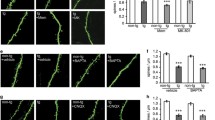
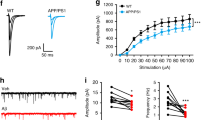
Abstract
Amyloid beta (Aβ) peptides represent one of the most studied etiological factors of Alzheimer’s disease. Nevertheless, the effects elicited by different molecular forms of amyloid beta peptides widely vary between the studies, mostly depending on experimental conditions. Despite the enormous amount of accumulated evidences concerning the pathological effects of amyloid beta peptides, the exact identity of the amyloid beta species is still controversial, and even less is clear as regards to the downstream effectors that mediate the devastating impact of these peptides on synapses in the central nervous system. Recent publications indicate that some of the neurotoxic effects of amyloid beta peptides may be mediated via the activation of proteins belonging to the Abelson non-receptor tyrosine kinase (Abl) family, that are known to regulate actin cytoskeleton structure as well as phosphorylate microtubule-associated tau protein, a hallmark of Alzheimer’s disease. By performing series of miniature excitatory postsynaptic currents (mEPSC) recordings in cultured hippocampal cells, we demonstrate that activation of Abl kinases by acute application of 42 amino acid-length monomeric amyloid beta (Aβ1-42) peptides reduces spontaneous synaptic release, while this effect can be rescued by pharmacologic inhibition of Abl kinase activity, or by reduction of Abl expression with small interfering RNAs. Our electrophysiological data are further reinforced by a subsequent biochemical analysis, showing enhanced phosphorylation of Abl kinase substrate CT10 Regulator of Kinase-homolog-Like (Crkl) upon treatment of hippocampal neurons with Aβ peptides. Thus, we conclude that Abl kinase activation may be involved in Aβ-induced weakening of synaptic transmission.








Similar content being viewed by others
Abbreviations
- Abl:
-
Abelson non-receptor tyrosine kinase
- Aβ:
-
Amyloid beta peptide
- CrkL:
-
CT10 Regulator of Kinase-homolog-Like protein
- DIV:
-
Days in vitro
- DMSO:
-
Dimethyl sulfoxide
- DPH:
-
5-(1,3-Diaryl-1H-pyrazol-4-yl) hydantoin
- ECS:
-
Extracellular recording solution
- IEIs:
-
Inter-event intervals
- mEPSC:
-
Miniature excitatory postsynaptic currents
- siRNA:
-
Small interfering RNAs
References
Abramov E, Dolev I, Fogel H, Ciccotosto GD, Ruff E, Slutsky I (2009) Amyloid-beta as a positive endogenous regulator of release probability at hippocampal synapses. Nat Neurosci 12(12):1567–1576. https://doi.org/10.1038/nn.2433
Alvarez AR, Sandoval PC, Leal NR, Castro PU, Kosik KS (2004) Activation of the neuronal c-Abl tyrosine kinase by amyloid-beta-peptide and reactive oxygen species. Neurobiol Dis 17 (2):326–336. S0969–9961(04)00135–4 Doi: 10.1016/j.nbd.2004.06.007
Bate C, Williams A (2018) Monomeric amyloid-beta reduced amyloid-beta oligomer-induced synapse damage in neuronal cultures. Neurobiol Dis 111:48–58. https://doi.org/10.1016/j.nbd.2017.12.007
Beazely MA, Weerapura M, MacDonald JF (2008) Abelson tyrosine kinase links PDGFbeta receptor activation to cytoskeletal regulation of NMDA receptors in CA1 hippocampal neurons. Mol Brain 1:20. 1756–6606–1–20 doi: 10.1186/1756–6606–1–20
Bliss JM, Gray EE, Dhaka A, O'Dell TJ, Colicelli J (2010) Fear learning and extinction are linked to neuronal plasticity through Rin1 signaling. J Neurosci Res 88(4):917–926. https://doi.org/10.1002/jnr.22252
Borovok N, Nesher E, Levin Y, Reichenstein M, Pinhasov A, Michaelevski I (2016) Dynamics of Hippocampal Protein Expression During Long-term Spatial Memory Formation. Molecular & cellular proteomics : MCP 15(2):523–541. https://doi.org/10.1074/mcp.M115.051318
Buchdunger E, Mett H, Trinks U, Regenass U, Muller M, Meyer T, Beilstein P, Wirz B, Schneider P, Traxler P et al (1995) 4,5-bis(4-fluoroanilino)phthalimide: A selective inhibitor of the epidermal growth factor receptor signal transduction pathway with potent in vivo antitumor activity. Clinical cancer research : an official journal of the American Association for Cancer Research 1(8):813–821
Cancino GI, Perez de Arce K, Castro PU, Toledo EM, von Bernhardi R, Alvarez AR (2011) c-Abl tyrosine kinase modulates tau pathology and Cdk5 phosphorylation in AD transgenic mice. Neurobiol Aging 32(7):1249–1261. https://doi.org/10.1016/j.neurobiolaging.2009.07.007
Cancino GI, Toledo EM, Leal NR, Hernandez DE, Yevenes LF, Inestrosa NC, Alvarez AR (2008) STI571 prevents apoptosis, tau phosphorylation and behavioural impairments induced by Alzheimer's beta-amyloid deposits. Brain 131(9):2425–2442. https://doi.org/10.1093/brain/awn125
Cao C, Leng Y, Liu X, Yi Y, Li P, Kufe D (2003) Catalase is regulated by ubiquitination and proteosomal degradation Role of the c-Abl and Arg tyrosine kinases. Biochemistry 42(35):10348–10353. https://doi.org/10.1021/bi035023f
Chapman PF, White GL, Jones MW, Cooper-Blacketer D, Marshall VJ, Irizarry M, Younkin L, Good MA, Bliss TV, Hyman BT, Younkin SG, Hsiao KK (1999) Impaired synaptic plasticity and learning in aged amyloid precursor protein transgenic mice. Nat Neurosci 2(3):271–276. https://doi.org/10.1038/6374
Choi BJ, Imlach WL, Jiao W, Wolfram V, Wu Y, Grbic M, Cela C, Baines RA, Nitabach MN, McCabe BD (2014) Miniature neurotransmission regulates Drosophila synaptic structural maturation. Neuron 82(3):618–634. https://doi.org/10.1016/j.neuron.2014.03.012
Cirrito JR, Yamada KA, Finn MB, Sloviter RS, Bales KR, May PC, Schoepp DD, Paul SM, Mennerick S, Holtzman DM (2005) Synaptic activity regulates interstitial fluid amyloid-beta levels in vivo. Neuron 48(6):913–922. https://doi.org/10.1016/j.neuron.2005.10.028
Colicelli J (2010) ABL tyrosine kinases: evolution of function, regulation, and specificity. Sci Signal 3(139):6. https://doi.org/10.1126/scisignal.3139re6
de Arce KP, Varela-Nallar L, Farias O, Cifuentes A, Bull P, Couch BA, Koleske AJ, Inestrosa NC, Alvarez AR (2010) Synaptic clustering of PSD-95 is regulated by c-Abl through tyrosine phosphorylation. The Journal of neuroscience : the official journal of the Society for Neuroscience 30(10):3728–3738. https://doi.org/10.1523/JNEUROSCI.2024-09.2010
Derkinderen P, Scales TM, Hanger DP, Leung KY, Byers HL, Ward MA, Lenz C, Price C, Bird IN, Perera T, Kellie S, Williamson R, Noble W, Van Etten RA, Leroy K, Brion JP, Reynolds CH, Anderton BH (2005) Tyrosine 394 is phosphorylated in Alzheimer's paired helical filament tau and in fetal tau with c-Abl as the candidate tyrosine kinase. The Journal of neuroscience : the official journal of the Society for Neuroscience 25(28):6584–6593. https://doi.org/10.1523/JNEUROSCI.1487-05.2005
Deshpande A, Kawai H, Metherate R, Glabe CG, Busciglio J (2009) A role for synaptic zinc in activity-dependent Abeta oligomer formation and accumulation at excitatory synapses. The Journal of neuroscience : the official journal of the Society for Neuroscience 29(13):4004–4015. https://doi.org/10.1523/JNEUROSCI.5980-08.2009
Dewachter I, Ris L, Croes S, Borghgraef P, Devijver H, Voets T, Nilius B, Godaux E, Van Leuven F (2008) Modulation of synaptic plasticity and Tau phosphorylation by wild-type and mutant presenilin1. Neurobiol Aging 29(5):639–652. https://doi.org/10.1016/j.neurobiolaging.2006.11.019
Forloni G, Artuso V, La Vitola P, Balducci C (2016) Oligomeropathies and pathogenesis of Alzheimer and Parkinson's diseases. Movement disorders : official journal of the Movement Disorder Society. https://doi.org/10.1002/mds.26624
Giuffrida ML, Caraci F, Pignataro B, Cataldo S, De Bona P, Bruno V, Molinaro G, Pappalardo G, Messina A, Palmigiano A, Garozzo D, Nicoletti F, Rizzarelli E, Copani A (2009) Beta-amyloid monomers are neuroprotective. The Journal of neuroscience : the official journal of the Society for Neuroscience 29(34):10582–10587. https://doi.org/10.1523/JNEUROSCI.1736-09.2009
Grochowska KM, Yuanxiang P, Bar J, Raman R, Brugal G, Sahu G, Schweizer M, Bikbaev A, Schilling S, Demuth HU, Kreutz MR (2017) Posttranslational modification impact on the mechanism by which amyloid-beta induces synaptic dysfunction. EMBO Rep 18(6):962–981. https://doi.org/10.15252/embr.201643519
Grove M, Demyanenko G, Echarri A, Zipfel PA, Quiroz ME, Rodriguiz RM, Playford M, Martensen SA, Robinson MR, Wetsel WC, Maness PF, Pendergast AM (2004) ABI2-deficient mice exhibit defective cell migration, aberrant dendritic spine morphogenesis, and deficits in learning and memory. Mol Cell Biol 24(24):10905–10922. https://doi.org/10.1128/MCB.24.24.10905-10922.2004
Guglielmotto M, Monteleone D, Piras A, Valsecchi V, Tropiano M, Ariano S, Fornaro M, Vercelli A, Puyal J, Arancio O, Tabaton M, Tamagno E (2014) Abeta1-42 monomers or oligomers have different effects on autophagy and apoptosis. Autophagy 10(10):1827–1843. https://doi.org/10.4161/auto.30001
Hernandez SE, Krishnaswami M, Miller AL, Koleske AJ (2004) How do Abl family kinases regulate cell shape and movement? Trends Cell Biol 14(1):36–44.
Hossain S, Dubielecka PM, Sikorski AF, Birge RB, Kotula L (2012) Crk and ABI1: binary molecular switches that regulate abl tyrosine kinase and signaling to the cytoskeleton. Genes & cancer 3(5–6):402–413. https://doi.org/10.1177/1947601912460051
Jacobsen JS, Wu CC, Redwine JM, Comery TA, Arias R, Bowlby M, Martone R, Morrison JH, Pangalos MN, Reinhart PH, Bloom FE (2006) Early-onset behavioral and synaptic deficits in a mouse model of Alzheimer's disease. Proc Natl Acad Sci USA 103(13):5161–5166. https://doi.org/10.1073/pnas.0600948103
Jing Z, Caltagarone J, Bowser R (2009) Altered subcellular distribution of c-Abl in Alzheimer's disease. J Alzheimers Dis 17(2):409–422. https://doi.org/10.3233/JAD-2009-1062
Kamenetz F, Tomita T, Hsieh H, Seabrook G, Borchelt D, Iwatsubo T, Sisodia S, Malinow R (2003) APP processing and synaptic function. Neuron 37(6):925–937
Kavalali ET (2015) The mechanisms and functions of spontaneous neurotransmitter release. Nat Rev Neurosci 16(1):5–16. https://doi.org/10.1038/nrn3875
Kavalali ET (2018) Spontaneous neurotransmission: A form of neural communication comes of age. J Neurosci Res 96(3):331–334. https://doi.org/10.1002/jnr.24207
Kimura S, Naito H, Segawa H, Kuroda J, Yuasa T, Sato K, Yokota A, Kamitsuji Y, Kawata E, Ashihara E, Nakaya Y, Naruoka H, Wakayama T, Nasu K, Asaki T, Niwa T, Hirabayashi K, Maekawa T (2005) NS-187, a potent and selective dual Bcr-Abl/Lyn tyrosine kinase inhibitor, is a novel agent for imatinib-resistant leukemia. Blood 106(12):3948–3954. https://doi.org/10.1182/blood-2005-06-2209
Kruh GD, King CR, Kraus MH, Popescu NC, Amsbaugh SC, McBride WO, Aaronson SA (1986) A novel human gene closely related to the abl proto-oncogene. Science 234(4783):1545–1548
Lacor PN, Buniel MC, Furlow PW, Clemente AS, Velasco PT, Wood M, Viola KL, Klein WL (2007) Abeta oligomer-induced aberrations in synapse composition, shape, and density provide a molecular basis for loss of connectivity in Alzheimer's disease. The Journal of neuroscience : the official journal of the Society for Neuroscience 27(4):796–807. https://doi.org/10.1523/JNEUROSCI.3501-06.2007
Lauren J, Gimbel DA, Nygaard HB, Gilbert JW, Strittmatter SM (2009) Cellular prion protein mediates impairment of synaptic plasticity by amyloid-beta oligomers. Nature 457(7233):1128–1132. https://doi.org/10.1038/nature07761
Lin YC, Yeckel MF, Koleske AJ (2013) Abl2/Arg controls dendritic spine and dendrite arbor stability via distinct cytoskeletal control pathways. The Journal of neuroscience : the official journal of the Society for Neuroscience 33(5):1846–1857. https://doi.org/10.1523/JNEUROSCI.4284-12.2013
Lindgren M, Hammarstrom P (2010) Amyloid oligomers: spectroscopic characterization of amyloidogenic protein states. The FEBS journal 277(6):1380–1388. https://doi.org/10.1111/j.1742-4658.2010.07571.x
Lindgren M, Sorgjerd K, Hammarstrom P (2005) Detection and characterization of aggregates, prefibrillar amyloidogenic oligomers, and protofibrils using fluorescence spectroscopy. Biophys J 88(6):4200–4212. https://doi.org/10.1529/biophysj.104.049700
Lindholm D, Pham DD, Cascone A, Eriksson O, Wennerberg K, Saarma M (2016) c-Abl Inhibitors Enable Insights into the Pathophysiology and Neuroprotection in Parkinson's Disease. Front Aging Neurosci 8:254. https://doi.org/10.3389/fnagi.2016.00254
Moresco EM, Donaldson S, Williamson A, Koleske AJ (2005) Integrin-mediated dendrite branch maintenance requires Abelson (Abl) family kinases. The Journal of neuroscience : the official journal of the Society for Neuroscience 25(26):6105–6118. https://doi.org/10.1523/JNEUROSCI.1432-05.2005
Moresco EM, Koleske AJ (2003) Regulation of neuronal morphogenesis and synaptic function by Abl family kinases. Curr Opin Neurobiol 13(5):535–544.
Moresco EM, Scheetz AJ, Bornmann WG, Koleske AJ, Fitzsimonds RM (2003) Abl family nonreceptor tyrosine kinases modulate short-term synaptic plasticity. J Neurophysiol 89(3):1678–1687. https://doi.org/10.1152/jn.00892.2002
Nimmrich V, Grimm C, Draguhn A, Barghorn S, Lehmann A, Schoemaker H, Hillen H, Gross G, Ebert U, Bruehl C (2008) Amyloid beta oligomers (A beta(1–42) globulomer) suppress spontaneous synaptic activity by inhibition of P/Q-type calcium currents. The Journal of neuroscience : the official journal of the Society for Neuroscience 28(4):788–797. https://doi.org/10.1523/JNEUROSCI.4771-07.2008
Nishimura N, Furukawa Y, Sutheesophon K, Nakamura M, Kishi K, Okuda K, Sato Y, Kano Y (2003) Suppression of ARG kinase activity by STI571 induces cell cycle arrest through up-regulation of CDK inhibitor p18/INK4c. Oncogene 22(26):4074–4082. https://doi.org/10.1038/sj.onc.1206498
Okuda K, Weisberg E, Gilliland DG, Griffin JD (2001) ARG tyrosine kinase activity is inhibited by STI571. Blood 97(8):2440–2448
Palop JJ, Chin J, Roberson ED, Wang J, Thwin MT, Bien-Ly N, Yoo J, Ho KO, Yu GQ, Kreitzer A, Finkbeiner S, Noebels JL, Mucke L (2007) Aberrant excitatory neuronal activity and compensatory remodeling of inhibitory hippocampal circuits in mouse models of Alzheimer's disease. Neuron 55(5):697–711. https://doi.org/10.1016/j.neuron.2007.07.025
Parameshwaran K, Sims C, Kanju P, Vaithianathan T, Shonesy BC, Dhanasekaran M, Bahr BA, Suppiramaniam V (2007) Amyloid beta-peptide Abeta(1–42) but not Abeta(1–40) attenuates synaptic AMPA receptor function. Synapse 61(6):367–374. https://doi.org/10.1002/syn.20386
Parihar MS, Brewer GJ (2010) Amyloid-beta as a modulator of synaptic plasticity. J Alzheimers Dis 22(3):741–763. https://doi.org/10.3233/JAD-2010-101020
Penzes P, Vanleeuwen JE (2011) Impaired regulation of synaptic actin cytoskeleton in Alzheimer's disease. Brain Res Rev 67(1–2):184–192. https://doi.org/10.1016/j.brainresrev.2011.01.003
Perkinton MS, Standen CL, Lau KF, Kesavapany S, Byers HL, Ward M, McLoughlin DM, Miller CC (2004) The c-Abl tyrosine kinase phosphorylates the Fe65 adaptor protein to stimulate Fe65/amyloid precursor protein nuclear signaling. The Journal of biological chemistry 279(21):22084–22091. https://doi.org/10.1074/jbc.M311479200
Puzzo D, Privitera L, Leznik E, Fa M, Staniszewski A, Palmeri A, Arancio O (2008) Picomolar amyloid-beta positively modulates synaptic plasticity and memory in hippocampus. The Journal of neuroscience : the official journal of the Society for Neuroscience 28(53):14537–14545. https://doi.org/10.1523/JNEUROSCI.2692-08.2008
Reichenstein M, Rehavi M, Pinhasov A (2008) Involvement of pituitary adenylate cyclase activating polypeptide (PACAP) and its receptors in the mechanism of antidepressant action. Journal of molecular neuroscience : MN 36(1–3):330–338. https://doi.org/10.1007/s12031-008-9116-0
Roder S, Danober L, Pozza MF, Lingenhoehl K, Wiederhold KH, Olpe HR (2003) Electrophysiological studies on the hippocampus and prefrontal cortex assessing the effects of amyloidosis in amyloid precursor protein 23 transgenic mice. Neuroscience 120(3):705–720
Santos FP, Kantarjian H, Cortes J, Quintas-Cardama A (2010) Bafetinib, a dual Bcr-Abl/Lyn tyrosine kinase inhibitor for the potential treatment of leukemia. Curr Opin Investig Drugs 11(12):1450–1465
Selkoe DJ (2008) Soluble oligomers of the amyloid beta-protein impair synaptic plasticity and behavior. Behav Brain Res 192(1):106–113. https://doi.org/10.1016/j.bbr.2008.02.016
Selkoe DJ, Hardy J (2016) The amyloid hypothesis of Alzheimer's disease at 25 years. EMBO molecular medicine. https://doi.org/10.15252/emmm.201606210
Sfakianos MK, Eisman A, Gourley SL, Bradley WD, Scheetz AJ, Settleman J, Taylor JR, Greer CA, Williamson A, Koleske AJ (2007) Inhibition of Rho via Arg and p190RhoGAP in the postnatal mouse hippocampus regulates dendritic spine maturation, synapse and dendrite stability, and behavior. The Journal of neuroscience : the official journal of the Society for Neuroscience 27(41):10982–10992. https://doi.org/10.1523/JNEUROSCI.0793-07.2007
Shankar GM, Walsh DM (2009) Alzheimer's disease: synaptic dysfunction and Abeta. Molecular neurodegeneration 4:48. https://doi.org/10.1186/1750-1326-4-48
Smith LM, Strittmatter SM (2017) Binding Sites for Amyloid-beta Oligomers and Synaptic Toxicity. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a024075
Teplow DB (2006) Preparation of amyloid beta-protein for structural and functional studies. Methods Enzymol 413:20–33. https://doi.org/10.1016/S0076-6879(06)13002-5
Tremblay MA, Acker CM, Davies P (2010) Tau phosphorylated at tyrosine 394 is found in Alzheimer's disease tangles and can be a product of the Abl-related kinase. Arg J Alzheimers Dis 19(2):721–733. https://doi.org/10.3233/JAD-2010-1271
Vargas LM, Leal N, Estrada LD, Gonzalez A, Serrano F, Araya K, Gysling K, Inestrosa NC, Pasquale EB, Alvarez AR (2014) EphA4 activation of c-Abl mediates synaptic loss and LTP blockade caused by amyloid-beta oligomers. PLoS ONE 9(3):e92309. https://doi.org/10.1371/journal.pone.0092309
Vazquez MC, Vargas LM, Inestrosa NC, Alvarez AR (2009) c-Abl modulates AICD dependent cellular responses: transcriptional induction and apoptosis. J Cell Physiol 220(1):136–143. https://doi.org/10.1002/jcp.21743
Walsh DM, Klyubin I, Fadeeva JV, Cullen WK, Anwyl R, Wolfe MS, Rowan MJ, Selkoe DJ (2002) Naturally secreted oligomers of amyloid beta protein potently inhibit hippocampal long-term potentiation in vivo. Nature 416(6880):535–539. https://doi.org/10.1038/416535a
Wang JY, Ledley F, Goff S, Lee R, Groner Y, Baltimore D (1984) The mouse c-abl locus: molecular cloning and characterization. Cell 36(2):349–356
Woodring PJ, Hunter T, Wang JY (2003) Regulation of F-actin-dependent processes by the Abl family of tyrosine kinases. J Cell Sci 116(Pt 13):2613–2626. https://doi.org/10.1242/jcs.00622
Yang J, Campobasso N, Biju MP, Fisher K, Pan XQ, Cottom J, Galbraith S, Ho T, Zhang H, Hong X, Ward P, Hofmann G, Siegfried B, Zappacosta F, Washio Y, Cao P, Qu J, Bertrand S, Wang DY, Head MS, Li H, Moores S, Lai Z, Johanson K, Burton G, Erickson-Miller C, Simpson G, Tummino P, Copeland RA, Oliff A (2011) Discovery and characterization of a cell-permeable, small-molecule c-Abl kinase activator that binds to the myristoyl binding site. Chem Biol 18(2):177–186. https://doi.org/10.1016/j.chembiol.2010.12.013
Zambrano N, Bruni P, Minopoli G, Mosca R, Molino D, Russo C, Schettini G, Sudol M, Russo T (2001) The beta-amyloid precursor protein APP is tyrosine-phosphorylated in cells expressing a constitutively active form of the Abl protoncogene. The Journal of biological chemistry 276(23):19787–19792. https://doi.org/10.1074/jbc.M100792200
Zou K, Gong JS, Yanagisawa K, Michikawa M (2002) A novel function of monomeric amyloid beta-protein serving as an antioxidant molecule against metal-induced oxidative damage. The Journal of neuroscience : the official journal of the Society for Neuroscience 22(12):4833–4841
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MR designed and performed electrophysiological and biochemical experiments, analyzed data, prepared figures, participated in manuscript writing, AS performed electrophysiological experiments, analyzed data, NB performed biochemical experiments, analyzed Western blot data, TB performed HPLC experiments and analyzed HPLC data, IM designed experiments, integrated data, supervised, wrote the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all the authors of the manuscript, as the corresponding author, I declare NO competing financial interests.
Research Involving Human Participants and/or Animals
This research does not include human participants. This research includes animal resources approved the IACUC of Tel Aviv University (IL-12–024) and Ariel University (IL-147–07-17).
Informed Consent
On behalf of all the authors of the manuscript, as the corresponding author, I declare that Informed consent is not applicable since no human participants were involved in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Reichenstein, M., Borovok, N., Sheinin, A. et al. Abelson Kinases Mediate the Depression of Spontaneous Synaptic Activity Induced by Amyloid Beta 1–42 Peptides. Cell Mol Neurobiol 41, 431–448 (2021). https://doi.org/10.1007/s10571-020-00858-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-00858-7