Abstract
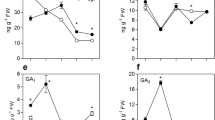
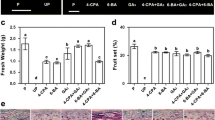
In tomato (Solanum lycopersicum L.), auxin and gibberellins (GAs) cross-talk plays an important role during fruit-set. The entire tomato mutant has been previously reported to carry a deletion in the coding region of the SlIAA9 gene, a member of the auxin signal repressor family Aux/IAA. In this paper, we examined the role of ENTIRE gene in controlling GAs metabolism and directing spontaneous fruit initiation and early ovary growth. It was shown that, similarly to pollinated fruits, facultative parthenocarpy in entire depends on active GA metabolism, since fruit growth is suppressed when GA biosynthesis is blocked. Analysis of endogenous GAs during the first 10 days after flower emasculation revealed that entire fruits accumulated higher amounts of active GAs (GA1 and GA3) in comparison to wild type pollinated fruits, suggesting that a different GA homeostasis regulation occurs. Transcript analysis of the main GA biosynthesis genes showed that differently from unpollinated and non parthenocarpic wild type ovaries, in entire active GA flux modulation is regulated by the activation of SlGA20ox1 and SlGA20ox2 and also by a marked reduction of GA catabolism (reduced transcription of GA 2β-oxidase genes) during the early fruit expansion phase.







Similar content being viewed by others
Abbreviations
- AC:
-
Ailsa Craig
- GA:
-
Gibberellin
- GC–MS/MS:
-
Gas chromatography-tandem mass spectrometry
- Aux/IAA:
-
Auxin/indoleacetic acid
- GA20ox:
-
GA 20-oxidase
- GA3ox:
-
GA 3β-oxidase
- GA2ox:
-
GA 2β-oxidase
References
Aubert D, Chevillard M, Dorne AM, Arlaud G, Herzog M (1998) Expression patterns of GASA genes in Arabidopsis thaliana: the GASA4 gene is up-regulated by gibberellins in meristematic regions. Plant Mol Biol 36:871–883
Audran-Delalande C, Bassa C, Mila I, Regad F, Zouine M, Bouzayen M (2012) Genome-wide identification, functional analysis and expression profiling of the Aux/IAA gene family in tomato. Plant Cell Physiol 53:659–672
Bangerth F (1989) Dominance among fruits/sinks and the search for a correlative signal. Physiol Plantarum 76:608–614
Ben-Nissan G, Weiss D (1996) The petunia homologue of tomato gast1: transcript accumulation coincides with gibberellin-induced corolla cell elongation. Plant Mol Biol 32:1067–1074
Bohner J, Hedden P, Bora-Haber E, Bangerth F (1988) Identification and quantitation of gibberellins in fruits of Lycopersicon esculentum, and their relationship to fruit size in L. esculentum and L. pimpinellifolium. Physiol Plantarum 73:348–353
Bünger-Kibler S, Bangerth F (1982) Relationship between cell number, cell size and fruit size of seeded fruits of tomato (Lycopersicon esculentum Mill.), and those induced parthenocarpically by the application of plant growth regulators. Plant Growth Regul 1:143–154
Coles JP, Phillips AL, Crocker SJ, García-Lepe R, Mervyn JL, Hedden P (1999) Modification of gibberellin production and plant development in Arabidopsis by sense and antisense expression of gibberellin 20-oxidase genes. Plant J 17:547–556
de Jong M, Wolters-Arts M, García-Martínez JL, Mariani C, Vriezen WH (2011) The Solanum lycopersicum auxin response factor 7 (SlARF7) mediates cross-talk between auxin and gibberellin signalling during tomato fruit set and development. J Exp Bot 62:617–626
Dharmasiri N, Dharmasiri S, Estelle M (2005) The F-box protein TIR1 is an auxin receptor. Nature 435:441–445
di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW. InfoStat versión 2012. InfoStat Group, Facultad de Ciencias Agropecuarias, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar
Dorcey E, Urbez C, Blázquez MA, Carbonell J, Perez-Amador MA (2009) Fertilization-dependent auxin response in ovules triggers fruit development through the modulation of gibberellin metabolism in Arabidopsis. Plant J 58:318–332
Fos M, Nuez F, García-Martínez JL (2000) The gene pat-2, which induces natural parthenocarpy, alters the gibberellin content in unpollinated tomato ovaries. Plant Physiol 122:471–480
Fos M, Proaño K, Nuez F, García-Martínez JL (2001) Role of gibberellins in parthenocarpic fruit development induced by the genetic system pat-3/pat-4 in tomato. Physiol Plantarum 111:545–550
Gillaspy G, Ben-David H, Gruissem W (1993) Fruits: a developmental perspective. Plant Cell 5:1439–1451
Hagen G, Guilfoyle TJ (2002) Auxin-responsive gene expression: genes, promoters and regulatory factors. Plant Mol Biol 49:373–385
Hedden P, Thomas SG (2012) Gibberellin biosynthesis and its regulation. Biochemical J 444:11–25
Koenig D, Bayer E, Kang J, Kuhlemeier C, Sinha N (2009) Auxin patterns Solanum lycopersicum leaf morphogenesis. Development 136:2997–3006
Koshioka M, Nishijima T, Yamazaki H, Liu Y, Nonaka M, Mander LN (1994) Analysis of gibberellins in growing fruits of Lycopersicon esculentum after pollination or treatment with 4-chlorophenoxyacetic acid. J Hortic Sci 69:171–180
Mapelli S, Frova C, Torti G, Soressi GP (1978) Relationship between set, development and activities of growth regulators in tomato fruits. Plant Cell Physiol 19:1281–1288
Mariotti L, Picciarelli P, Lombardi L, Ceccarelli N (2011) Fruit-set and early fruit growth in tomato are associated with increases in indoleacetic acid, cytokinin, and bioactive gibberellin contents. J Plant Growth Regul 30:405–415
Martí C, Orzáez D, Ellul P, Moreno V, Carbonell J, Granell A (2007) Silencing of DELLA induces facultative parthenocarpy in tomato fruits. Plant J 52:865–876
Martí E, Carrera E, Ruiz-Rivero O, García-Martínez JL (2010) Hormonal regulation of tomato gibberellin 20-oxidase1 expressed in Arabidopsis. J Plant Phisiol 167:1188–1196
McGiffen ME, Manthey JA (1996) The role of methanol in promoting plant growth: a current evaluation. Hort Sci 31:1092–1096
Mignolli F, Mariotti L, Lombardi L, Vidoz ML, Ceccarelli N, Picciarelli P (2012) Tomato fruit development in the auxin-resistant dgt mutant is induced by pollination but not by auxin treatment. J Plant Physiol 169:1165–1172
Nadeau CD, Ozga JA, Kurepin LV, Jin A, Pharis RP, Reinecke DM (2011) Tissue-specific regulation of gibberellin biosynthesis in developing pea seeds. Plant Physiol 156:897–912
Ngo P, Ozga JA, Reinecke DM (2002) Specificity of auxin regulation of gibberellin 20-oxidase expression in pea pericarp. Plant Mol Biol 49:439–448
O’Neill DP, Davidson SE, Clarke VC, Yamauchi Y, Yamaguchi S, Kamiya Y, Reid JB, Ross JJ (2010) Regulation of the gibberellin pathway by auxin and DELLA proteins. Planta 232:1141–1149
Olimpieri I, Siligato F, Caccia R, Mariotti L, Ceccarelli N, Soressi GP, Mazzucato A (2007) Tomato fruit set driven by pollination or by the parthenocarpic fruit allele are mediated by transcriptionally regulated gibberellin biosynthesis. Planta 226:877–888
Ozga JA, van Huizen R, Reinecke DM (2002) Hormone and seed-specific regulation of pea fruit growth. Plant Physiol 128:1379–1389
Ozga JA, Reinecke DM, Ayele BT, Ngo P, Nadeau C, Wickramarathna AD (2009) Developmental and hormonal regulation of gibberellin biosynthesis and catabolism in pea fruit. Plant Physiol 150:448–462
Pattison RJ, Catalá C (2012) Evaluating auxin distribution in tomato (Solanum lycopersicum) through an analysis of the PIN and AUX/LAX gene families. Plant J 70:585–598
Rademacher W (2000) Growth retardants: effect on gibberellin biosynthesis and other metabolic pathways. Annu Rev Plant Physiol Plant Mol Biol 51:501–531
Rebers M, Kaneta T, Kawaide H, Yamaguchi S, Yang YY, Imai R, Sekimoto H, Kamiya Y (1999) Regulation of gibberellin biosynthesis genes during flower and early fruit development of tomato. Plant J 17:241–250
Rowe RN, Farr DJ, Richards BA (1994) Effects of foliar and root application of methanol and ethanol on the growth of tomato plants (Lycopersicum esculentum Mill.). New Zeal J Crop Hort 22:335–337
Ruan YL, Patrick JW, Bouzayen M, Osorio S, Fernie AR (2012) Molecular regulation of seed and fruit set. Trends Plant Sci 17:656–665
Serrani JC, Fos M, Atarés A, García-Martínez JL (2007a) Effect of gibberellin and auxin on parthenocarpic fruit growth induction in the cv Micro-Tom of tomato. J Plant Growth Regul 26:211–221
Serrani JC, Sanjuán R, Ruiz-Rivero O, Fos M, García-Martínez JL (2007b) Gibberellin regulation of fruit set and growth in tomato. Plant Physiol 145:246–257
Serrani JC, Ruiz-Rivero O, Fos M, García-Martínez JL (2008) Auxin-induced fruit-set in tomato is mediated in part by gibberellins. Plant J 56:922–934
Shi L, Gast RT, Gopalraj M, Olszewski NE (1992) Characterization of a shoot-specific, GA3- and ABA-regulated gene from tomato. Plant J 2:153–159
Sundberg E, Østergaard L (2009) Distinct and dynamic auxin activities during reproductive development. Cold Spring Harb Perspect Biol 1:a001628
Thomas SG, Phillips AL, Hedden P (1999) Molecular cloning and functional expression of gibberellin 2-oxidases, multifunctional enzymes involved in gibberellin deactivation. Proc Natl Acad Sci USA 96:4698–4703
Tiwari SB, Hagen G, Guilfoyle T (2003) The roles of auxin response factor domains in auxin-responsive transcription. Plant Cell 15:533–543
Vivian-Smith A, Koltunow AM (1999) Genetic analysis of growth-regulator-induced parthenocarpy in Arabidopsis. Plant Physiol 121:437–451
Wang H, Jones B, Li Z, Frasse P, Delalande C, Regad F, Chaabouni S, Latché A, Pech JC, Bouzayen M (2005) The tomato Aux/IAA transcription factor IAA9 is involved in fruit development and leaf morphogenesis. Plant Cell 17:2676–2692
Wang H, Shauer N, Usadel B, Frasse P, Zouine M, Hernould M, Latché A, Pech JC, Fernie AR, Bouzayen M (2009) Regulatory features underlying pollination-dependent and -independent tomato fruit set revealed by transcript and primary metabolite profiling. Plant Cell 21:1428–1452
Weiss D, Ori N (2007) Mechanism of cross talk between gibberellin and other hormones. Plant Physiol 144:1240–1246
Worley CK, Zenser N, Ramos J, Rouse D, Leyser O, Theologis A, Callis J (2000) Degradation of Aux/IAA proteins is essential for normal auxin signalling. Plant J 21:553–562
Wu J, Peng Z, Liu S, He Y, Cheng L, Kong F, Wang J, Lu G (2012) Genome-wide analysis of Aux/IAA gene family in Solanaceae species using tomato as a model. Mol Genet Genomics 287:295–311
Zhang J, Cheng R, Xiao J, Quian C, Wang T, Li H, Ouyang B, Ye Z (2007) A single-base deletion mutation in SlIAA9 gene causes tomato (Solanum lycopersicum) entire mutant. J Plant Res 120:671–678
Zimmermann R, Sakai H, Hochholdinger F (2010) The Gibberellic acid stimulated-like gene family in maize and its role in lateral root development. Plant Physiol 152:356–365
Acknowledgments
We would like to thank Dr. A. Pardossi for providing greenhouse facilities and helpful advice during the plant growing season. We are also grateful to Dr. N. Ceccarelli for his critical remarks about the experimental design.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10725_2014_2_MOESM1_ESM.tif
Supplementary material 1 Ratio between GA19 and GA20 plus GA29 concentration in AC pollinated and parthenocarpic entire fruits (TIFF 206 kb)
10725_2014_2_MOESM2_ESM.tif
Supplementary material 2 Diameter of 10-day-old fruits treated with mock (1 % ethanol, 0.1 % Tween 20), 20 ng ovary−1 and 100 ng ovary−1 of the synthetic auxin 4-chlorophenoxyacetic acid (4-CPA) (TIFF 74 kb)
10725_2014_2_MOESM3_ESM.tif
Supplementary material 3 Relationship between flower position in the truss (lower Roman numbers indicate proximity to the stem) and frequency of emasculated flowers that developed parthenocarpic fruits (grey bars) or remained as static ovaries (white bars) (TIFF 124 kb)
Rights and permissions
About this article
Cite this article
Mignolli, F., Vidoz, M.L., Mariotti, L. et al. Induction of gibberellin 20-oxidases and repression of gibberellin 2β-oxidases in unfertilized ovaries of entire tomato mutant, leads to accumulation of active gibberellins and parthenocarpic fruit formation. Plant Growth Regul 75, 415–425 (2015). https://doi.org/10.1007/s10725-014-0002-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-014-0002-1