Abstract
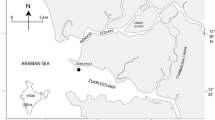
This study presents the response of autotrophic pico- and nanoplankton to southwest monsoon-associated hydrographical transformations in the Cochin backwaters (CBW), the largest monsoonal estuary along the west coast of India. By the onset of the southwest monsoon, the euhaline/mesohaline conditions in the downstream/upstream of CBW usually transform into oligohaline/limnohaline. The flow cytometer analysis revealed the dominance of picoeukaryotes > Synechococcus > nanoautotrophs, with Prochlorococcus either very low or entirely absent. Synechococcus abundance was high during the pre-southwest monsoon (106 L−1), which dwindled with heavy fresh water influx during the southwest monsoon (105 L−1). The drastic drop in salinity and faster flushing of the CBW during the southwest monsoon replaced the euhaline/mesohaline strain of Synechococcus with an oligohaline/limnohaline strain. Epifluorescence microscopy analyses showed that, among the two strains of Synechococcus, the phycoerythrin-rich (PE-rich) one was dominant in the mesohaline/euhaline conditions, whereas the phycocyanin-rich (PC-rich) strain dominated in oligohaline/limnohaline conditions. Although Synechococcus abundance diminished during the southwest monsoon, the total abundance of picoplankton community remained virtually unchanged in the upstream due to an increase in the abundance of picoeukaryotes. On the other hand, the autotrophic nanoplankton abundance increased from pre-monsoon levels of av. 3.8 × 106–av. 9.5 × 106 L−1 at the onset of the southwest monsoon. Utilizing suitable multivariate analyses, the study illustrated the differential response and niche preference of various smaller communities of autotrophs to the southwest monsoon-associated hydrographical ramifications in a large monsoonal estuary, which may be applicable to similar such estuaries situated along the Indian coastline.









Similar content being viewed by others
References
Ahlgren, N. A., & Rocap, G. (2006). Culture isolation and culture-independent clone libraries reveal new marine Synechococcus ecotypes with distinctive light and N physiologies. Applied and Environmental Microbiology, 72, 7193–7204.
Azam, F., Fenchel, T., Field, J. G., Gray, J. S., Meyer-Reil, L. A., & Thingstad, F. (1983). The ecological role of water-column microbes in the sea. Marine Ecology Progress Series, 10, 257–263.
Badylak, S., & Philips, E. J. (2004). Spatial and temporal patterns of phytoplankton composition in a subtropical coastal lagoon, the Indian River Lagoon, Florida, USA. Journal of Plankton Research., 26, 1229–1247.
Birks, H. J. B. (1998). Numerical tools in palaeolimnology – progress, potentialities, and problems. Journal of Paleolimnology., 20, 307–332.
Bloem, J., Marie-Jose, B.-G., & Cappenberg, T. E. (1986). Fixation, counting, and manipulation of heterotrophic nanoflagellates. Applied Environmental Microbiology, 52, 1266–1272.
Burkill, P. H., Leakey, R. J. G., Owens, N. J. P., & Mantoura, R. F. C. (1993). Synechococcus and its importance to the microbial food web of the northwestern Indian Ocean. Deep sea research part II., 40, 773–782.
Callieri, C., Amicucci, E., Bertoni, R., & Voros, L. (1996). Fluorometric characterization of two picocyanobacteria strains from different underwater light quality. International Revue der gesamten Hydrobiologie und Hydrographie, 81, 13–23.
Cermeno, P., Maranon, E., Perez, V., Serret, P., Fernandez, E., & Castr, C. G. (2006). Phytoplankton size structure and primary production in highly dynamic coastal ecosystem (Ria de Vigo, NW-Spain): Ssasonal and short-time scale variability. Estuarine Coastal and Shelf Science., 67, 251–266.
Clarke, K. R., & Warwick, R. M. (1994). Similarity-based testing for community pattern: the 2-way layout with no replication. Marine Biology, 118, 167–176.
Ernst, A. (1991). Cyanobacterial picoplankton from Lake Constance: I. Isolation by fluorescence characteristics. Journal of Plankton Research, 13, 1307–1312.
Fuller, N. J., Marie, D., Partensky, F., Vaulot, D., Post, A. F., & Scanlan, D. J. (2003). Clade-specific 16S rDNA oligonucleotides reveal the predominance of an single marine Synechococcus clade throughout a stratified water column in the Red Sea. Applied Environmental Microbiology, 69, 2430–2443.
Grassholf, K., Ehrhardt, M., & Kremling, K. (1983). In K. Grassholf, M. Ehrhardt, & K. Kremling (Eds.), Methods of Seawater Analysis (pp. 89–224). Weinheim: Verlag Chemie.
Jyothibabu, R., Madhu, N. V., Jayalakshmi, K. V., Balachandran, K. K., Shiyas, C. A., & Martin, G. D. (2006). Impact of freshwater influx on microzooplankton mediated food web in a tropical estuary (Cochin backwaters – India). Estuarine, Coastal and Shelf Science, 69, 505–18.
Jyothibabu, R., Mohan, A. P., Jagadeesan, L., Anjusha, A., Muraleedharan, K. R., Lallu, K. R., Krishna, K., & Ullas, N. (2013). Ecology and trophic preference of picoplankton and nanoplankton in the Gulf of Mannar and the Palk Bay, southeast coast of India. Journal of Marine Systems, 111-112, 29–44.
Jyothibabu, R, Madhu, NV, Martin, GD, Aneesh, C, Sooria, P, Vineetha, G. (2014). Waning of plankton food web in the upstream region of the Cochin backwaters during the southwest monsoon. Indian Journal of Marine Sciences. (In Press).
Jyothibabu, R., Madhu, N. V., Jagadeesan, L., Anjusha, A., Mohan, A. P., Ullas, N., Sudheesh, N., & Karnan, C. (2014b). Why do satellite imageries show exceptionally high chlorophyll in the Gulf of Mannar and the Palk Bay during the northeast monsoon? Environmental Monitoring Assessment, 186, 7781–7792.
Lampitt, R., Wishner, K. F., Turley, C. M., & Angel, M. V. (1993). Marine snow studies in the Northeast Atlantic: distribution composition and role as a food source for migrating plankton. Marine Biology., 116, 689–702.
Leps, J., & Smilauer, P. S. (2003). Multivariate analysis of ecological data using CANOCO. Cambridge, UK: Cambridge University Press.
Li, W. K. W., & Wood, A. M. (1988). Vertical distribution of North Atlantic ultraphytoplankton: analysis by flow cytometry and epifluorescence microscopy. Deep-Sea Research I., 35, 1615–1638.
Li, W. K. W., Rao, D. V. S., Harrison, W. G., Smith, J. C., Cullen, J. J., Irwin, B., & Platt, T. (1983). Autotrophic picoplankton in the tropical ocean. Science New Series, 219, 292–295.
Madhu, N. V., Jyothibabu, R., Balachandran, K. K., Honey, U. K., Martin, G. D., Vijay, J. G., Shiyas, C. A., Gupta, G. V. M., & Achuthankutty, C. T. (2007). Monsoonal impact on planktonic standing stock and abundance in a tropical estuary (Cochin Backwaters – India). Estuarine Coastal and Shelf Science., 73, 54–64.
Madhupratap, M. (1987). Status and strategy of zooplankton of tropical Indian estuaries: a review. Bulletin of the Plankton Society of Japan, 34, 65–81.
Mann, E. L., Ahlgren, N., Moffett, J. W., & Chisholm, S. W. (2002). Copper toxicity and cyanobacteria ecology in the Sargasso Sea. Limnology and Oceanography, 47, 976–988.
Mclusky, D. S. (1993). Marine and estuarine gradients – an overview. Netherlands Journal of Aquatic Ecology, 27, 489–493.
Mitbavkar, S., & Anil, A. C. (2011). Tiniest primary producers in the marine environment: an appraisal from the context of waters around India. Current Science, 100, 986–988.
Mitbavkar, S., Rajaneesh, K., Anil, A., & Sundar, D. (2012). Picophytoplankton community in a tropical estuary: detection of Prochlorococcus-like populations. Estuarine Coastal and Shelf Science, 107, 159–164.
Naik, R. K., Anil, A. C., Narale, D. D., Chitari, R. R., & Kulkarni, V. V. (2011). Primary description of surface waters phytoplankton pigment patterns in the Bay of Bengal. Journal of Sea Research, 65, 435–441.
Partensky, F., Blanchot, J., Lantoine, F., Neveux, J., & Marie, D. (1996). Vertical structure of picophytoplankton at different trophic sites of the tropical northeastern Atlantic Ocean. Deep-Sea Research I, 43, 1191–1213.
Partensky, F., Hess, W., & Vaulot, D. (1999). Prochlorococcus, a marine photosynthetic prokaryote of global significance. Microbiology and Molecular Biology, 63, 106–127.
Philips, E. J., Badylak, S., & Lynch, T. C. (1999). Blooms of the picoplanktonic cyanobacterium Synechococcus in Florida Bay, a subtropical inner-shelf lagoon. Limnology and Oceanography, 44, 1166–1175.
Pick, F. R., & Agbeti, D. M. (1991). The seasonal dynamic and composition of photosynthetic picoplankton communities in temperate lakes in Ontario, Canada. Internationale Revue der gesamten Hydrobiologie, 76, 565–580.
Pomeroy, L. R. (1974). The Ocean’s Food Web, A Changing Paradigm. BioScience., 24, 499–504.
Qasim, S. Z. (2003). Cochin backwaters and Vembanad. In S. Z. Qasim (Ed.), Indian estuaries (pp. 305–382). Mumbai: Allied Publishers Pvt. Ltd. ISBN 81-7764-369-X.
Rajaneesh, K. M., & Mitbavkar, S. (2013). Factors controlling the temporal and spatial variations in Synechococcus abundance in a monsoonal estuary. Marine Environmental Research., 92, 133–143.
Saraladevi, K., Venugopal, P., Remani, K. N., Zacharias, D., & Unnithan, R. V. (1983). Nutrients in some estuaries of Kerala. Mahasagar – The Bulletin of National Institute of Oceanography Goa, 16, 161–173.
Stockner, J., & Anita, N. (1986). Algal pico plankton from marine and freshwater ecosystems: a multidisciplinary perspective. Canadian Journal of Fisheries and Aquatic Sciences., 43, 2472–2502.
Stomp, M., Huisman, J., Voros, L., Pick, F. R., Laamanen, M., Haverkamp, T., & Stal, L. J. (2007). Colourful coexistence of red and green picocyanobacteria in lakes and seas. Ecology Letters, 10, 290–298.
Tan, Y., Huang, L., Chen, Q., & Huang, X. (2004). Seasonal variation in zooplankton composition and grazing impact on phytoplankton standing stock in the Pearl River estuary, China. Continental Shelf Research, 24, 1949–1968.
UNESCO. (1994). Protocols for the Joint Global Ocean Flux Study (JGOFS). Core measurements, IOC manuals and guides 29 (p. 170). Paris: UNESCO.
Vaulot, D., Lebot, N., Marie, D., & Fukai, E. (1996). Effect of phosphorous on the Synechococcus cell cycle in surface Mediteranean waters during summer. Applied and Environmental Microbiology, 62, 2527–2533.
Veldhuis, M. J. W., Kraay, G. W., & Gieskes, W. W. C. (1993). Growth and fluorescence characteristics of ultra-plankton on a north-south transect in the eastern North Atlantic. Deep-Sea Research II, 40, 609–626.
Vijith, V., Sundar, D., & Shetye, S. R. (2009). Time-dependence of salinity in monsoonal estuaries. Estuarine Coastal and Shelf Science, 85, 601–608.
Voros, L., Callieri, C., Balogh, K. V., & Bertoni, R. (1998). Freshwater picocyanobacteria along a tropical gradient and light quality range. Hydrobiologia, 369/370, 117–125.
Wang, K., Wommack, K. E., & Chen, F. (2011). Abundance and distribution of Synechococcus spp. and cyanophages in the Chesapeake Bay. Applied. Environmental. Microbiology., 77, 7459–7468.
Wellershaus, S. (1974). Seasonal changes in the zooplankton population in the Cochin backwater (a south Indian estuary). Hydrobiological Bulletin, 8, 213–223.
Wood, A. M., Horan, P. K., Muirhead, K., Phinney, D. A., Yentsch, C. M., & Waterbury, J. B. (1985). Discrimination between types of pigments in marine Synechococcus spp. by scanning spectroscopy, epifluorescence microscopy, and flow cytometry. Limnol Oceanography, 30, 1303–1315.
Worden, A. Z., & Binder, B. J. (2003). Growth regulation of rRNA content of Prochlorococcus and Synechococcus (marine cyanobacteria) measured by whole-cell hybridization of rRNA targeted peptide nucleic acids. Journal of Phycology, 39, 527–534.
Acknowledgments
The authors thank the director of CSIR-National Institute of Oceanography (NIO), India, for their facilities. The authors thank the scientist-in-charge of CSIR NIO, RC Kochi, for encouragement. ICMAM-PD, Chennai, provided financial support. This is NIO contribution 5853.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohan, A.P., Jyothibabu, R., Jagadeesan, L. et al. Summer monsoon onset-induced changes of autotrophic pico- and nanoplankton in the largest monsoonal estuary along the west coast of India. Environ Monit Assess 188, 93 (2016). https://doi.org/10.1007/s10661-016-5096-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-016-5096-7