Abstract
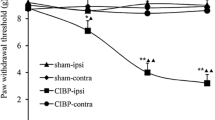
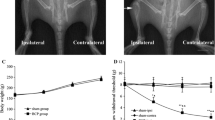
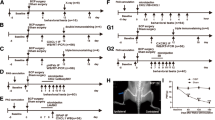
The hypernociceptive role played by the chemokine CCL2, and its main receptor, CCR2, in pathological settings is being increasingly recognized. We aimed to characterize the involvement of spinal CCL2 in the hyperalgesia due to the intratibial inoculation of fibrosarcoma NCTC 2472 cells in mice. The intrathecal (i.t.) administration of the CCR2 antagonist RS 504393 (1–3 μg) or an anti-CCL2 antibody inhibited tumoral hyperalgesia. No change in the expression of spinal CCR2 was detected by western blot, whereas immunohistochemical experiments demonstrated increased CCL2 staining at the superficial laminae of the spinal cord ipsilateral to the tumor. This spinal CCL2 does not seem to be released from nociceptors since CCL2 mRNA and CCL2 levels in DRGs, as measured by RT-PCR and ELISA, remain unmodified in tumor-bearing mice. In contrast, immunohistochemical assays demonstrated the spinal up-regulations of GFAP and Iba-1, respective markers of astroglia and microglia, and the expression of CCL2 in both types of glial cells at the superficial laminae of the spinal cord of tumor-bearing mice. Finally, since CCL2 could induce astroglial or microglial activation, we studied whether the blockade of CCR2 could inhibit the increased spinal glial expression. GFAP, but not Iba-1, up-regulation was reduced in tumor-bearing mice treated for 3 days with i.t. RS 504393, indicating that spinal CCL2 acts as an astroglial activator in this setting. The participation at spinal level of CCL2/CCR2 in tumoral hypernociception, together with its previously described involvement at periphery, makes attractive the modulation of this system for the alleviation of neoplastic pain.









Similar content being viewed by others
References
Abbadie C, Lindia JA, Cumiskey AM, Peterson LB, Mudgett JS, Bayne EK, DeMartino JA, MacIntyre DE, Forrest MJ (2003) Impaired neuropathic pain responses in mice lacking the chemokine receptor CCR2. Proc Natl Acad Sci USA 100:7947–7952
Abbadie C, Bhangoo S, De Koninck Y, Malcangio M, Melik-Parsadaniantz S, White FA (2009) Chemokines and pain mechanisms. Brain Res Rev 60:125–134
Andjelkovic AV, Song L, Dzenko KA, Cong H, Pachter JS (2002) Functional expression of CCR2 by human fetal astrocytes. J Neurosci Res 70:219–231
Baamonde A, Hidalgo A, Menéndez L (2011) Involvement of glutamate NMDA and AMPA receptors, glial cells and IL-1β in the spinal hyperalgesia evoked by the chemokine CCL2 in mice. Neurosci Lett 502:178–181
Babcock AA, Kuziel WA, Rivest S, Owens T (2003) Chemokine expression by glial cells directs leukocytes to sites of axonal injury in the CNS. J Neurosci 23:7922–7930
Banisadr G, Quéraud-Lesaux F, Boutterin MC, Pélaprat D, Zalc B, Rostène W, Haour F, Parsadaniantz SM (2002) Distribution, cellular localization and functional role of CCR2 chemokine receptors in adult rat brain. J Neurochem 81:257–269
Chehl N, Gong Q, Chipitsyna G, Aziz T, Yeo CJ, Arafat HA (2009) Angiotensin II regulates the expression of monocyte chemoattractant protein- pancreatic cancer cells. J Gastrointest Surg 13:2189–2200
Curto-Reyes V, Llames S, Hidalgo A, Menéndez L, Baamonde A (2010) Spinal and peripheral analgesic effects of the CB2 cannabinoid receptor agonist AM1241 in two models of bone cancer-induced pain. Br J Pharmacol 160:561–573
Dansereau MA, Gosselin RD, Pohl M, Pommier B, Mechighel P, Mauborgne A, Rostene W, Kitabgi P, Beaudet N, Sarret P, Melik-Parsadaniantz S (2008) Spinal CCL2 pronociceptive action is no longer effective in CCR2 receptor antagonist-treated rats. J Neurochem 106:757–769
Gao YJ, Zhang L, Samad OA, Suter MR, Yasuhiko K, Xu ZZ, Park JY, Lind AL, Ma Q, Ji RR (2009) JNK-induced MCP-1 production in spinal cord astrocytes contributes to central sensitization and neuropathic pain. J Neurosci 29:4096–4108
Gao YJ, Zhang L, Ji RR (2010) Spinal injection of TNF-α-activated astrocytes produces persistent pain symptom mechanical allodynia by releasing monocyte chemoattractant protein-1. Glia 58:1871–1880
Gosselin RD, Varela C, Banisadr G, Mechighel P, Rostene W, Kitabgi P, Melik-Parsadaniantz S (2005) Constitutive expression of CCR2 chemokine receptor and inhibition by MCP-1/CCL2 of GABA-induced currents in spinal cord neurones. J Neurochem 95:1023–1034
Hald A, Nedergaard S, Hansen RR, Ding M, Heegaard AM (2009) Differential activation of spinal cord glial cells in murine models of neuropathic and cancer pain. Eur J Pain 13:138–145
Heesen M, Tanabe S, Berman MA, Yoshizawa I, Luo Y, Kim RJ, Post TW, Gerard C, Dorf ME (1996) Mouse astrocytes respond to the chemokines MCP-1 and KC, but reverse transcriptase-polymerase chain reaction does not detect mRNA for the KC or new MCP-1 receptor. J Neurosci Res 45:382–391
Honore P, Rogers SD, Schwei MJ, Salak-Johnson JL, Luger NM, Sabino MC, Clohisy DR, Mantyh PW (2000) Murine models of inflammatory, neuropathic and cancer pain each generates a unique set of neurochemical changes in the spinal cord and sensory neurons. Neuroscience 98:585–598
Hu JH, Zheng XY, Yang JP, Wang LN, Ji FH (2012) Involvement of spinal monocyte chemoattractant protein-1 (MCP-1) in cancer-induced bone pain in rats. Neurosci Lett 17:60–63
Hu JH, Wu MY, Tao M, Yang JP (2013) Changes in protein expression and distribution of spinal CCR2 in a rat model of bone cancer pain. Brain Res 6:1–7
Ivanusic JJ (2009) Size, neurochemistry, and segmental distribution of sensory neurons innervating the rat tibia. J Comp Neurol 517:276–283
Jeon SM, Lee KM, Park ES, Jeon YH, Cho HJ (2008) Monocyte chemoattractant protein-1 immunoreactivity in sensory ganglia and hindpaw after adjuvant injection. Neuroreport 19:183–186
Jeon SM, Sung JK, Cho HJ (2011) Expression of monocyte chemoattractant protein-1 and its induction by tumor bnecrosis factor receptor sensory neurons in the ventral rhizotomy model of neuropathic pain. Neuroscience 190:354–366
Jung H, Bhangoo S, Banisadr G, Freitag C, Ren D, White FA, Miller RJ (2009) Visualization of chemokine receptor activation in transgenic mice reveals peripheral activation of CCR2 receptors in states of neuropathic pain. J Neurosci 29:8051–8062
Khasabova IA, Stucky CL, Harding-Rose C, Eikmeier L, Beitz AJ, Coicou LG, Hanson AE, Simone DA, Seybold VS (2007) Chemical interactions between fibrosarcoma cancer cells and sensory neurons contribute to cancer pain. J Neurosci 27:10289–10298
Knerlich-Lukoschus F, Juraschek M, Blömer U, Lucius R, Mehdorn HM, Held-Feindt J (2008) Force-dependent development of neuropathic central pain and time-related CCL2/CCR2 expression after graded spinal cord contusion injuries of the rat. J Neurotrauma 25:427–448
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Mao-Ying QL, Wang XW, Yang CJ, Li X, Mi WL, Wu GC, Wang YQ (2012) Robust spinal neuroinflammation mediates mechanical allodynia in Walker 256 induced bone cancer rats. Mol Brain 5:16
Menéndez L, Lastra A, Hidalgo A, Baamonde A (2002) Unilateral hot plate test: a simple and sensitive method for detecting central and peripheral hyperalgesia in mice. J Neurosci Meth 113:91–97
Menéndez L, Lastra A, Fresno MF, Llames S, Meana A, Hidalgo A, Baamonde A (2003) Initial thermal heat hypoalgesia and delayed hyperalgesia in a murine model of bone cancer pain. Brain Res 969:102–109
Miller RE, Tran PB, Das R, Ghoreishi-Haack N, Ren D, Miller RJ, Malfait AM (2012) CCR2 chemokine receptor signaling mediates pain in experimental osteoarthritis. Proc Natl Acad Sci USA 109:20602–20607
Mirzadegan T, Diehl F, Ebi B, Bhakta S, Polsky I, McCarley D, Mulkins M, Weatherhead GS, Lapierre JM, Dankwardt J, Morgans D Jr, Wilhelm R, Jarnagin K (2000) Identification of the binding site for a novel class of CCR2b chemokine receptor antagonists: binding to a common chemokine receptor motif within the helical bundle. J Biol Chem 275:25562–25571
Ogura N, Satoh K, Akutsu M, Tobe M, Kuyama K, Kuboyama N, Sakamaki H, Kujiraoka H, Kondoh T (2010) MCP-1 production in temporomandibular joint inflammation. J Dent Res 89:1117–1122
Old EA, Malcangio M (2012) Chemokine mediated neuron–glia communication and aberrant signalling in neuropathic pain states. Curr Opin Pharmacol 12:67–73
Peters CM, Eisenach JC (2010) Contribution of the chemokine (C-C motif) ligand 2 (CCL2) to mechanical hypersensitivity after surgical incision in rats. Anesthesiology 112:1250–1258
Pevida M, González-Rodríguez S, Lastra A, Hidalgo A, Menéndez L, Baamonde A (2012) CCL2 released at tumoral level contributes to the hyperalgesia evoked by intratibial inoculation of NCTC 2472 but not B16-F10 cells in mice. N-S Arch Pharmacol 385:1053–1061
Pevida M, Lastra A, Hidalgo A, Baamonde A, Menéndez L (2013) Spinal CCL2 and microglial activation are involved in paclitaxel-evoked cold hyperalgesia. Brain Res Bull 95:21–27
Preobrazhensky AA, Dragan S, Kawano T, Gavrilin MA, Gulina IV, Chakravarty L, Kolattukudy PE (2000) Monocyte chemotactic protein-1 receptor CCR2B is a glycoprotein that has tyrosine sulfation in a conserved extracellular N-terminal region. J Immunol 165:5295–5303
Quan Y, Jiang CT, Xue B, Zhu SG, Wang X (2011) High glucose stimulates TNFα and MCP-1 expression in rat microglia via ROS and NF-κB pathways. Acta Pharmacol Sin 32:188–193
Quinones MP, Kalkonde Y, Estrada CA, Jimenez F, Ramirez R, Mahimainathan L, Mummidi S, Choudhury GG, Martinez H, Adams L, Mack M, Reddick RL, Maffi S, Haralambous S, Probert L, Ahuja SK, Ahuja SS (2008) Role of astrocytes and chemokine systems in acute TNFalpha induced demyelinating syndrome: CCR2-dependent signals promote astrocyte activation and survival via NF-kappaB and Akt. Mol Cell Neurosci 37:96–109
Schiller KR, Zillhardt MR, Alley J, Borjesson DL, Beitz AJ, Mauro LJ (2009) Secretion of MCP-1 and other paracrine factors in a novel tumor-bone coculture model. BMC Cancer 9:45
Serrano A, Paré M, McIntosh F, Elmes SJ, Martino G, Jomphe C, Lessard E, Lembo PM, Vaillancourt F, Perkins MN, Cao CQ (2000) Blocking spinal CCR2 with AZ889 reversed hyperalgesia in a model of neuropathic pain. Mol Pain 6:90
Shirotake S, Miyajima A, Kosaka T, Tanaka N, Kikuchi E, Mikami S, Okada Y, Oya M (2012) Regulation of monocyte chemoattractant protein-1 through angiotensin II type 1 receptor in prostate cancer. Am J Pathol 180:1008–1016
Soria G, Ofri-Shahak M, Haas I, Yaal-Hahoshen N, Leider-Trejo L, Leibovich-Rivkin T, Weitzenfeld P, Meshel T, Shabtai E, Gutman M, Ben-Baruch A (2011) Inflammatory mediators in breast cancer: coordinated expression of TNFα & IL-1β with CCL2 & CCL5 and effects on epithelial-to-mesenchymal transition. BMC Cancer 11:130
Tanaka T, Minami M, Nakagawa T, Satoh M (2004) Enhanced production of monocyte chemoattractant protein- the dorsal root ganglia in a rat model of neuropathic pain: possible involvement in the development of neuropathic pain. Neurosci Res 48:463–469
Thacker MA, Clark AK, Bishop T, Grist J, Yip PK, Moon LD, Thompson SW, Marchand F, McMahon SB (2009) CCL2 is a key mediator of microglia activation in neuropathic pain states. Eur J Pain 13:263–272
Van Steenwinckel J, Reaux-Le Goazigo A, Pommier A, Mauborgne B, Dansereau MA, Kitabgi P, Sarret P, Pohl M, Mélik Parsadaniantz S (2011) CCL2 released from neuronal synaptic vesicles in the spinal cord is a major mediator of local inflammation and pain after peripheral nerve injury. J Neurosci 31:5865–5875
Vit JP, Ohara PT, Tien DA, Fike JR, Eikmeier L, Beitz A, Wilcox GL, Jasmin L (2006) The analgesic effect of low dose focal irradiation in a mouse model of bone cancer is associated with spinal changes in neuro-mediators of nociception. Pain 120:188–201
Wang LN, Yang JP, Zhan Y, Ji FH, Wang XY, Zuo JL, Xu QN (2012) Minocycline-induced reduction of brain-derived neurotrophic factor expression in relation to cancer-induced bone pain in rats. J Neurosci Res 90:672–681
Watkins LR, Milligan ED, Maier SF (2001) Glial activation: a driving force pathological pain. Trends Pharmacol Sci 24:450–455
White FA, Bhangoo SK, Miller RJ (2005) Chemokines: integrators of pain and inflammation. Nat Rev Drug Discov 4:834–844
Zhang J, De Koninck Y (2006) Spatial and temporal relationship between monocyte chemoattractant protein-1 expression and spinal glial activation following peripheral nerve injury. J Neurochem 97:772–783
Zhang J, Shi XQ, Echeverry S, Mogil JS, De Koninck Y, Rivest S (2007) Expression of CCR2 in both resident and bone marrow-derived microglia plays a critical role in neuropathic pain. J Neurosci 27:12396–12406
Zhang ZJ, Dong YL, Lu Y, Cao S, Zhao ZQ, Gao YJ (2012) Chemokine CCL2 and its receptor CCR2 in the medullary dorsal horn are involved in trigeminal neuropathic pain. J Neuroinflammation 9:136
Acknowledgments
Grants were provided by MEC (SAF2012-36271). M.P. is the recipient of a grant from the Instituto Universitario de Oncología (IUOPA). IUOPA is supported by Obra Social Cajastur-Asturias, Spain. The authors appreciate the excellent technical assistance given by Marta Alonso Guervós and Ángel Martínez Nistal (Servicios Científico-Técnicos, Universidad de Oviedo) for fluorescence microscopy and imaging processing.
Conflict of interest
The authors declare that they have no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pevida, M., González-Rodríguez, S., Lastra, A. et al. Involvement of Spinal Chemokine CCL2 in the Hyperalgesia Evoked by Bone Cancer in Mice: A Role for Astroglia and Microglia. Cell Mol Neurobiol 34, 143–156 (2014). https://doi.org/10.1007/s10571-013-9995-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-013-9995-7