The low I k at low temperatures enabled the active photosynthesis of overwintering populations at low irradiance levels under ice and snow cover. The optimum of the photosynthesis was above 20°C at irradiances above 150 μmol m−2 s−1. At lower irradiance levels (7.5–30 μmol m−2 s−1), the photosynthesis was the most intensive in the temperature range of 2–5°C. The interaction between light and temperature allowed the proliferation of A. flos-aquae in Lake Stechlin resulting in winter water bloom in this oligo-mesotrophic lake. The applied 2°C is the lowest experimental temperature ever in the photosynthesis/growth studies of A. flos-aquae, and the results of the P–I and P–T measurements provide novel information about the tolerance and physiological plasticity of this species.




Similar content being viewed by others
References
Ahlgren, G., 1987. Temperature functions in biology and their application to algal growth constants. Oikos 49: 177–190.
APHA – American Public Health Association, 1998. Standard Methods for the Examination of Water and Wastewater, 20th ed. United Book Press, Inc., Baltimore, MD.
Bissinger, J. E., D. J. S. Montagnes, J. Sharples & D. Atkinson, 2008. Predicting marine phytoplankton maximum growth rates from temperature: improving on the Eppley curve using quantile regression. Limnology and Oceanography 53: 487–493.
Butterwick, C., S. I. Heaney & J. F. Talling, 2005. Diversity in the influence of temperature on the growth rates of freshwater algae, and its ecological relevance. Freshwater Biology 50: 291–300.
De Nobel, W. T., C. P. H. Matthijs, E. von Elert & L. R. Mur, 1998. Comparison of the light-limited growth of the nitrogen-fixing cyanobacteria Anabaena and Aphanizomenon. New Phytologist 138: 579–587.
Dokulil, M. T. & K. Teubner, 2000. Cyanobacterial dominance in lakes. Hydrobiologia 438: 1–12.
Gervais, F., J. Padisák & R. Koschel, 1997. Do light quality and low nutrient concentration favour picocyanobacteria below the thermocline of the oligotrophic Lake Stechlin? Journal of Plankton Research 19: 771–781.
Huisman, J., R. R. Jonker, C. Zonneveld & F. J. Weissing, 1999. Competition for light between phytoplankton species: experimental tests of mechanistic theory. Ecology 80: 211–222.
Jones, R. I., 1979. Notes on the growth and sporulation of a natural population of Aphanizomenon flos-aquae. Hydrobiologia 62: 55–58.
Kirk, J. T. O., 1994. Light and Photosynthesis in Aquatic Ecosystems, 2nd ed. Cambridge University Press, Cambridge.
Konopka, A. & T. D. Brock, 1978. Effect of temperature on blue-green algae (cyanobacteria) in Lake Mendota. Applied and Environmental Microbiology 36: 572–576.
Krause, G. H., 1994. Photoinhibition induced by low temperatures. In Baker, N. R. & J. R. Bower (eds), Photoinhibition of Photosynthesis from Molecular Mechanisms to the Field. Bios Scientific Publishers, Oxford: 331–348.
Lazarova, D., A. Popova, D. Stanoeva & M. Velitchkova, 2009. Involvement of reactive oxygen radicals in photoinhibition of primary photosynthetic reactions—effect of temperature and oxygen radical scavengers. Biotechnology & Biotechnological Equipment 23: 511–515.
Lei, R., M. Leppäranta, A. Erm, E. Jaatinen & O. Pärn, 2011. Field investigations of apparent optical properties of ice cover in Finnish and Estonian lakes in winter 2009. Estonian Journal of Earth Sciences 60: 50–64.
Mehnert, G., F. Leunert, S. Cirés, K. D. Jöhnk, J. Rücker, B. Nixdorf & C. Wiedner, 2010. Competitiveness of invasive and native cyanobacteria from temperate freshwaters under various light and temperature conditions. Journal of Plankton Research 32: 1009–1021.
Padisák, J., 1997. Cylindrospermopsis raciborskii (Woloszynska) Seenayya et Subba Raju, an expanding highly adaptive bluegreen algal species: worldwide distribution and review of its ecology. Archiv für Hydrobiologie Supplement 107: 563–593.
Padisák, J., É. Hajnal, L. Krienitz, J. Lakner & V. Üveges, 2010. Rarity, ecological memory, rate of floral change in phytoplankton—and the mystery of the Red Cock. Hydrobiologia 653: 45–64.
Paerl, H. W. & J. Huisman, 2008. Blooms like it hot. Science 320: 57–58.
Platt, T., C. L. Gallegos & W. G. Harrison, 1980. Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton. Journal of Marine Research 38: 687–701.
Preußel, K., G. Wessel, J. Fastner & I. Chorus, 2009. Response of cylindrospermopsin production and release in Aphanizomenon flos-aquae (Cyanobacteria) to varying light and temperature conditions. Harmful Algae 8: 645–650.
Price, G. D., D. Sültemeyer, B. Klughammer, M. Ludwig & M. R. Badger, 1998. The functioning of the CO2 concentrating mechanism in several cyanobacterial strains: a review of general physiological characteristics, genes, proteins, and recent advances. Canadian Journal of Botany 76: 973–1002.
Reynolds, C. S., 1984. The Ecology of Freshwater Phytoplankton. Cambridge University Press, Cambridge.
Reynolds, C. S., V. Huszar, C. Kruk, L. Naselli-Flores & S. Melo, 2002. Towards a functional classification of the freshwater phytoplankton. Journal of Plankton Research 24: 417–428.
Scherer, S., E. Stürlz & P. Boger, 1981. Arrhenius plots indicate localization of photosynthetic and respiratory electron transport in different membrane regions of Anabaena. Zeitschrift für Naturforschung Teil C 36: 1036–1040.
Seaburg, K. G., B. C. Parker, R. A. Wharton & G. M. Simmons, 1981. Temperature growth responses of algal isolates from Antarctic oases. Journal of Phycology 17: 353–360.
Simona, M., 2003. Winter and spring mixing depths affect the trophic status and composition of phytoplankton in the northern meromictic basin of Lake Lugano. Journal of Limnology 62: 190–206.
Steemann-Nielsen, E. J., 1952. The use of radioactive carbon (14C) for measuring organic production in the sea. Journal du Conseil Permanent International pour l’Exploration de la Mer 18: 117–140.
Tsujimura, S., K. Ishikawa & H. Tsukada, 2001. Effect of temperature on growth of the cyanobacterium Aphanizomenon flos-aquae in Lake Biwa and Lake Yogo. Phycological Research 49: 275–280.
Uehlinger, U., 1981. Experimental studies on the autecology of Aphanizomenon flos-aquae (Experimentelle Untersuchungen zur Autökologie von Aphanizomenon flos-aquae). Archiv für Hydrobiologie Supplement 60: 260–288.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitteilungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 9: 1–38.
Üveges, V., L. Vörös, J. Padisák & A. W. Kovács, 2011. Primary production of epipsammic algal communities in Lake Balaton (Hungary). Hydrobiologia 660: 17–27.
Vincent, W. F., 2000. Cyanobacterial dominance on Polar regions. In Whitton, B. A. & M. Potts (eds), The Ecology of Cyanobacteria. Kluwer, Netherlands: 321–340.
Vincent, W. F., 2007. Cold tolerance in cyanobacteria and life in the cryosphere. In Seckbach, J. (ed.), Algae and Cyanobacteria in Extreme Environments. Springer, UK: 287–301.
Vincent, W. F., L. G. Whyte, C. Lovejoy, C. W. Greer, I. Laurion, C. A. Suttle, J. Corbeil & D. R. Mueller, 2009. Arctic microbial ecosystems and impacts of extreme warming during the International Polar Year. Polar Science 3: 171–180.
Vincent, W. F., T. V. Callaghan, D. Dahl-Jensen, M. Johansson, K. M. Kovacs, C. Michel, T. Prowse, J. D. Reist & M. Sharp, 2011. Ecological implications of changes in the arctic cryosphere. AMBIO 40: 87–99.
Vyverman, W., E. Verleyen, A. Wilmotte, D. A. Hodgson, A. Willems, K. Peeters, B. Van de Vijver, A. De Wever, F. Leliaert & K. Sabbe, 2010. Evidence for widespread endemism among Antarctic micro-organisms. Polar Science 4: 103–113.
Wagner, C. & R. Adrian, 2009. Cyanobacteria dominance: quantifying the effects of climate change. Limnology and Oceanography 54: 2460–2468.
Walsby, A. E., 1994. Gas vesicles. Microbiological Reviews 58: 94–144.
Wetzel, R. G. & G. E. Likens, 2000. Limnological Analyses. Springer, New York.
Wildman, R., J. H. Loescher & C. L. Winger, 1975. Development and germination of akinetes of Aphanizomenon flos-aquae. Journal of Phycology 11: 96–104.
Wilhelm, S. & R. Adrian, 2008. Impact of summer warming on the thermal characteristics of a polymictic lake and consequences for oxygen, nutrients and phytoplankton. Freshwater Biology 53: 226–237.
Yamamoto, Y., 2009. Environmental factors that determine the occurrence and seasonal dynamics of Aphanizomenon flos-aquae. Journal of Limnology 68: 122–132.
Yamamoto, Y. & H. Nakahara, 2005. The formation and degradation of cyanobacterium Aphanizomenon flos-aquae blooms: the importance of pH, water temperature, and day length. Limnology 6: 1–6.
Yamamoto, Y. & H. Nakahara, 2006. Importance of interspecific competition in the abundance of Aphanizomenon flos-aquae (Cyanophyceae). Limnology 7: 163–170.
Yamamoto, Y. & H. Nakahara, 2007. Factors affecting the fluctuation of akinete and heterocyst numbers of Aphanizomenon flos-aquae (Cyanobacteria) population. Algological Studies 125: 79–96.
Yamamoto, Y. & H. Nakahara, 2009a. Seasonal variations in the morphology of bloom-forming cyanobacteria in a eutrophic pond. Limnology 10: 185–193.
Yamamoto, Y. & H. Nakahara, 2009b. Life cycle of cyanobacterium Aphanizomenon flos-aquae. Taiwania 54: 113–117.
Acknowledgments
This research was implemented through the CENTRAL EUROPE Programme (European Lakes Under Environmental Stressors, 2CE243P3) and was supported by the Leibniz Gemeinschaft (Klimabetriebene Veränderung der Biodiversität von Mikrobiota—TemBi), and by the Hungarian National Science Foundation (OTKA Nr. 105827). We thank Dr. Lajos Vörös, Dr. Attila W. Kovács (Balaton Limnological Research Institute of the Hungarian Academy of Science), Dr. János Somlai and Borbála Máté (University of Pannonia, Institute of Radiochemistry and Radioecology) for their help in the radiocarbon measurements and the technical assistance by the staff of the Leibniz-Institute of Freshwater Ecology and Inland Fisheries for their contributions to the samplings and field measurements.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: N. Salmaso, L. Naselli-Flores, L. Cerasino, G. Flaim, M. Tolotti & J. Padisák / Phytoplankton responses to human impacts at different scales: 16th workshop of the International Association of Phytoplankton Taxonomy and Ecology (IAP)
Rights and permissions
About this article
Cite this article
Üveges, V., Tapolczai, K., Krienitz, L. et al. Photosynthetic characteristics and physiological plasticity of an Aphanizomenon flos-aquae (Cyanobacteria, Nostocaceae) winter bloom in a deep oligo-mesotrophic lake (Lake Stechlin, Germany). Hydrobiologia 698, 263–272 (2012). https://doi.org/10.1007/s10750-012-1103-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-012-1103-3