Abstract
Background
Chemokine receptors are now known to play an important role in cancer growth and metastasis. However, there is little information regarding chemokine expression in gastric cancer. In this study, we examined CXCL12 expression in gastric cancer and also evaluated whether the down-regulation of CXCL12 is due to aberrant methylation of the gene.
Methods
CXCL12 expression was examined using real-time reverse-transcription polymerase chain reaction (RT-PCR), immunofluorescence, flow cytometry, and immunohistochemistry, and the methylation status of the gene was evaluated by methylation-specific PCR (MSP) in normal gastric and gastric cancer cell lines and 35 primary gastric carcinomas and corresponding nonmalignant gastric tissues.
Results
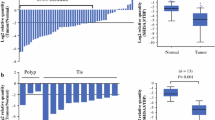
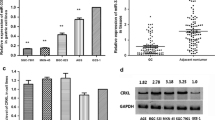
The down-regulation of CXCL12 was observed in gastric cancer cell lines and primary gastric carcinomas, while decreased expression of CXCL12 protein was significantly associated with lymph node metastasis and histological grade. And this down-regulation was found to be in accordance with aberrant methylation of the gene. Hypermethylation of the gene was observed in 65.7% (23 of 35) of the primary gastric carcinomas, while it was found in only 11.4% (4/35) of the corresponding nonmalignant tissues. Furthermore, CXCL12 expression was restored in gastric cancer cell lines after treatment with the demethylating agent, 5-aza-2′-deoxycytidine (5-Aza-dC), and demethylation of the highly metastatic cells SGC-7901 induced invasion suppression of the cells. For two CXCL12 receptors, CXCR4 and CXCR7, the mRNA levels remained almost unchanged with the 5-Aza-dC treatment.
Conclusions
Collectively, our results suggest that the aberrant methylation of CXCL12 frequently occurs in the down-regulation of CXCL12 in gastric cancers and that it may play a role in the metastasis of gastric cancer.





Similar content being viewed by others
Abbreviations
- 5-Aza-dC:
-
5-Aza-2′-deoxycytidine
- RT-PCR:
-
Reverse-transcription polymerase chain reaction
- MSP:
-
Methylation-specific PCR
References
Zlotnik A, Yoshie O. Chemokines: a new classification system and their role in immunity. Immunity. 2000;12:121–127. doi:10.1016/S1074-7613(00)80165-X.
Scotton CJ, Wilson JL, Milliken D, Stamp G, Balkwill FR. Epithelial cancer cell migration: a role for chemokine receptors? Cancer Res. 2001;61:4961–4965.
Koizumi K, Hojo S, Akashi T, Yasumoto K, Saiki I. Chemokine receptors in cancer metastasis and cancer cell-derived chemokines in host immune response. Cancer Sci. 2007;98:1652–1658. doi:10.1111/j.1349-7006.2007.00606.x.
Ben-Baruch A. The multifaceted roles of chemokines in malignancy. Cancer Metastasis Rev. 2006;25:357–371. doi:10.1007/s10555-006-9003-5.
Balkwill F. The significance of cancer cell expression of the chemokine receptor CXCR4. Semin Cancer Biol. 2004;14:171–179. doi:10.1016/j.semcancer.2003.10.003.
Ratajczak MZ, Zuba-Surma E, Kucia M, Reca R, Wojakowski W, Ratajczak J. The pleiotropic effects of the SDF-1-CXCR4 axis in organogenesis, regeneration and tumorigenesis. Leukemia. 2006;20:1915–1924. doi:10.1038/sj.leu.2404357.
Balkwill F. Cancer and the chemokine network. Nat Rev Cancer. 2004;4:540–550. doi:10.1038/nrc1388.
Zlotnik A. Chemokines in neoplastic progression. Semin Cancer Biol. 2004;14:181–185. doi:10.1016/j.semcancer.2003.10.004.
Salcedo R, Oppenheim JJ. Role of chemokines in angiogenesis: CXCL12/SDF-1 and CXCR4 interaction, a key regulator of endothelial cell responses. Microcirculation. 2003;10:359–370. doi:10.1038/sj.mn.7800200.
Luker KE, Luker GD. Functions of CXCL12 and CXCR4 in breast cancer. Cancer Lett. 2006;238:30–41. doi:10.1016/j.canlet.2005.06.021.
Wang J, Loberg R, Taichman RS. The pivotal role of CXCL12 (SDF-1)/CXCR4 axis in bone metastasis. Cancer Metastasis Rev. 2006;25:573–587. doi:10.1007/s10555-006-9019-x.
Dewan MZ, Ahmed S, Iwasaki Y, Ohba K, Toi M, Yamamoto N. Stromal cell-derived factor-1 and CXCR4 receptor interaction in tumor growth and metastasis of breast cancer. Biomed Pharmacother. 2006;60:273–276. doi:10.1016/j.biopha.2006.06.004.
Burger JA, Kipps TJ. CXCR4: a key receptor in the crosstalk between tumor cells and their microenvironment. Blood. 2006;107:1761–1767. doi:10.1182/blood-2005-08-3182.
Epstein RJ. The CXCL12-CXCR4 chemotactic pathway as a target of adjuvant breast cancer therapies. Nat Rev Cancer. 2004;4:901–909. doi:10.1038/nrc1473.
Zlotnik A. Involvement of chemokine receptors in organ-specific metastasis. Contrib Microbiol. 2006;13:191–199. doi:10.1159/000092973.
Burns JM, Summers BC, Wang Y, et al. A novel chemokine receptor for SDF-1 and I-TAC involved in cell survival, cell adhesion, and tumor development. J Exp Med. 2006;203:2201–2213. doi:10.1084/jem.20052144.
Wendt MK, Cooper AN, Dwinell MB. Epigenetic silencing of CXCL12 increases the metastatic potential of mammary carcinoma cells. Oncogene. 2008;27:1461–1471. doi:10.1038/sj.onc.1210751.
Wendt MK, Johanesen PA, Kang-Decker N, Binion DG, Shah V, Dwinell MB. Silencing of epithelial CXCL12 expression by DNA hypermethylation promotes colonic carcinoma metastasis. Oncogene. 2006;25:4986–4997. doi:10.1038/sj.onc.1209505.
Suzuki M, Mohamed S, Nakajima T, et al. Aberrant methylation of CXCL12 in non-small cell lung cancer is associated with an unfavorable prognosis. Int J Oncol. 2008;33:113–119.
Ramos EA, Camargo AA, Braun K, et al. Simultaneous CXCL12 and ESR1 CpG island hypermethylation correlates with poor prognosis in sporadic breast cancer. BMC Cancer. 2010;10:23. doi:10.1186/1471-2407-10-23.
Zhou W, Jiang Z, Liu N, et al. Down-regulation of CXCL12 mRNA expression by promoter hypermethylation and its association with metastatic progression in human breast carcinomas. J Cancer Res Clin Oncol. 2009;135:91–102. doi:10.1007/s00432-008-0435-x.
Yasumoto K, Koizumi K, Kawashima A, et al. Role of the CXCL12/CXCR4 axis in peritoneal carcinomatosis of gastric cancer. Cancer Res. 2006;66:2181–2187. doi:10.1158/0008-5472.CAN-05-3393.
Ishigami S, Natsugoe S, Okumura H, et al. Clinical implication of CXCL12 expression in gastric cancer. Ann Surg Oncol. 2007;14:3154–3158. doi:10.1245/s10434-007-9521-6.
Zieker D, Königsrainer I, Traub F, et al. PGK1 a potential marker for peritoneal dissemination in gastric cancer. Cell Physiol Biochem. 2008;21:429–436. doi:10.1159/000129635.
Zieker D, Königsrainer I, Tritschler I, et al. Phosphoglycerate kinase 1 a promoting enzyme for peritoneal dissemination in gastric cancer. Int J Cancer. 2010;126:1513–1520. doi:10.1002/ijc.24835.
Raggo C, Ruhl R, McAllister S, et al. Novel cellular genes essential for transformation of endothelial cells by Kaposi’s sarcoma-associated herpesvirus. Cancer Res. 2005;65:5084–5095. doi:10.1158/0008-5472.CAN-04-2822.
Richard CL, Tan EY, Blay J. Adenosine upregulates CXCR4 and enhances the proliferative and migratory responses of human carcinoma cells to CXCL12/SDF-1alpha. Int J Cancer. 2006;119:2044–2053. doi:10.1002/ijc.22084.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi:10.1006/meth.2001.1262.
Herman JG, Graff JR, Myöhänen S, Nelkin BD, Baylin SB. Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc Natl Acad Sci USA. 1996;93:9821–9826.
Müller A, Homey B, Soto H, et al. Involvement of chemokine receptors in breast cancer metastasis. Nature. 2001;410:50–56. doi:10.1038/35065016.
Taichman RS, Cooper C, Keller ET, Pienta KJ, Taichman NS, McCauley LK. Use of the stromal cell-derived factor-1/CXCR4 pathway in prostate cancer metastasis to bone. Cancer Res. 2002;62:1832–1837.
Oonakahara K, Matsuyama W, Higashimoto I, Kawabata M, Arimura K, Osame M. Stromal-derived factor-1alpha/CXCL12-CXCR 4 axis is involved in the dissemination of NSCLC cells into pleural space. Am J Respir Cell Mol Biol. 2004;30:671–677. doi:10.1165/rcmb.2003-0340OC.
Nagasawa T, Kikutani H, Kishimoto T. Molecular cloning and structure of a pre-B-cell growth-stimulating factor. Proc Natl Acad Sci USA. 1994;91:2305–2309.
Zou YR, Kottmann AH, Kuroda M, Taniuchi I, Littman DR. Function of the chemokine receptor CXCR4 in haematopoiesis and in cerebellar development. Nature. 1998;393:595–599. doi:10.1038/31269.
Dwinell MB, Eckmann L, Leopard JD, Varki NM, Kagnoff MF. Chemokine receptor expression by human intestinal epithelial cells. Gastroenterology. 1999;117:359–367. doi:S0016508599001535.
Yoshitake N, Fukui H, Yamagishi H, et al. Expression of SDF-1 alpha and nuclear CXCR4 predicts lymph node metastasis in colorectal cancer. Br J Cancer. 2008;98:1682–1689. doi:10.1038/sj.bjc.6604363.
Brand S, Dambacher J, Beigel F, et al. CXCR4 and CXCL12 are inversely expressed in colorectal cancer cells and modulate cancer cell migration, invasion and MMP-9 activation. Exp Cell Res. 2005;310:117–130. doi:10.1016/j.yexcr.2005.07.006.
Meng CF, Zhu XJ, Peng G, Dai DQ. Role of histone modifications and DNA methylation in the regulation of O6-methylguanine-DNA methyltransferase gene expression in human stomach cancer cells. Cancer Invest. 2010;28:331–339. doi:10.3109/07357900903179633.
Wendt MK, Drury LJ, Vongsa RA, Dwinell MB. Constitutive CXCL12 expression induces anoikis in colorectal carcinoma cells. Gastroenterology. 2008;135:508–517. doi:10.1053/j.gastro.2008.05.033.
Balabanian K, Lagane B, Infantino S, et al. The chemokine SDF-1/CXCL12 binds to and signals through the orphan receptor RDC1 in T lymphocytes. J Biol Chem. 2005;280:35760–35766. doi:10.1074/jbc.M508234200.
Drury LJ, Wendt MK, Dwinell MB. CXCL12 chemokine expression and secretion regulates colorectal carcinoma cell anoikis through bim-mediated intrinsic apoptosis. PLoS One. 2010;5:e12895. doi:10.1371/journal.pone.0012895.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant number: 30572162).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhi, Y., Chen, J., Zhang, S. et al. Down-Regulation of CXCL12 by DNA Hypermethylation and Its Involvement in Gastric Cancer Metastatic Progression. Dig Dis Sci 57, 650–659 (2012). https://doi.org/10.1007/s10620-011-1922-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1922-5