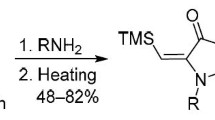
A convenient preparative method has been developed for obtaining a vinylacetylene phosphonate. The reaction of arylnitrile oxides with 4-(diethylphosphoryl)-2-methylbut-1-en-3-yne proceeds as a regioselective 1,3-dipolar cycloaddition exclusively at the terminal double bond of vinylacetylene phosphonate.
Similar content being viewed by others
References
J. S. D. Kurmar, M. M. Ho, J. M. Leung, and T. Toyokuni, Adv. Synth. Catal., 344, 1146 (2002).
Y. Hagiwara, M. Tanaka, A. Kajitani, and S. Yasumoto, Jpn. Pat. Appl. 3220180; Chem. Abstr., 116, 128944s (1992).
N. Minami, M. Sato, K. Hasumi, N. Yamamoto, K. Keino, T. Matsui, A. Kaneda, S. Ota, N. Saitou, S. Sato, A. Asau, S. Doi, M. Kobayashi, J. Sato, and S. Asano, Jpn. Pat. Appl. 2000086657; Chem. Abstr., 132, 251139r (2000).
O. Dannhardt, P. Dominiak, and S. Laufer, Arzneim. Forsch., 43, 441 (1993).
P. Skolnick, J. Clin. Psychiatry, 63, 19 (2002).
A. J. Scheen and M. Malaise, Rev. Med. Liege, 59, 251 (2004).
Q. Chen, F. Liu, F. Xu, and C. Yang, J. Heterocycl. Chem., 45, 77 (2008).
R. Eskandari, L. Navidpour, M. Amini, H. Shafaroodi, and A. Shafiee, J. Heterocycl. Chem., 44, 449 (2007).
E. de Clercq, Biochem. Pharmacol., 82, 99 (2011).
V. K. Brel, Heteroat. Chem., 17, 547 (2006).
V. K. Brel, Synthesis, 2674 (2007).
V. K. Brel, Synthesis, 3405 (2009).
V. K. Brel, V. K. Belsky, A. I. Stash, V. E. Zavodnik, and P. J. Stang, Org. Biomol. Chem., 1, 4220 (2003).
V. K. Brel, Synthesis, 463 (1999).
K.-C. Liu, B. R. Shelton, and R. K. Howe, J. Org. Chem., 45, 3916 (1980).
T. Olsson, K. Stern, and S. Sundell, J. Org. Chem., 53, 2468 (1988).
A. Weissberger, E. Proskauer, J. A. Riddick, and E. E. Toops, Jr., Organic Solvents, Wiley-Interscience, New York (1955).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 977-981, June, 2012.
Rights and permissions
About this article
Cite this article
Brel’, V.K. Synthesis of 3-aryl-5-diethoxyphosphorylethyne-4,5-dihydroisoxazoles. Chem Heterocycl Comp 48, 907–911 (2012). https://doi.org/10.1007/s10593-012-1075-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-012-1075-2