Abstract
Introduction
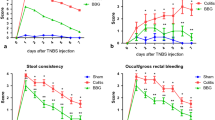
Our work analyzed the effects of a P2X7 receptor antagonist, Brilliant Blue G (BBG), on rat ileum myenteric plexus following ischemia and reperfusion (ISR) induced by 45 min of ileal artery occlusion with an atraumatic vascular clamp with 24 h (ISR 24-h group) or 14 d of reperfusion (ISR 14-d group).
Material and methods
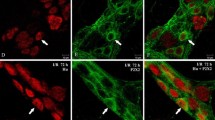
Either BBG (50 mg/kg or 100 mg/kg, BBG50 or BBG100 groups) or saline (vehicle) was administered subcutaneously 1 h after ischemia in the ISR 24-h group or once daily for the 5 d after ischemia in the ISR 14-d group (n = 5 per group). We evaluated the neuronal density and profile area by examining the number of neutrophils in the intestinal layers, protein expression levels of the P2X7 receptor, intestinal motility and immunoreactivity for the P2X7 receptor, nitric oxide synthase, neurofilament-200, and choline acetyl transferase in myenteric neurons.
Results
The neuronal density and profile area were restored by BBG following ISR. The ischemic groups showed alterations in P2X7 receptor protein expression and the number of neutrophils in the intestine and decreased intestinal motility, all of which were recovered by BBG treatment.
Conclusion
We concluded that ISR morphologically and functionally affected the intestine and that its effects were reversed by BBG treatment, suggesting the P2X7 receptor as a therapeutic target.









Similar content being viewed by others
References
Furness JB. The enteric nervous system. Oxford: Blackwell; 2006.
Furness JB. The enteric nervous system and neurogastroenterology. Nat Rev Gastroenterol Hepatol. 2012;9:286–294.
Guan Y, Worrell RT, Pritts TA, Montrose MH. Intestinal ischemia–reperfusion injury: reversible and irreversible damage imaged in vivo. Am J Physiol Gastrointest Liver Physiol. 2009;297:187–196.
Haglund U, Bergqvist D. Intestinal ischemia—the basics. Langenbeck’s Arch Surg. 1999;384:233–238.
Oldenburg A, Lau LL, Rodenberg TJ, Edmonds HJ, Burger CD. Acute mesenteric ischemia. A clinical review. Arc Intern Med. 2004;164:1054–1061.
Lindestrom L, Ekblad E. Structural and neuronal changes in rat ileum after ischemia with reperfusion. Dig Dis Sci. 2004;49:1212–1222. https://doi.org/10.1023/B:DDAS.0000037815.63547.08.
Paulino AS, Palombit K, Cavriani G, et al. Effects of ischemia and reperfusion on P2X2 receptor expressing neurons of the rat ileum enteric nervous system. Dig Dis Sci. 2011;56:2262–2277. https://doi.org/10.1007/s10620-011-1588-z.
Palombit K, Mendes CE, Tavares-De-Lima W, Castelucci P. Effects of ischemia and reperfusion on subpopulations of rat enteric neurons expressing the P2X7 receptor. Dig Dis Sci. 2013;58:3429–3439. https://doi.org/10.1007/s10620-013-2847-y.
Marosti AR, da Silva MV, Palombit K, Mendes CE, Tavares-de-Lima W, Castelucci P. Differential effects of intestinal ischemia and reperfusion in rat enteric neurons and glial cells expressing P2X2 receptors. Histol Histopathol. 2015;30:489–501.
Mendes CE, Palombit K, Vieira C, Silva I, Correia-de-Sá P, Castelucci P. The effect of ischemia and reperfusion on enteric glial cells and contractile activity in the ileum. Dig Dis Sci. 2015;60:2677–2689. https://doi.org/10.1007/s10620-015-3663-3.
Ralevic V, Burnstock G. Receptors for purines and pyrimidines. Pharmacol Rev. 1998;50:413–492.
Burnstock G. A basis for distinguishing two types of purinergic receptor. In: Straub RW, Bolis L, eds. Cell membrane receptors for drugs and hormones. A multidisciplinary approach. New York: Raven Press; 1978:107–118.
North RA, Surprenant A. Pharmacology of cloned P2X receptors. Annu Rev Pharmacol Toxicol. 2000;40:563–580.
North A. Molecular physiology of P2X receptors. Physiol Rev. 2002;82:1013–1067.
Burnstock G, Kennedy C. Is there a basis for distinguishing two types of P2-purinoceptor? Gen Pharmacol. 1985;16:433–440.
Galligan JJ. Ligand-gated ion channels in the enteric nervous system. Neurogastroenterol Motil. 2002;14:611–623.
Abbrachio MP, Burnstock G, Verkhratsky A, Zimmermann H. Purinergic signaling in the nervous system: an overview. Trends Neurosci. 2009;32:19–29.
Burnstock G. Purinergic signalling in the gastrointestinal tract and related organs in health and disease. Purinergic Signal. 2014;10:3–50.
Franke H, Krüel U, Illes P. P2 receptors and neuronal injury. Euro J Physiol. 2006;452:622–644.
Sperlágh B, Illes P. P2X7 receptor, an emerging target in central nervous system diseases. Trends Pharmacol Sci. 2014;35:537–547.
Vulchanova L, Arvidsson U, Riedl M, et al. Differential distribution of two ATP-gated ion channels (P2X receptors) determined by immunohistochemistry. Proc Natl Acad Sci. 1996;93:8063–8067.
Hu HZ, Gao N, Lin Z, et al. P2X7 receptors in the enteric nervous system of guinea-pig small intestine. J Comp Neurol. 2001;440:299–310.
Castelucci P, Robbins HL, Poole DP, Furness JB. The distribution of purine P2X2 receptors in the guinea pig enteric nervous system. Histochem Cell Biol. 2002;117:415–422.
Poole DP, Castelucci P, Robbins HL, Chiocchetti R, Furness JB. The distribution of P2X3 purine receptor subunits in the guinea-pig enteric nervous system. Auton Neurosci. 2002;101:39–47.
Van Nassauw L, Brouns I, Adraensen D, Burnstock G, Timmermans JP. Neurochemical identification of enteric neurons expressing P2X(3) receptors in the guinea-pig ileum. Histochem Cell Biol. 2002;118:193–203.
Xiang Z, Burnstock G. Distribution of P2Y2 receptors in the guinea pig enteric nervous system and its coexistence with P2X2 and P2X3 receptors, neuropeptide Y, nitric oxide synthase and calretinin. Histochem Cell Biol. 2005;124:379–390.
Xiang Z, Burnstock G. P2X2 and P2X3 purinoceptors in the rat enteric nervous system. Histochem Cell Biol. 2004;12:169–179.
Yu Q, Zhao Z, Sun J, Guo W, Fu J, Burnstock G. Expression of P2X6 receptors in the enteric nervous system of the rat gastrointestinal tract. Histochem Cell Biol. 2010;133:177–188.
Giaroni C, Knight GE, Ruan H-Z, et al. P2 receptors in the murine gastrointestinal tract. Neuropharmacology. 2002;43:1313–1323.
Ruan HZ, Burnstock G. The distribution of P2X5 purinergic receptors in the enteric nervous system of mouse. Cell Tissue Res. 2005;319:191–200.
Sperlágh B, Vizi ES, Wirkner K, Illes P. P2X7 receptors in the nervous system. Prog Neurobiol. 2006;78:327–346.
Burnstock G. An introduction to the roles of purinergic signalling in neurodegeneration, neuroprotection and neuroregeneration. Neuropharmacology. 2016;104:4–17.
Girotti PA, Misawa R, Palombit K, Mendes CE, Bittencourt JC, Castelucci P. Differential effects of undernourishment on the differentiation and maturation of rat enteric neurons. Cell Tissue Res. 2013;353:367–380.
Da Silva MV, Marosti AR, Mendes CE, Palombit K, Castelucci P. Differential effects of experimental ulcerative colitis on P2X7 receptor expression in enteric neurons. Histochem Cell Biol. 2015;143:171–184.
Da Silva MV, Marosti AR, Mendes CE, Palombit K, Castelucci P. Submucosal neurons and enteric glial cells expressing the P2X7 receptor in rat experimental colitis. Acta Histochem. 2017;119:481–494.
Wang X, Arcuino G, Takano T, et al. P2X7 receptor inhibition improves recovery after spinal cord injury. Nat Med. 2004;10:821–827.
Volonté C, Apolloni S, Skaper SD, Burnstock G. P2X7 receptors, channels, pores and more. CNS Neurol Disord Drug Targets. 2012;11:705–721.
Jiang LH, Mackenzie AB, North RA, Surprenant A. Brilliant Blue G selectively blocks ATP-gated rat P2X7 receptors. Mol Pharmacol. 2000;58:82–88.
Remy M, Thaler S, Schumann RG, et al. An in vivo evaluation of Brilliant Blue G in animals and humans. Br J Ophtalmol. 2008;92:1142–1147.
Ryu JK, Mclarnon JG. Block of purinergic P2X(7) receptor is neuroprotective in an animal model of Alzheimer’s disease. Neuroreport. 2008;19:1715–1719.
Díaz-Hernández M, Díez-Zaera M, Sánchez-Nogueiro J, et al. Altered P2X7-receptor level and function in mouse models of Huntington’s disease and therapeutic efficacy of antagonist administration. FASEB J. 2009;23:1893–1906.
Peng W, Cotrina ML, Han X, et al. Systemic administration of an antagonist of the ATP-sensitive receptor P2X7 improves recovery after spinal cord injury. Proc Natl Acad Sci. 2009;106:12489–12493.
Arbeloa J, Pérez-Samartín A, Gottlieb M, Matute C. P2X7 receptor blockade prevents ATP excitotoxicity in neurons and reduces brain damage after ischemia. Neurobiol Dis. 2012;45:954–961.
Chu K, Yin B, Wang J, et al. Inhibition of P2X7 receptor ameliorates transient global cerebral ischemia/reperfusion injury via modulating inflammatory responses in the rat hippocampus. J. Neuroinflamm. 2012;18:69–75.
Rivera LR, Thacker M, Castelucci P, Poole DP, Frugier T, Furness JB. Knock out of neuronal nitric oxide synthase exacerbates intestinal ischemia/reperfusion injury in mice. Cell Tissue Res. 2012;349:565–576.
Franke H, Illes P. Involvement of P2 receptors in the growth and survival of neurons in the CNS. Pharmacol Ther. 2006;109:297–324.
Gulbransen BD, Bashashati M, Hirota SA, et al. Activation of neuronal P2X7 receptor–pannexin-1 mediates death of enteric neurons during colitis. Nat Med. 2012;18:600–604.
Roberts JA, Lukewich MK, Sharkey KA, Furness JB, Mawe GM, Lomax AE. The roles of purinergic signaling during gastrointestinal inflammation. Curr Opin Pharmacol. 2012;12:659–666.
Antonioli L, Giron MC, Colucci R, et al. The role of purinergic pathways in the pathophysiology of gut diseases, pharmacological modulation and potential therapeutic applications. Pharmacol Ther. 2013;139:157–188.
Coutinho-Silva R, Persechini PM, Bisaggio RD, et al. P2Z/P2X7 receptor-dependent apoptosis of dendritic cells. Am J Physiol. 1999;276:C1139–C1147.
Slater M, Barden JA, Murphy CR. The purinergic calcium channels P2X1,2,5,7 are down-regulated while P2X3,4,6 are up-regulated during apoptosis in the ageing rat prostate. Histochem J. 2000;32:571–580.
Castelucci P, De Souza RR, De Angelis RC, Furness JB, Liberti EA. Effects of pre- and postnatal protein deprivation and postnatal re-feeding on myenteric neurons of the rat large intestine: a quantitative morphological study. Cell Tissue Res. 2002;310:1–7.
Gomes OA, Castelucci P, Fontes RBV, Liberti EA. Effects of pre- and postnatal protein and postnatal re-feeding on myenteric neurons of the rat small intestine, a quantitative morphological study. Auton Neurosci. 2006;126:277–284.
Misawa R, Girotti PA, Mizuno MS, Liberti EA, Furness JB, Castelucci P. Effects of protein deprivation and re-feeding on P2X2 receptors in enteric neurons. World J Gastroenterol. 2010;16:3651–3663.
Mizuno MS, Crisma AR, Borelli P, Castelucci P. Expression of the P2X2 receptor in different classes of ileum myenteric neurons in the female obese ob/ob mouse. World J Gastroenterol. 2012;18:4693–4703.
Mizuno MS, Crisma AR, Borelli P, Schäfer BT, Silveira MP, Castelucci P. Distribution of the P2X2 receptor and chemical coding in ileal enteric neurons of obese male mice (ob/ob). World J Gastroenterol. 2014;20:13911–13919.
Rivera LR, Thacker M, Castelucci P, Bron R, Furness JB. The reactions of specific neuron types to intestinal ischemia in the guinea pig enteric nervous system. Acta Neuropathol. 2009;118:261–270.
Fellin T, Pozzan T, Carmignoto G. Purinergic receptors mediate two distinct glutamate release pathways in hippocampal astrocytes. J Biol Chem. 2006;281:4274–4284.
Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg. 1998;228:652–663.
Hierholzer C, Kalff JC, Audolfsson G, et al. Molecular and functional contractile sequelae of rat intestinal ischemia/reperfusion injury. Transplantation. 1999;68:1244–1254.
Massberg S, Messmer K. The nature of ischemia/reperfusion injury. Transplant Proc. 1998;30:4217–4223.
Filpa V, Carpanese E, Marchet S, et al. Nitric oxide regulates homeoprotein OTX1 and OTX2 expression in the rat myenteric plexus after intestinal ischemia–reperfusion injury. Am J Physiol Gastrointest Liver Physiol. 2017;312:G374–G389.
Pedata F, Dettori I, Coppi E, Melani A, Tweardy DJ, Bauer AJ. Purinergic signalling in brain ischemia. Neuropharmacology. 2016;104:105–130.
Morris GP, Beck PL, Herridge MS, et al. Hapten-induced model of chronic inflammation and ulceration in the rat colon. Gastroenterology. 1989;96:795–803.
Corriden R, Chen Y, Inoue Y, et al. GEcto-nucleoside triphosphate diphosphohydrolase 1 (E-NTPDase1/CD39) regulates neutrophil chemotaxis by hydrolyzing released ATP to adenosine. J Biol Chem. 2008;283:28480–28486.
Di Virgilio F. Liaisons dangereuses, P2X(7) and the inflammasome. Trends Pharmacol Sci. 2007;28:465–472.
Csölle C, Sperlágh B. Peripheral origin of IL-1beta production in the rodent hippocampus under in vivo systemic bacterial lipopolysaccharide (LPS) challenge and its regulation by P2X(7) receptors. J Neuroimmunol. 2010;219:38–46.
Hassoun HT, Weisbrodt NW, Mercer DW, Kozar RA, Mooy FG, Moore FA. Inducible nitric oxide synthase mediates gut ischemia/reperfusion-induced ileus only after severe insults. J Surg Res. 2001;97:150–154.
Takahashi A, Tomomasa TH, Kaneko H, et al. Intestinal motility in an in vivo rat model of intestinal ischemia–reperfusion with special reference to the effects of nitric oxide on the motility changes. J Pediatr Gastroenterol Nutr. 2001;33:283–288.
Taha MO, Miranda-Ferreira R, Paez RP, et al. Role of l-arginine, a substrate of nitric oxide biosynthesis, on intestinal ischemia–reperfusion in rabbits. Transplant Proc. 2010;42:448–450.
Caplan MS, Hedlund E, Hill N, Mackendrick W. The role of endogenous nitric oxide and platelet-activating factor in hypoxia-induced intestinal injury in rats. Gastroenteroly. 1994;106:346–352.
Ragy M, Elbassuoni E. The role of nitric oxide and L-type calcium channel blocker in the contractility of rabbit ileum in vitro. J Physiol Biochem. 2012;68:521–528.
Ballabeni V, Barocelli E, Bertoni S, Impicciatore M. Alterations of intestinal motor responsiveness in a model of mild mesenteric ischemia/reperfusion in rats. Life Sci. 2002;71:2025–2035.
Giaroni C, Marchet S, Carpanese E, et al. Role of neuronal and inducible nitric oxide synthases in the guinea pig ileum myenteric plexus during in vitro ischemia and reperfusion. Neurogastroenterol Motil. 2013;25:e114–e126.
Anitha M, Vijay-Kumar M, Sitaraman SV, Gewirtz AT, Srinivasan S. Gut microbial products regulate murine gastrointestinal motility via Toll-like receptor 4 signaling. Gastroenterology. 2012;143:1006.e4–1016.e4.
Caputi V, Marsilio I, Cerantola S, et al. Toll-like receptor 4 modulates small intestine neuromuscular function through nitrergic and purinergic pathways. Front Pharmacol. 2017;8:350. https://doi.org/10.3389/fphar.2017.00350.
Tran L, Greenwood-Van Meerveld B. In a non-human primate model, aging disrupts the neural control of intestinal smooth muscle contractility in a region-specific manner. Neurogastroenterol Motil. 2014;26:410–418.
Acknowledgments
We would like to thank Rosana Prisco for the statistical analysis. These studies were supported by São Paulo Foundation Research (FAPESP Grants Nos. 2010/10510-8, 2012/00259-1, 2014/25927-2) and CAPES.
Author information
Authors and Affiliations
Contributions
KP performed the experiments and analyzed the results. CEM helped with the immunohistochemistry study and ISR surgery. WTL helped edit the manuscript. MLBC helped in the Western blotting study. PC planned experiments, analyzed the results, and wrote and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Palombit, K., Mendes, C.E., Tavares-de-Lima, W. et al. Blockage of the P2X7 Receptor Attenuates Harmful Changes Produced by Ischemia and Reperfusion in the Myenteric Plexus. Dig Dis Sci 64, 1815–1829 (2019). https://doi.org/10.1007/s10620-019-05496-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-019-05496-8