Abstract
Background
TOB1, a member of the transducer of erbB-2 /B-cell translocation gene family, was detected to be down-regulated in ESCC by RNA sequencing. TOB1-AS1, a head-to-head antisense lncRNA with TOB1, was down-regulated in several cancers. However, the roles of them in esophageal squamous cell carcinoma (ESCC) remained unclarified.
Aims
To investigate the roles and functions of TOB1-AS1 and TOB1 in ESCC tumorigenesis.
Materials and Methods
The expression levels, methylation status, biological function and mechanisms of TOB1-AS1 and TOB1 in ESCC were, respectively, detected.
Results
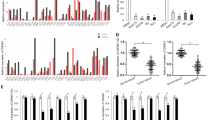
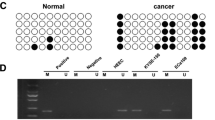
Frequent down-regulation of TOB1-AS1 and TOB1 was verified in esophageal cancer cells and ESCC tissues, and there was a positive correlation between them in ESCC tissues. The CpG sites hypermethylation within proximal promoter of TOB1-AS1 and TOB1 could lead to transcriptional inhibition of both genes. Furthermore, expression and proximal promoter methylation status of TOB1-AS1 or TOB1 may be associated with ESCC patients’ prognosis. TOB1-AS1 and TOB1 may function as tumor suppressors by inhibiting growth, migration, and invasion of esophageal cancer cells. Up-regulation of TOB1-AS1 increased expression level of TOB1, and TOB1-AS1 could work as a ceRNA to modulate ATF3 expression via competitively binding with miR-103a-2-5p. Meanwhile, ATF3, as a transcription factor, could regulate transcription of TOB1; down-regulation of TOB1-AS1 in ESCC led to decreased expression of ATF3 through ceRNA mechanism, and further influenced the transcription of TOB1.
Conclusion
TOB1-AS1 and TOB1 may act as tumor suppressors and may serve as potential targets for antitumor therapy in ESCC.







Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68:394–424.
Lagergren J, Smyth E, Cunningham D, Lagergren P. Oesophageal cancer. Lancet 2017;390:2383–2396.
Prabhu A, Obi KO, Rubenstein JH. The synergistic effects of alcohol and tobacco consumption on the risk of esophageal squamous cell carcinoma: a meta-analysis. Am J Gastroenterol 2014;109:822–827.
Chen Y, Tong Y, Yang C, Gan Y, Sun H et al. Consumption of hot beverages and foods and the risk of esophageal cancer: a meta-analysis of observational studies. BMC Cancer 2015;15:449.
Wu C, Wang Z, Song X, Feng XS, Abnet CC et al. Joint analysis of three genome-wide association studies of esophageal squamous cell carcinoma in Chinese populations. Nat Genet 2014;46:1001–1006.
Slack FJ, Chinnaiyan AM. The Role of Non-coding RNAs in Oncology. Cell 2019;179:1033–1055.
Bach DH, Lee SK. Long noncoding RNAs in cancer cells. Cancer Lett 2018;419:152–166.
Chen L, Dzakah EE, Shan G. Targetable long non-coding RNAs in cancer treatments. Cancer Lett 2018;418:119–124.
Rosikiewicz W, Makałowska I. Biological functions of natural antisense transcripts. Acta Biochim Pol 2016;63:665–673.
Faghihi MA, Wahlestedt C. Regulatory roles of natural antisense transcripts. Nat Rev Mol Cell Biol 2009;10:637–643.
Wanowska E, Kubiak MR, Rosikiewicz W, Makałowska I, Szcześniak MW. Natural antisense transcripts in diseases: From modes of action to targeted therapies. Wiley Interdiscip Rev RNA 2018;9:e1461.
Wang W, Wei C, Li P, Wang L, Li W et al. Integrative analysis of mRNA and lncRNA profiles identified pathogenetic lncRNAs in esophageal squanous cell carcinoma. Gene 2018;661:169–175.
Yao J, Li Z, Yang Z, Xue H, Chang H et al. Long noncoding RNA TOB1-AS1, an epigenetically silenced gene, functioned as a novel tumor suppressor by sponging miR-27b in cervical cancer. Am J Cancer Res 2018;8:1483–1498.
Jiang K, Zhi XH, Ma YY, Zhou LQ. Long non-coding RNA TOB1-AS1 modulates cell proliferation, apoptosis, migration and invasion through miR-23a/NEU1 axis via Wnt/b-catenin pathway in gastric cancer. Eur Rev Med Pharmacol Sci 2019;23:9890–9899.
Shangguan WJ, Liu HT, Que ZJ, Qian FF, Liu LS et al. TOB1-AS1 suppresses non-small cell lung cancer cell migration and invasion through a ceRNA network. Exp Ther Med 2019;18:4249–4258.
Guan R, Peng L, Wang D, He H, Wang D et al. Decreased TOB1 expression and increased phosphorylation of nuclear TOB1 promotes gastric cancer. Oncotarget 2017;8:75243–75253.
Guo H, Zhang R, Afrifa J, Wang Y, Yu J. Decreased expression levels of DAL-1 and TOB1 are associated with clinicopathological features and poor prognosis in gastric cancer. Pathol Res Pract 2019;215:152403.
Bai Y, Qiao L, Xie N, Li Y, Nie Y et al. TOB1 suppresses proliferation in K-Ras wild-type pancreatic cancer. Cancer Med 2020;9:1503–1514.
O’Malley S, Su H, Zhang T, Ng C, Ge H et al. TOB suppresses breast cancer tumotigenesis. Int J Cancer 2009;125:1805–1813.
Iwanaga K, Sueoka N, Sato A, Sakuragi T, Sakao Y et al. Alteration of expression or phosphorylation status of tob, a novel tumor suppressor gene product, is an early event in lung cancer. Cancer Lett 2003;202:71–79.
Wang J, Dong S, Zhang J, Jing D, Wang W et al. LncRNA NR2F1-AS1 regulates miR-371a-3p/TOB1 axis to suppress the proliferation of colorectal cancer cells. Cancer Biother Radiopharm 2020;35:760–764.
Li D, Xiao L, Ge Y, Fu Y, Zhang W et al. High expression of Tob1 indicates poor survival outcome and promotes tumour progression via a Wnt positive feedback loop in colon cancer. Mol Cancer 2018;17:159.
Cameron EE, Bachman KE, Myöhänen S, Herman JG, Baylin SB. Synergy of demethylation and histone deacetylase inhibition in the re-expression of genes silenced in cancer. Nat Genet 1999;21:103–107.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001;25:402–408.
Umemoto M, Yokoyama Y, Sato S, Tsuchida S, Al-Mulla F et al. Carbonyl reductase as a significant predictor of survival and lymph node metastasis in epithelial ovarian cancer. Br J Cancer 2001;85:1032–1036.
Yu L, Liu C, Vandeusen J, Becknell B, Dai Z et al. Global assessment of promoter methylation in a mouse model of cancer identifies ID4 as a putative tumor-suppressor gene in human leukemia. Nat Genet 2005;37:265–274.
Yuan JH, Yang F, Wang F, Ma JZ, Guo YJ et al. A long noncoding RNA activated by TGF-β promotes the invasion-metastasis cascade in hepatocellular carcinoma. Cancer Cell 2014;25:666–681.
Pelechano V, Steinmetz LM. Gene regulation by antisense transcription. Nat Rev Genet 2013;14:880–893.
Liang H, Tang Y, Zhang H, Zhang C. MiR-32-5p regulates radiosensitization, migration and invasion of colorectal cancer cells by targeting TOB1 gene. Onco Targets Ther 2019;12:9651–9661.
Zinad HS, Natasya I, Werner A. Natural antisense transcripts at the interface between host genome and mobile genetic elements. Front Microbiol 2017;8:2292.
Huang B, Song JH, Cheng Y, Abraham JM, Ibrahim S et al. Long non-coding antisense RNA KRT7-AS is activated in gastric cancers and supports cancer cell progression by increasing KRT7 expression. Oncogene 2016;35:4927–4936.
Modarresi F, Faghihi MA, Lopez-Toledano MA, Fatemi RP, Magistri M et al. Inhibition of natural antisense transcripts in vivo results in gene-specific transcriptional upregulation. Nat Biotechnol 2012;30:453–459.
Xie JJ, Xie YM, Chen B, Pan F, Guo JC et al. ATF3 functions as a novel tumor suppressor with prognostic significance in esophageal squamous cell carcinoma. Oncotarget 2014;5:8569–8582.
Acknowledgments
This study was supported by Grants from the National Natural Science Foundation of China (No. 81772612), Natural Science Foundation of Hebei Province (No. H2020206363).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10620_2022_7664_MOESM1_ESM.tif
Supplementary figure 1. Influence of knocking down TOB1-AS1 and TOB1 on proliferation, migration, and invasion ability of KYSE170 cells. A. Knock-down efficiency of TOB1-AS1 siRNAs transfection in KYSE170 cells. B. Influence of siRNA-TOB1-AS1-1 transfection on KYSE170 cells viability. C. Influence of siRNA-TOB1-AS1-1 transfection on KYSE170 cells migration ability. D. Influence of siRNA-TOB1-AS1-1 transfection on KYSE170 cells invasion ability. E. Knock-down efficiency of TOB1 siRNAs transfection in KYSE170 cells. F. Influence of siRNA-TOB1-1 transfection on KYSE170 cells viability. G. Influence of siRNA-TOB1-1 transfection on KYSE170 cells migration ability. H. Influence of siRNA-TOB1-1 transfection on KYSE170 cells invasion ability. * P < 0.05. (TIF 140 kb)
10620_2022_7664_MOESM2_ESM.tif
Supplementary figure 2. The expression levels of miR-103a-2-5p in databases. A. The expression level of miR-103a-2-5p in different kinds of tumor types in ONCOMIR database. B. The expression level of miR-103a-2-5p in different kinds of tumor types in YM500v2 database. (TIF 250 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dong, Z., Zhang, G., Lu, J. et al. Methylation Mediated Downregulation of TOB1-AS1 and TOB1 Correlates with Malignant Progression and Poor Prognosis of Esophageal Squamous Cell Carcinoma. Dig Dis Sci 68, 1316–1331 (2023). https://doi.org/10.1007/s10620-022-07664-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-022-07664-9