Abstract
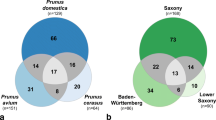
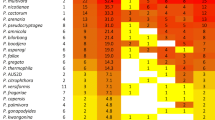
Endophytic fungal hitch-hikers have been difficult to detect in the past, and have potentially spread these latent pathogens via the global plant trade. The African genera Protea, Leucospermum and Leucadendron, commercially referred to as proteas, form the basis of a global flower production industry. The largest producers of proteas are Australia and South Africa, followed by Portugal and Spain. In the 1990s propagation material from South Africa was used to establish protea orchards in Portugal. We utilized metabarcoding to determine if this plant trade has carried host-specific fungal pathogens to a new environment. Wood samples collected from asymtomatic twigs from Portuguese farms, where propagation material had been imported from South Africa, was compared to material from South African farms that originally produced and supplied rooted and unrooted cuttings. DNA metabarcoding, using fungal-specific primers for the ITS2 gene region, produced 1237 OTUs. Focusing only on known pathogens of protea, we found that the Portuguese orchards contained fungal disease agents associated with Proteaceae or other plant families from the Southern Hemisphere. Our sampling technique could be used by agencies and applied to other plant material and pathogens to reduce the spread of pathogens.

Similar content being viewed by others
References
Barrow, J. R., Osuna-avila, P., Range, J. E., Box, P. O., & Cruces, L. (2004). Fungal endophytes intrinsically associated with micropropagated plants regenerated from native Bouteloua eriopoda Torr. And Atriplex canescens (Pursh) Nutt. In Vitro Cellular & Developmental Biology - Plant, 109(5), 608–612. https://doi.org/10.1079/IVP2004584.
Bezerra, J. D. P., Sandoval-denis, M., Paiva, L. M., Silva, G. A., & Groenewald, J. Z. (2017). New endophytic Toxicocladosporium species from cacti in Brazil , and description of Neocladosporium gen. nov. IMA Fungus, 8(1), 77–97. https://doi.org/10.5598/imafungus.2017.08.01.06.
Brasier, C. M. (2008). The biosecurity threat to the UK and global environment from international trade in plants. Plant Pathology, 57(5), 792–808. https://doi.org/10.1111/j.1365-3059.2008.01886.x.
Burgess, T., Crous, C. J., Slippers, B., Hantula, J., & Wingfield, M. J. (2016). Tree invasions and biosecurity: Eco-evolutionary dynamics of hitchhiking fungi. Annals of Botany Plants, 8, plw076. https://doi.org/10.1093/aobpla/plw076.
Burgess, T. I., & Wingfield, M. J. (2002). Quarantine is important in restricting the spread of exotic seed-borne tree pathogens in the southern hemisphere. International Forestry Review, 4(1), 56–65 https://www.jstor.org/stable/43740945.
Burgess, T., & Wingfield, M. J. (2016). Pathogens on the move: A 100-year global experiment with planted eucalypts. BioScience, 67(1), 14–25. https://doi.org/10.1093/biosci/biw146.
Burgess Tan, Y. P., Garnas, J., Edwards, J., Scarlett, K. A., Shuttleworth, L. A., et al. (2019). Current status of the Botryosphaeriaceae in Australia. Australasian Plant Pathology, 48, 35–44. https://doi.org/10.1007/s13313-018-0577-5.
Caporaso, J. G., Kuczynski, J., Stombaugh, J., Bittinger, K., Bushman, F. D., Costello, E. K., Fierer, N., Peña, A. G., Goodrich, J. K., Gordon, J. I., Huttley, G. A., Kelley, S. T., Knights, D., Koenig, J. E., Ley, R. E., Lozupone, C. A., McDonald, D., Muegge, B. D., Pirrung, M., Reeder, J., Sevinsky, J. R., Turnbaugh, P. J., Walters, W. A., Widmann, J., Yatsunenko, T., Zaneveld, J., & Knight, R. (2010). QIIME allows analysis of high- throughput community sequencing data intensity normalization improves color calling in SOLiD sequencing. Nature Methods, 7(5), 335–336. https://doi.org/10.1038/nmeth0510-335.
Cleary, M., Oskay, F., Doğmuş, H. T., Lehtijärvi, A., Woodward, S., & Vettraino, A. M. (2019). Cryptic risks to forest biosecurity associated with the global movement of commercial seed. Forests, 10(5). https://doi.org/10.3390/f10050459.
Coutinho, T. A., Wingfield, M. J., Alfenas, A. C., & Crous, P. W. (1998). Eucalyptus rust : A disease with the potential for serious international implications. Plant Disease, 82(7), 819–825. https://doi.org/10.1094/PDIS.1998.82.7.819.
Crone, M., Mccomb, J. A., Brien, P. A. O., Hardy, G. E. S. J., & Voglmayr, H. (2013). Survival of Phytophthora cinnamomi as oospores , stromata , and thick-walled chlamydospores in roots of symptomatic and asymptomatic annual and herbaceous perennial plant species. Fungal Biology, 117(2), 112–123. https://doi.org/10.1016/j.funbio.2012.12.004.
Crous, P. W., Denman, S., Taylor, J. E., Swart, L., Bezuidenhout, C. M., Hoffman, L., et al. (2013). Cultivation and diseases of proteaceae: Leucadendron, Leucospermum and Protea (2nd ed.). Utrecht, the Netherlands: CBS-KNAW fungal biodiversity Centre (CBSbiodiversity series 13).
Crous, P. W., & Groenewald, J. Z. (2011). Why everlastings don’t last. Persoonia: Molecular Phylogeny and Evolution of Fungi, 26, 70–84. https://doi.org/10.3767/003158511X574532.
Crous, P. W., Groenewald, J. Z., & Shivas, R. G. (2010). Devriesia fraseriae Crous & RG Shivas, sp. nov. Persoonia - Molecular Phylogeny and Evolution of Fungi, 25, 150–151. https://doi.org/10.3767/003158510X552636.
Crous, P. W., Groenewald, J. Z., Slippers, B., & Wingfield, M. J. (2016). Global food and fibre security threatened by current inefficiencies in fungal identification. Philosophical Transactions of the Royal Society B: Biological Sciences, 371(1709), 20160024. https://doi.org/10.1098/rstb.2016.0024.
Crous, P. W., Schoch, C. L., Hyde, K. D., Wood, A. R., Gueidan, C., De Hoog, G. S., & Groenewald, J. Z. (2009). Phylogenetic lineages in the Capnodiales. Studies in Mycology, 64, 17–47. https://doi.org/10.3114/sim.2009.64.02.
Crous, P. W., Schubert, K., Braun, U., De Hoog, G. S., Hocking, A. D., Shin, H. D., & Groenewald, J. Z. (2007). Opportunistic, human-pathogenic species in the Herpotrichiellaceae are phenotypically similar to saprobic or phytopathogenic species in the Venturiaceae. Studies in Mycology, 58(2005), 185–217. https://doi.org/10.3114/sim.2007.58.07.
Crous, P. W., Summerell, B. A., Mostert, L., & Groenewald, J. Z. (2008). Host specificity and speciation of Mycosphaerella and Teratosphaeria species associated with leaf spots of Proteaceae. Persoonia: Molecular Phylogeny and Evolution of Fungi, 20, 59–86. https://doi.org/10.3767/003158508X323949.
Crous, P. W., Summerell, B. A., Shivas, R. G., Romberg, M., Mel’nik, V. A., Verkley, G. J., & Groenewald, J. Z. (2011a). Fungal planet description sheets: 92-106. Persoonia: Molecular Phylogeny and Evolution of Fungi, 27, 130–162. https://doi.org/10.3767/003158511X617561.
Crous, P. W., Summerell, B. A., Swart, L., Denman, S., Taylor, J. E., Bezuidenhout, C. M., Palm, M. E., Marincowitz, S., & Groenewald, J. Z. (2011b). Fungal pathogens of Proteaceae. Persoonia: Molecular Phylogeny and Evolution of Fungi, 27, 20–45. https://doi.org/10.3767/003158511X606239.
Crous, P. W., Summerell, B. A., Taylor, J. E., & Bullock, S. (2000). Fungi occurring on Proteaceae in Australia: Selected foliicolous species. Australasian Plant Pathology, 29(4), 267–278. https://doi.org/10.1071/AP00050.
Crous, P. W., Wingfield, M. J., Schumacher, R. K., Summerell, B. A., Giraldo, A., Gené, J., et al. (2014). Fungal Planet description sheets : 281–319, 212–289.
Denman, S., Crous, P. W., Groenewald, J. Z. E., Slippers, B., Wingfield, B. D., Wingfield, M. J., et al. (2003). Circumscription of Botryosphaeria species associated with Proteaceae based on morphology and DNA sequence data circumscription of Botryosphaeria species associated with Proteaceae based on morphology and DNA sequence data. Mycologia, 95(2), 294–307. https://doi.org/10.1080/15572536.2004.11833114.
Dissanayake, A. J., Purahong, W., Wubet, T., Hyde, K. D., Zhang, W., Xu, H., Zhang, G., Fu, C., Liu, M., Xing, Q., Li, X., & Yan, J. (2018). Direct comparison of culture-dependent and culture-independent molecular approaches reveal the diversity of fungal endophytic communities in stems of grapevine (Vitis vinifera). Fungal Diversity, 90(1), 85–107. https://doi.org/10.1007/s13225-018-0399-3.
Edgar, R. C. (2010). Search and clustering orders of magnitude faster than BLAST. Bioinformatics, 26(19), 2460–2461. https://doi.org/10.1093/bioinformatics/btq461.
Edgar, R. C., Haas, B. J., Clemente, J. C., Quince, C., & Knight, R. (2011). UCHIME improves sensitivity and speed of chimera detection. Bioinformatics, 27(16), 2194–2200. https://doi.org/10.1093/bioinformatics/btr381.
Eschen, R., Douma, J. C., Grégoire, J.-C., Mayer, F., Rigaux, L., & Potting, R. P. J. (2017). A risk categorisation and analysis of the geographic and temporal dynamics of the European import of plants for planting. Forest Invasions, 19, 3243–3257. https://doi.org/10.1007/s10530-017-1465-6.
Fernandez-Conradi, P., Fort, T., Castagneyrol, B., Jactel, H., & Robin, C. (2019). Fungal endophyte communities differ between chestnut galls and surrounding foliar tissues. Fungal Ecology, 42, 100876. https://doi.org/10.1016/j.funeco.2019.100876.
Franić, I., Prospero, S., Hartmann, M., Allan, E., Auger-Rozenberg, M., Grünwald, N. J., et al. (2019). Are traded forest tree seeds a potential source of nonnative pests? Ecological Applications, 29(7), e01971. https://doi.org/10.1002/eap.1971.
Gladieux, P., Feurtey, A., Hood, M. E., Snirc, A., Clavel, J., & Dutech, C. (2015). The population biology of fungal invasions. Molecular Ecology, 24, 1969–1986. https://doi.org/10.1111/mec.13028.
Griffin, E. A., Harrison, J. G., McCormick, M. K., Burghardt, K. T., & Parker, J. D. (2019). Tree diversity reduces fungal endophyte richness and diversity in a large-scale temperate forest experiment. Diversity, 11(12), 234. https://doi.org/10.3390/d11120234.
Gryzenhout, M., Myburg, H., Merwe, N. A., Wingfield, B. D., & Wingfield, M. J. (2004). Chrysoporthe, a new genus to accommodate Cryphonectria cubensis. Studies in Mycology, 50, 119–142.
Gryzenhout, M., Rodas, C. A., Portales, J. M., Clegg, P., Wingfield, B. D., & Wingfield, M. J. (2006). Novel hosts of the Eucalyptus canker pathogen Chrysoporthe cubensis and a new Chrysoporthe species from Colombia. Mycological Research, 110(7), 833–845. https://doi.org/10.1016/j.mycres.2006.02.010.
Hernandez-Escribano, L., Iturritxa, E., Aragon, A., Mesanza, N., Berbegal, M., Raposo, R., & Elvira-recuenco, M. (2018). Root infection of canker pathogens, Fusarium circinatum and Diplodia sapinea, in asymptomatic trees in Pinus radiata and Pinus pinaster plantations. Forests, 9(128), 1–15. https://doi.org/10.3390/f9030128.
https://blast.ncbi.nlm.nih.gov/Blast.cgi?P. (n.d.). https://blast.ncbi.nlm.nih.gov/Blast.cgi?P. Accessed 1 February 2018.
Https://www.ncbi.nlm.nih.gov/sra/PRJNA548865, S. reference number P. (n.d.). NCBI/sra. https://www.ncbi.nlm.nih.gov/sra/PRJNA548865, SRA reference number PRJNA548865. Accessed 20 June 2019.
Ihrmark, K., Bödeker, I., Cruz-Martinez, K., Friberg, H., Kubartova, A., Schenck, J., et al. (2012). New primers to amplify the fungal ITS2 region – Evaluation by 454-sequencing of artificial and natural communities ¨. FEMS Microbiology Ecology, 82(3), 666–677. https://doi.org/10.1111/j.1574-6941.2012.01437.x.
Jimu, L., Kemler, M., Mujuru, L., & Mwenje, E. (2017). Illumina DNA metabarcoding of Eucalyptus plantation soil reveals the presence of mycorrhizal and pathogenic fungi. Forestry An International Journal ofForest Research, 91(2), 238–245. https://doi.org/10.1093/forestry/cpx046.
Jimu, L., Kemler, M., Wingfield, M. J., Mwenje, E., & Roux, J. (2016). The Eucalyptus stem canker pathogen Teratosphaeria zuluensis detected in seed samples. Forestry, 89(3), 316–324. https://doi.org/10.1093/forestry/cpv037.
Kearse, M., Moir, R., Wilson, A., Stones-havas, S., Sturrock, S., Buxton, S., et al. (2012). Geneious basic : An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics, 28(12), 1647–1649. https://doi.org/10.1093/bioinformatics/bts199.
Kõljalg, U., Nilsson, R. H., Abarenkov, K., Tedersoo, L., Taylor, A. F. S., Bahram, M., Bates, S. T., Bruns, T. D., Bengtsson-Palme, J., Callaghan, T. M., Douglas, B., Drenkhan, T., Eberhardt, U., Dueñas, M., Grebenc, T., Griffith, G. W., Hartmann, M., Kirk, P. M., Kohout, P., Larsson, E., Lindahl, B. D., Lücking, R., Martín, M. P., Matheny, P. B., Nguyen, N. H., Niskanen, T., Oja, J., Peay, K. G., Peintner, U., Peterson, M., Põldmaa, K., Saag, L., Saar, I., Schüßler, A., Scott, J. A., Senés, C., Smith, M. E., Suija, A., Taylor, D. L., Telleria, M. T., Weiss, M., & Larsson, K. H. (2013). Towards a unified paradigm for sequence-based identification of fungi. Molecular Ecology, 22(21), 5271–5277. https://doi.org/10.1111/mec.12481.
Liebhold, A. M., Brockerhoff, E. G., Garrett, L. J., Parke, J. L., & Britton, K. O. (2012). Live plant imports: The major pathway for forest insect and pathogen invasions of the US. Frontiers in Ecology and the Environment, 10(3), 135–143. https://doi.org/10.1890/110198.
Martinez Arbizu, P. (2017). pairwiseAdonis: Pairwise multilevel comparison using Adonis. R package version 0.3. https://github.com/pmartinezarbizu/pairwiseAdonis
Migliorini, D., Ghelardini, L., Tondini, E., Luchi, N., & Santini, A. (2015). The potential of symptomless potted plants for carrying invasive soilborne plant pathogens. Diversity and Distributions, 21(10), 1218–1229. https://doi.org/10.1111/ddi.12347.
Oksanen, J., Blanchet, F. G., Kindt, R., Legendre, P., Minchin, P. R., O’Hara, R., et al. (2018). Vegan: Community ecology package. R package version, 2, 5–3 https://cran.r-project.org/package=vegan.
Paap, T., Brouwers, N. C., Burgess, T. I., & Hardy, G. E. S. J. (2017). Importance of climate , anthropogenic disturbance and pathogens ( Quambalaria coyrecup and Phytophthora spp .) on marri ( Corymbia calophylla ) tree health in southwest Western Australia. Annals of Forest Science, 74(3), 62. https://doi.org/10.1007/s13595-017-0658-6.
Pirttila, A. M., Laukkanen, H., & Hohtola, A. (2002). Chitinase production in pine callus ( Pinus sylvestris L .): a defense reaction against endophytes ? Planta, 214, 848–852. https://doi.org/10.1007/s00425-001-0709-x.
Pirttila, A. M., Pospiech, H., Laukkanen, H., Myllyla, R., & Hohtola, A. (2003). Two endophytic fungi in different tissues of scots pine buds ( Pinus sylvestris L.). Microvial Ecology, 45, 53–62. https://doi.org/10.1007/s00248-002-1038-8.
Qian, X., Chen, L., Guo, X., He, D., Shi, M., & Zhang, D. (2018). Shifts in community composition and co-occurrence patterns of phyllosphere fungi inhabiting Mussaenda shikokiana along an elevation gradient. PeerJ, 6, e5767. https://doi.org/10.7717/peerj.5767.
Quaedvlieg, W., Binder, M., Groenewald, J. Z., Summerell, B. A., Carnegie, A. J., Burgess, T. I., & Crous, P. W. (2014). Introducing the consolidated species concept to resolve species in the Teratosphaeriaceae. Persoonia: Molecular Phylogeny and Evolution of Fungi, 33, 1–40. https://doi.org/10.3767/003158514X681981.
R Core Team. (2018). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.r-project.org
Rassati, D., Faccoli, M., Toffolo, E. P., & Battisti, A. (2015). Improving the early detection of alien wood-boring beetles in ports and surrounding forests. Journal of Applied Ecology, 52, 50–58. https://doi.org/10.1111/1365-2664.12347.
Rodas, C. A., Gryzenhout, M., Myburg, H., Wingfield, B. D., & Wingfield, M. J. (2005). Discovery of the Eucalyptus canker pathogen Chrysoporthe cubensis on native Miconia (Melastomataceae) in Colombia. Plant Pathology, 54(4), 460–470. https://doi.org/10.1111/j.1365-3059.2005.01223.x.
Sakalidis, M. L., Slippers, B., Wingfield, B. D., & Hardy, G. E. S. J. (2013). The challenge of understanding the origin , pathways and extent of fungal invasions : global populations of the Neofusicoccum parvum – N. ribis species complex. Diversity and Distributions, 19(8), 873–883. https://doi.org/10.1111/ddi.12030.
Santini, A., Ghelardini, L., De Pace, C., Desprez-Loustau, M. L., Capretti, P., Chandelier, A., et al. (2013). Biogeographical patterns and determinants of invasion by forest pathogens in Europe. New Phytologist, 197(1), 238–250. https://doi.org/10.1111/j.1469-8137.2012.04364.x.
Slippers, B., Stenlid, J., & Wingfield, M. J. (2005). Emerging pathogens : Fungal host jumps following anthropogenic introduction. Trends in Ecology and Evolution, 20(8), 420–421. https://doi.org/10.1016/j.tree.2005.04.024.
Slippers, B., & Wingfield, M. J. (2007). Botryosphaeriaceae as endophytes and latent pathogens of woody plants : diversity , ecology and impact. Fungal Biology Reviewes, 21, 90–106. https://doi.org/10.1016/j.fbr.2007.06.002.
Unterseher, M., Siddique, A. B., Brachmann, A., & Per, D. (2016). Diversity and composition of the leaf mycobiome of beech ( Fagus sylvatica ) are affected by local habitat conditions and leaf biochemistry. PLoS One, 11(4), e0152878. https://doi.org/10.1371/journal.pone.0152878.
Valenzuela-Lopez, N., Cano-Lira, J. F., Guarro, J., Sutton, D. A., Wiederhold, N., Crous, P. W., & Stchigel, A. M. (2018). Coelomycetous Dothideomycetes with emphasis on the families Cucurbitariaceae and Didymellaceae. Studies in Mycology, 90, 1–69. https://doi.org/10.1016/j.simyco.2017.11.003.
Vettraino, A. M., Li, H. M., Eschen, R., Morales-Rodriguez, C., & Vannini, A. (2017). The sentinel tree nursery as an early warning system for pathway risk assessment: Fungal pathogens associated with Chinese woody plants commonly shipped to Europe. PLoS One, 12(11), e0188800. https://doi.org/10.1371/journal.pone.0188800.
Vettraino, A. M., Roques, A., Yart, A., Fan, J. T., Sun, J. H., & Vannini, A. (2015). Sentinel trees as a tool to forecast invasions of alien plant pathogens. PLoS One, 10(3), e0120571. https://doi.org/10.1371/journal.pone.0120571.
Wang, M. M., Shenoy, B. D., Li, W., & Cai, L. (2017). Molecular phylogeny of neodevriesia, with two new species and several new combinations. Mycologia, 109(6), 965–974. https://doi.org/10.1080/00275514.2017.1415075.
White, T., Bruns, T., Lee, S., & Taylor, J. (1990). Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications (Innis MA, Gelfland DH, Sninsky JJ & White TJ, eds) (academic P., pp. 315–322). San Diego, CA.
Wickham, H. (2016). ggplot2: Elegant graphics for data analysis. (springer-Verlag New York, Ed.). New York.
Wingfield, M. J., Brockerhoff, E. G., Wingfield, B. D., & Slippers, B. (2015). Planted forest health: The need for a global strategy. Science, 349(6250), 832–836. https://doi.org/10.1126/science.aac6674.
Acknowledgments
We are grateful for the assistance provided by the European Cooperation in Science and Technology (Action FP1401: A global network of nurseries as early warning system against alien tree pests - Global Warning). We also thank Kay Howard for assistance with the manuscript preparation and Chris Shaw for his work on the statistics.
Data availability
The data that support the findings of this study are openly available in NCBI at https://www.ncbi.nlm.nih.gov/sra/PRJNA548865, SRA reference number PRJNA548865. Temporary Submission ID: SUB5845073.
Code availability
(Not applicable)
Author information
Authors and Affiliations
Contributions
DM, TB and MW have made major contributions to the conception of the study and the writing of the manuscript, DM collected all the samples. DM and MM were responsible for the acquisition, analysis, and interpretation of the data. AS, AR and PT facilitated sampling and were involved in the critical editing for important intellectual content.
Corresponding author
Ethics declarations
Ethics approval
(Not applicable)
Consent to participate
(Not applicable)
Consent for publication
(Not applicable)
Conflicts of interest/competing interests
The authors declare no conflicts of financial or personal interest.
Rights and permissions
About this article
Cite this article
Migliorini, D., Messal, M., Santini, A. et al. Metabarcoding reveals southern hemisphere fungal endophytes within wood of cultivated Proteaceae in Portugal. Eur J Plant Pathol 160, 173–184 (2021). https://doi.org/10.1007/s10658-021-02233-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-021-02233-8