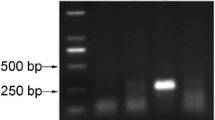
Abstract
Members of the genus Tospovirus which can infect plants in the family Bunyaviridae, possess quasi-spherical enveloped particles (80–120 nm in diameter) and a segmented tripartite single-stranded RNA genome, with large (L), medium (M) and small (S) RNA segments. Tospoviruses are vectored by thrips in a persistent manner and cause yield losses of numerous economic crops worldwide. Inspection is an important measure to prevent invasion of tospoviruses. However, the increase of new virus species makes inspection challenging. In this study, a degenerate primer pair dTospo-F2/dTospo-R2 was designed from the conserved regions of the tospoviral L RNA sequences and used for the tospovirus detection in SYBR Green I-based quantitative real-time reverse transcription-polymerase chain reaction (RT-qPCR). The designed primers could amplify specific products from all tested 20 tospovirus species. The specificity of amplifications was validated by melting curve assays, agarose gel electrophoresis and direct sequencing of amplicons. Furthermore, the degenerate primer pair was used in RT-qPCR to detect tospovirus infections from field cowpea and sweet pepper samples. Sequencing of the amplicons confirmed the identity of the viruses as Groundnut chlorotic fan-spot virus (GCFSV) from the cowpea and Tomato spotted wilt virus from the sweet pepper. Our results demonstrated that the degenerate primer pair dTospo-F2/dTospo-R2 is highly specific to tospoviruses, and when used in RT-qPCR, it greatly simplifies and enhances the efficacy of the inspection of tospoviruses. Additionally, this is the first report of GCFSV infected cowpea.




Similar content being viewed by others
References
Boonham, N., Smith, P., Walsh, K., Tame, J., Morris, J., Spence, N., Bennison, J., & Barker, I. (2002). The detection of Tomato spotted wilt virus (TSWV) in individual thrips using real time fluorescent RT-PCR (TaqMan). Journal of Virological Methods, 101, 37–48.
Bucher, E., Sijen, T., de Haan, P., Goldbach, R., & Prins, M. (2003). Negative-strand tospoviruses and tenuiviruses carry a gene for a suppressor of gene silencing at analogous genomic positions. Journal of Virology, 77, 1329–1336.
Chen, C. C., & Chiu, R. J. (1996). A tospovirus infecting peanut in Taiwan. Acta Horticulturae, 431, 57–67.
Chen, T. C., & Jan, F.-J. (2015). Tomato spotted wilt. In P. Tennant & G. Fermin (Eds.), Virus diseases of tropical and subtropical crops (pp. 162–176). Oxfordshire: CAB International.
Chen, C. C., Chen, T. C., Lin, Y. H., Yeh, S. D., & Hsu, H. T. (2005a). A chlorotic spot disease on calla lilies (Zantedeschia spp.) is caused by a tospovirus serologically but distantly related to Watermelon silver mottle virus. Plant Disease, 89, 440–445.
Chen, T. C., Huang, C. W., Liu, F. L., Hsu, H. T., Jain, R. K., Lin, C. H., & Yeh, S. D. (2005b). Purification and serological analyses of tospoviral nucleocapsid proteins expressed by Zucchini yellow mosaic virus vector. Journal of Virological Methods, 129, 113–124.
Chen, T. C., Lu, Y. Y., Cheng, Y. H., Chang, C. A., & Yeh, S. D. (2008). Melon yellow spot virusin watermelon: a first record from Taiwan. Plant Pathology, 54, 765.
Chen, T. C., Lu, Y. Y., Cheng, Y. H., Li, J. T., Yeh, Y. C., Kang, Y. C., Chang, C. P., Huang, L. H., Peng, J. C., & Yeh, S. D. (2010). Serological relationship between Melon yellow spot virus and Watermelon silver mottle virus and differential detection of the two viruses in cucurbits. Archives of Virology, 155, 1085–1095.
Chen, T. C., Li, J. T., Lin, Y. P., Yeh, Y. C., Kang, Y. C., Huang, L. H., & Yeh, S. D. (2012). Genomic characterization of Calla lily chlorotic spot virus and design of broad-spectrum primers for detection of tospoviruses. Plant Pathology, 61, 183–194.
Chen, X., Xu, X., Li, Y., & Liu, Y. (2013). Development of a real-time fluorescent quantitative PCR assay for detection of Impatiens necrotic spot virus. Journal of Virological Methods, 189, 299–304.
Chen, S. M., Wang, Y. C., Wu, P. R., & Chen, T. C. (2014). Production of antiserum against the nucleocapsid protein of Tomato spotted wilt virus and investigation of its serological relationship with other tospoviruses. Plant Protection Bulletin, 56, 55–74 (In Chinese).
Chen, T. C., Tsai, W. T., Kang, Y. C., Wang, Y. C., & Yeh, S. D. (2016a). Using monoclonal antibodies against the common epitopes of NSs proteins for the prompt detection and differentiation of tospoviruses prevalent in Euro-America and Asia regions. European Journal of Plant Pathology, 144, 509–524.
Chen, Y. H., Dong, J., Chien, W. C., Zheng, K., Wu, K., Yeh, S. D., Sun, J. H., Wang, Y. C., & Chen, T. C. (2016b). Monoclonal antibodies for differentiating infections of three serological-related tospoviruses prevalent in Southwestern China. Virology Journal, 13, 72.
Cheng, Y. H., Zheng, Y. X., Tai, C. H., Yen, J. H., Chen, Y. K., & Jan, F.-J. (2014). Identification, characterization and detection of a new tospovirus on sweet pepper. Annals of Applied Biology, 164, 107–115.
Chu, F. H., Chao, C. H., Chung, M. H., Chen, C. C., & Yeh, S. D. (2001a). Completion of the genome sequence of Watermelon silver mottle virus and utilization of degenerate primers for detecting tospoviruses in five serogroups. Phytopathology, 91, 361–368.
Chu, F. H., Chao, C. H., Peng, Y. C., Lin, S. S., Chen, C. C., & Yeh, S. D. (2001b). Serological and molecular characterization of Peanut chlorotic fan-spot virus, a new species of the genus Tospovirus. Phytopathology, 91, 856–863.
Ciuffo, M., Tavella, L., Pacifico, D., Masenga, V., & Turina, M. (2008). A member of a new Tospovirus species isolated in Italy from wild buckwheat (Polygonum convolvulus). Archives of Virology, 153, 2059–2068.
Cortes, I., Livierations, I. C., Derks, A., Peters, D., & Kormelink, R. (1998). Molecular and serological characterization of iris yellow spot virus, a new and distinct tospovirus species. Phytopathology, 88, 1276–1282.
Cortez, I., Saaijer, J., Wongjkaew, K. S., Pereira, A. M., Goldbach, R., Peters, D., & Kormelink, R. (2001). Identification and characterization of a novel tospovirus species using a new RT-PCR approach. Archives of Virology, 146, 265–278.
Dai, H., Yang, C., Zhou, Q., & Yu, C. (2012). Development and comparative study of several PCR-based approaches for detection of Iris yellow spot virus (IYSV). Acta Phytopathologica Sinica, 42, 126–130.
de Avila, A. C., de Haan, P., Kormelink, R., Resende, R. de. O., Goldback, R. W., & Peter, D. (1993). Classification of tospoviruses based on phylogeny of nucleoprotein gene sequences. Journal of General Virology, 74, 153–159.
de Haan, P., Wagemakers, L., Peters, D., & Goldbach, R. W. (1990). The S RNA segment of Tomato spotted wilt virus has an ambisense character. Journal of General Virology, 71, 1001–1007.
de Haan, P., Kormelink, R., Resende, R. O., van Poelwijk, F., Peters, D., & Goldbach, R. (1991). Tomato spotted wilt virus L RNA encodes a putative RNA polymerase. Journal of General Virology, 72, 2207–2216.
de Oliveira, A. S., Melo, F. L., Inoue-Nagata, A. K., Nagata, T., Kitajima, E. W., & Resende, R. O. (2012). Characterization of Bean necrotic mosaic virus: a member of a novel evolutionary lineage within the genus Tospovirus. PLoS ONE, 7, e38634.
Debreczeni, D. E., Ruiz-Ruiz, S., Aramburu, J., Lopez, C., Belliure, B., Galipienso, L., Soler, S., & Rubio, L. (2011). Detection, discrimination and absolute quantitation of Tomato spotted wilt virus isolates using real time RT-PCR with TaqMan MGB probes. Journal of Virological Methods, 176, 32–37.
Dong, J. H., Yin, Y. Y., Fang, Q., McBeath, J. H., & Zhang, Z. K. (2013). A new tospovirus causing chlorotic ringspot on Hippeastrum sp. in China. Virus Genes, 46, 567–570.
Hassani-Mehraban, A., Saaijer, J., Peters, D., Goldbach, R. W., & Kormelink, R. (2005). A new tomato-infecting tospovirus from Iran. Phytopathology, 95, 852–858.
Hassani-Mehraban, A., Botermans, M., Verhoeven, J. T. J., Meekes, E., Saaijer, J., Peters, D., Goldbach, R., & Kormelink, R. (2010). A distinct tospovirus causing necrotic streak on Alstroemeria sp. in Colombia. Archives of Virology, 155, 423–428.
Kang, Y. C., Yeh, S. D., Liao, C. H., Chou, W. C., Liu, F. L., Dong, J. H., & Chen, T. C. (2014). Verification of serological relationship between two phylogenetically related peanut-infecting Tospovirus species. European Journal of Plant Pathology, 140, 815–828.
Kikkert, M., van Lent, J., Storms, M., Bodegom, P., Kormelink, R., & Goldbach, R. (1999). Tomato spotted wilt virus particle morphogenesis in plant cells. Journal of Virology, 73, 2288–2297.
King, A. M. Q., Lefkowitz, E., Adams, M. J., & Carstens, E. B. (2011). Virus taxonomy: Ninth report of the international committee on taxonomy of viruses (pp. 725–741). Amsterdam: Elsevier Academic Press.
Kormelink, R., Storms, M., Van Lent, J., Peters, D., & Goldbach, R. (1994). Expression and subcellular location of the NSm protein of Tomato spotted wilt virus (TSWV), a putative viral movement protein. Virology, 200, 56–65.
Law, M. D., & Moyer, J. W. (1990). A tomato spotted wilt-like virus with a serologically distinct N protein. Journal of General Virology, 71, 933–938.
Lewandowski, D. J., & Adkins, S. (2005). The tubule-forming NSm protein from Tomato spotted wilt virus complements cell-to-cell and long-distance movement of Tobacco mosaic virus hybrids. Virology, 342, 26–37.
Li, J. T., Yeh, Y. C., Yeh, S. D., Raja, J. A. J., Rajagopalan, P. A., Liu, L. Y., & Chen, T. C. (2011). Complete genomic sequence of watermelon bud necrosis virus. Archives of Virology, 156, 359–362.
Lima, R. N., De Oliveira, A. S., Leastro, M. O., Blawid, R., Nagata, T., Resende, R. O., & Melo, F. L. (2016). The complete genome of the tospovirus Zucchini lethal chlorosis virus. Virology Journal, 13, 123.
Lin, Y. H., Chen, T. C., Hsu, H. T., Liu, F. L., Chu, F. H., Chen, C. C., Lin, Y. Z., & Yeh, S. D. (2005). Serological comparison and molecular characterization for verification of Calla lily chlorotic spot virus as a new tospovirus species belonging to Watermelon silver mottle virus serogroup. Phytopathology, 95, 1482–1488.
Pang, S. Z., Slightom, J. L., & Gonsalves, D. (1993). The biological properties of a distinct tospovirus and sequence analysis of its S RNA. Phytopathology, 83, 728–733.
Pappu, H. R., Rosales, I. M., & Druffel, K. L. (2008). Serological and molecular assays for rapid and sensitive detection of Iris yellow spot virus infection of bulb and seed onion crops. Plant Disease, 92, 588–594.
Pappu, H. R., Jones, R. A., & Jain, R. K. (2009). Global status of tospovirus epidemics in diverse cropping systems: successes gained and challenges that lie ahead. Virus Research, 141, 219–236.
Roberts, C. A., Dietzgen, R. G., Heelan, L. A., & Maclean, D. J. (2000). Real-time RT-PCR fluorescent detection of Tomato spotted wilt virus. Journal of Virological Methods, 88, 1–8.
Shimomoto, Y., Kobayashi, K., & Okuda, M. (2014). Identification and characterization of Lisianthus necrotic ringspot virus, a novel distinct tospovirus species causing necrotic disease of lisianthus (Eustoma grandiflorum). Journal of General Plant Pathology, 80, 169–175.
Takeda, A., Sugiyama, K., Nagano, H., Mori, M., Kaido, M., Mise, K., Tsuda, S., & Okuno, T. (2002). Identification of a novel RNA silencing suppressor, NSs protein of Tomato spotted wilt virus. FEBS Letters, 532, 75–79.
Takeshita, M., Nagai, N., Okuda, M., Matsuura, S., Okuda, S., Furuya, N., & Tsuchiya, K. (2011). Molecular and biological characterization of Chrysanthemum stem necrosis virus isolates from distinct regions in Japan. European Journal of Plant Pathology, 131, 9–14.
Yeh, S. D., Lin, Y. C., Cheng, Y. H., Jih, C. L., Chen, M. J., & Chen, C. C. (1992). Identification of tomato spotted wilt-like virus infecting watermelon in Taiwan. Plant Disease, 76, 835–840.
Yin, Y., Zheng, K., Dong, J., Fang, Q., Wu, S., Wang, L., & Zhang, Z. (2014). Identification of a new tospovirus causing necrotic ringspot on tomato in China. Virology Journal, 11, 213.
Zheng, Y. X., Chen, C. C., Yang, C. J., Yeh, S. D., & Jan, F.-J. (2008). Identification and characterization of a tospovirus causing chlorotic ringspots on Phalaenopsis orchids. European Journal of Plant Pathology, 120, 199–209.
Zheng, Y. X., Huang, C. H., Cheng, Y. H., Kuo, F. Y., & Jan, F.-J. (2010). First report of Tomato spotted wilt virus in sweet pepper in Taiwan. Plant Disease, 94, 920.
Zhou, J., Kantartzi, S. K., Wen, R. H., Newman, M., Hajimorad, M. R., Rupe, J. C., & Tzanetakis, I. E. (2011). Molecular characterization of a new tospovirus infecting soybean. Virus Genes, 43, 289–295.
Acknowledgments
We are grateful to Dr. Chung-Jan Chang, emeritus professor of the University of Georgia, and Dr. Kuang-Ren Chung for their critical review of the manuscript. Our gratitude also goes to Michael Burton, Asia University. This study was partially supported by grants from the Bureau of Animal and Plant Health Inspection and Quarantine, Council of Agriculture, Executive Yuan, the Ministry of Science and Technology (MOST 105-2313-B-005-021-MY3) and the Ministry of Education, Taiwan, R.O.C. under the ATU plan. We thank the virus providers: Dr. R. Provvidenti for TSWV; Dr. Dennis Gonsalves for GRSV; Dr. James W. Moyer for INSV; Dr. Richard Kormelink for ANSV, IYSV and TYRV; Dr. Mitsuru Okuda for CSNV; Dr. Prem A. Rajagopalan for GBNV and WBNV; Dr. Ioannis E. Tzanetakis for SVNaV; Dr. Jiahong Dong for HCRV, TNSaV and TZSV; Dr. Massimo Turina for PolRSV; the DSMZ Plant Virus Collection, Germany for TCSV; and the permission of the Bureau of Animal and Plant Health Inspection and Quarantine, Council of Agriculture, Executive Yuan for the virus importation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study was funded by the Bureau of Animal and Plant Health Inspection and Quarantine, Council of Agriculture, Executive Yuan (106AS-9.5.1-BQ-B7), and the Ministry of Science and Technology (MOST 105-2313-B-005-021-MY3). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
Not applicable.
Electronic supplementary material
All data generated or analyzed during this study are included in this published article and its supplementary information files.
All data are fully available without restriction.
Supplementary Table S1
(DOCX 70 kb)
Supplementary Table S2
(PDF 1503 kb)
Supplementary Fig. S1
(DOCX 2027 kb)
Supplementary Fig. S2
(PDF 3761 kb)
Rights and permissions
About this article
Cite this article
Huang, KS., Li, SL., Sun, JH. et al. Development of a generic method for inspection of tospoviruses. Eur J Plant Pathol 150, 457–469 (2018). https://doi.org/10.1007/s10658-017-1295-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-017-1295-5