Abstract
Introduction
Cancer-associated fibroblasts (CAFs), as the activated fibroblasts in tumor stroma, are important modifiers of tumor progression. TGFβ1 has been the mostly accepted factor to fuel normal fibroblasts transformation into CAFs. Ca2+/calmodulin-dependent protein kinase II (CaMKII) is thought to play an important role in fibroblasts activation induced by TGFβ1. The aim of this study is to investigate the potential role of CaMKII in TGFβ1-induced fibroblasts activation and CAF-like differentiation. Cross talk between CaMKII-dependent fibroblasts and colon cancer in colon cancer progression also was addressed
Results
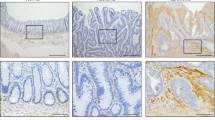
Immunostaining demonstrated that in colon cancer stroma, CaMKII overexpressed in stromal CAFs. In vitro, TGFβ1 increased CAF markers expression in human colon fibroblasts CCD-18Co, but not in CaMKII depletion fibroblasts. CaMKII knockdown by CaMKII shRNA significantly inhibited TGFβ1-induced fibroblasts activation and CAF-like differentiation. Smad3, AKT, and MAPK were targeted in TGFβ1–CaMKII-mediated pathway. Human colon cancer cell line HCT-116 activated fibroblasts directly, whereas CaMKII depletion dragged CCD-18Co fibroblasts undergoing CAF-associated trans-differentiation. Furthermore, increased proliferation, migration, and invasion of colon cancer cells were stimulated when co-cultured with normal fibroblasts, but not with CaMKII depletion fibroblasts.
Conclusions
These findings provide evidence that CaMKII is a critical mediator in TGFβ1-induced fibroblasts activation and is involved in the cross talk with colon cancer cells. CaMKII is a potentially effective target for future treatment of colon cancer.






Similar content being viewed by others
Abbreviations
- CaMKII:
-
Ca2+/calmodulin-dependent protein kinase II
- TGFβ1:
-
Transforming growth factor β1
- CAF:
-
Cancer-associated fibroblast
- CRC:
-
Colorectal cancer
- α-SMA:
-
α-Smooth muscle actin
- FAP:
-
Fibroblast activation protein
- FN:
-
Fibronectin
- PDGF:
-
Platelet-derived growth factor
- EGF:
-
Epidermal growth factor
- FGF:
-
Fibroblast growth factors
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–E386.
Lan YT, Yang SH, Chang SC, Liang WY, Li AF et al. Analysis of the seventh edition of American Joint Committee on colon cancer staging. Int J Colorectal Dis. 2012;27:657–663.
Bauer M, Su G, Casper C, He R, Rehrauer W et al. Heterogeneity of gene expression in stromal fibroblasts of human breast carcinomas and normal breast. Oncogene. 2010;29:1732–1740.
Navab R, Strumpf D, Bandarchi B, Zhu CQ, Pintilie M et al. Prognostic gene-expression signature of carcinoma-associated fibroblasts in non-small cell lung cancer. Proc Natl Acad Sci U S A. 2011;108:7160–7165.
Shiga K, Hara M, Nagasaki T, Sato T, Takahashi H et al. Cancer-associated fibroblasts: their characteristics and their roles in tumor growth. Cancers (Basel). 2015;7:2443–2458.
Yamamura Y, Asai N, Enomoto A, Kato T, Mii S et al. Akt-Girdin signaling in cancer-associated fibroblasts contributes to tumor progression. Cancer Res. 2015;75:813–823.
Franco OE, Shaw AK, Strand DW, Hayward SW. Cancer associated fibroblasts in cancer pathogenesis. Semin Cell Dev Biol. 2010;21:33–39.
Zhao H, Peehl DM. Tumor-promoting phenotype of CD90hi prostate cancer-associated fibroblasts. Prostate. 2009;69:991–1000.
Leung CS, Yeung TL, Yip KP, Pradeep S, Balasubramanian L et al. Calcium-dependent FAK/CREB/TNNC1 signalling mediates the effect of stromal MFAP5 on ovarian cancer metastatic potential. Nat Commun. 2014;5:5092.
Wang YY, Zhao R, Zhe H. The emerging role of CaMKII in cancer. Oncotarget. 2015;6:11725–11734.
Li B, Wang JH. Fibroblasts and myofibroblasts in wound healing: force generation and measurement. J Tissue Viability. 2011;20:108–120.
Rahaman SO, Grove LM, Paruchuri S, Southern BD, Abraham S et al. TRPV4 mediates myofibroblast differentiation and pulmonary fibrosis in mice. J Clin Invest. 2014;124:5225–5238.
Mukherjee S, Ayaub EA, Murphy J, Lu C, Kolb M et al. Disruption of calcium signaling in fibroblasts and attenuation of bleomycin-induced fibrosis by nifedipine. Am J Respir Cell Mol Biol. 2015;53:450–458.
Janssen LJ, Mukherjee S, Ask K. Calcium homeostasis and ionic mechanisms in pulmonary fibroblasts. Am J Respir Cell Mol Biol. 2015;53:135–148.
Chai S, Qian Y, Tang J, Liang Z, Zhang M et al. Ca(2+)/calmodulin-dependent protein kinase IIgamma, a critical mediator of the NF-kappaB network, is a novel therapeutic target in non-small cell lung cancer. Cancer Lett. 2014;344:119–128.
Britschgi A, Bill A, Brinkhaus H, Rothwell C, Clay I et al. Calcium-activated chloride channel ANO1 promotes breast cancer progression by activating EGFR and CAMK signaling. Proc Natl Acad Sci U S A. 2013;110:E1026-1034.
Wang T, Guo S, Liu Z, Wu L, Li M et al. CAMK2N1 inhibits prostate cancer progression through androgen receptor-dependent signaling. Oncotarget. 2014;5:10293–10306.
Wang C, Li N, Liu X, Zheng Y, Cao X. A novel endogenous human CaMKII inhibitory protein suppresses tumor growth by inducing cell cycle arrest via p27 stabilization. J Biol Chem. 2008;283:11565–11574.
Polanska UM, Orimo A. Carcinoma-associated fibroblasts: non-neoplastic tumour-promoting mesenchymal cells. J Cell Physiol. 2013;228:1651–1657.
Giannoni E, Bianchini F, Masieri L, Serni S, Torre E et al. Reciprocal activation of prostate cancer cells and cancer-associated fibroblasts stimulates epithelial-mesenchymal transition and cancer stemness. Cancer Res. 2010;70:6945–6956.
Calon A, Tauriello DV, Batlle E. TGF-beta in CAF-mediated tumor growth and metastasis. Semin Cancer Biol. 2014;25:15–22.
De Wever O, Demetter P, Mareel M, Bracke M. Stromal myofibroblasts are drivers of invasive cancer growth. Int J Cancer. 2008;123:2229–2238.
Hinz B, Phan SH, Thannickal VJ, Prunotto M, Desmouliere A et al. Recent developments in myofibroblast biology: paradigms for connective tissue remodeling. Am J Pathol. 2012;180:1340–1355.
Coleman DT, Gray AL, Stephens CA, Scott ML, Cardelli JA. Repurposed drug screen identifies cardiac glycosides as inhibitors of TGF-beta-induced cancer-associated fibroblast differentiation. Oncotarget. 2016;7:32200–32209.
Karagiannis GS, Poutahidis T, Erdman SE, Kirsch R, Riddell RH et al. Cancer-associated fibroblasts drive the progression of metastasis through both paracrine and mechanical pressure on cancer tissue. Mol Cancer Res. 2012;10:1403–1418.
Mazzocca A, Fransvea E, Dituri F, Lupo L, Antonaci S et al. Down-regulation of connective tissue growth factor by inhibition of transforming growth factor beta blocks the tumor-stroma cross-talk and tumor progression in hepatocellular carcinoma. Hepatology. 2010;51:523–534.
Henriksson ML, Edin S, Dahlin AM, Oldenborg PA, Oberg A et al. Colorectal cancer cells activate adjacent fibroblasts resulting in FGF1/FGFR3 signaling and increased invasion. Am J Pathol. 2011;178:1387–1394.
Evans RA, Tian YC, Steadman R, Phillips AO. TGF-beta1-mediated fibroblast-myofibroblast terminal differentiation-the role of Smad proteins. Exp Cell Res. 2003;282:90–100.
Midgley AC, Rogers M, Hallett MB, Clayton A, Bowen T et al. Transforming growth factor-beta1 (TGF-beta1)-stimulated fibroblast to myofibroblast differentiation is mediated by hyaluronan (HA)-facilitated epidermal growth factor receptor (EGFR) and CD44 co-localization in lipid rafts. J Biol Chem. 2013;288:14824–14838.
Kuperwasser C, Chavarria T, Wu M, Magrane G, Gray JW et al. Reconstruction of functionally normal and malignant human breast tissues in mice. Proc Natl Acad Sci U S A. 2004;101:4966–4971.
Wen S, Niu Y, Yeh S, Chang C. BM-MSCs promote prostate cancer progression via the conversion of normal fibroblasts to cancer-associated fibroblasts. Int J Oncol. 2015;47:719–727.
Eberlein C, Rooney C, Ross SJ, Farren M, Weir HM et al. E-Cadherin and EpCAM expression by NSCLC tumour cells associate with normal fibroblast activation through a pathway initiated by integrin alphavbeta6 and maintained through TGFbeta signalling. Oncogene. 2015;34:704–716.
Calon A, Espinet E, Palomo-Ponce S, Tauriello DV, Iglesias M et al. Dependency of colorectal cancer on a TGF-beta-driven program in stromal cells for metastasis initiation. Cancer Cell. 2012;22:571–584.
Zayzafoon M. Calcium/calmodulin signaling controls osteoblast growth and differentiation. J Cell Biochem. 2006;97:56–70.
Bourguignon LY, Gilad E, Brightman A, Diedrich F, Singleton P. Hyaluronan-CD44 interaction with leukemia-associated RhoGEF and epidermal growth factor receptor promotes Rho/Ras co-activation, phospholipase C epsilon-Ca2+ signaling, and cytoskeleton modification in head and neck squamous cell carcinoma cells. J Biol Chem. 2006;281:14026–14040.
Macias MJ, Martin-Malpartida P, Massague J. Structural determinants of Smad function in TGF-beta signaling. Trends Biochem Sci. 2015;40:296–308.
Piersma B, Bank RA, Boersema M. Signaling in fibrosis: TGF-beta, WNT, and YAP/TAZ Converge. Front Med (Lausanne). 2015;2:59.
Acknowledgments
This work was supported by National Natural Science Foundation of China (81302131), Medical Scientific Research Foundation of Guangdong Province (A2015297), and Shenzhen Science and Technology Project (JCYJ20150402152130164).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, W., Chen, Y., Su, J. et al. CaMKII Mediates TGFβ1-Induced Fibroblasts Activation and Its Cross Talk with Colon Cancer Cells. Dig Dis Sci 67, 134–145 (2022). https://doi.org/10.1007/s10620-021-06847-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-06847-0