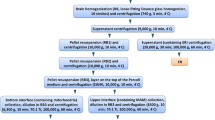
Abstract
Accumulating evidence suggests mitochondrial alterations are intimately associated with the pathogenesis of Alzheimer’s disease (AD). In order to determine if mutations of presenilin-1 (PS-1) affect levels of mitochondrial proteins at different ages we enriched mitochondrial fractions from 3-, 6-, 12-month-old knock-in mice expressing the M146V PS-1 mutation and identified, and quantified proteins using cleavable isotope-coded affinity tag labeling and two-dimensional liquid chromatography/tandem mass spectrometry (2D-LC/MS/MS). Using this approach, 165 non-redundant proteins were identified with 80 of them present in all three age groups. Specifically, at young ages (3 and 6 months), Na+/K+ ATPase and several signal transduction proteins exhibited elevated levels, but dropped dramatically at 12 months. In contrast, components of the oxidative phosporylation pathway (OXPHOS), the mitochondrial permeability transition pore (MPTP), and energy metabolism proteins remained unchanged at 3 months but significantly increased with age. We propose that alterations in calcium homeostasis induced by the PS-1 mutation have a major impact in young animals by inhibiting the function of relevant proteins and inducing compensatory changes. However, in older mice combination of the PS-1 mutation and accumulated oxidative damage results in a functional suppression of OXPHOS and MPTP proteins requiring a compensatory increase in expression levels. In contrast, signal transduction proteins showed decreased levels due to a break down in the compensatory mechanisms. The dysfunction of Na+/K+ ATPase and signal transduction proteins may induce impaired cognition and memory before neurodegeneration occurs.




Similar content being viewed by others
References
Aksenov M, Aksenova M, Butterfield DA et al (2000) Oxidative modification of creatine kinase BB in Alzheimer’s disease brain. J Neurochem 74(6):2520–2527. doi:10.1046/j.1471-4159.2000.0742520.x
Ashe KH (2001) Learning and memory in transgenic modeling Alzheimer’s disease. Learn Mem 8:301–308. doi:10.1101/lm.43701
Bailey SM, Landar A, Darley-Usmar V (2005) Mitochondrial proteomics in free radical research. Free Radic Biol Med 38:175–188. doi:10.1016/j.freeradbiomed.2004.10.011
Begley JG, Duan W, Chan S et al (1999) Altered calcium homeostasis and mitochondrial dysfunction in cortical synaptic compartments of presenilin-1 mutant mice. J Neurochem 1030–1039. doi:10.1046/j.1471-4159.1999.0721030.x
Beinert H, Kennedy MC, Stout CD (1996) Aconitase as iron-sulfur protein, enzyme, and iron-regulatory protein. Chem Rev 96(7):2335. doi:10.1021/cr950040z
Beinert H, Holm RH, Munck E (1997) Iron-sulfur clusters: nature’s modular, multipurpose structures. Science 277(5326):653–659. doi:10.1126/science.277.5326.653
Berkich DA, Ola MS, Cole J et al (2007) Mitochondrial transport proteins of the brain. J Neurosci Res 85:3367–3377. doi:10.1002/jnr.21500
Bojarski L, Herms J, Kuznicki J (2008) Calcium dysregulation in Alzheimer’s disease. Neurochem Int 52(4–5):621–633. doi:10.1016/j.neuint.2007.10.002
Boldyrev A, Bulygina E (1997) Na/K-ATPase and oxidative stress. Ann N Y Acad Sci 834:666–668. doi:10.1111/j.1749-6632.1997.tb52345.x
Brouwers B, Sleegers K, Van Broeckhoven C (2008) Molecular genetics of Alzheimer’s disease: an update. Ann Med 40(8):562–583. doi:10.1080/07853890802186905
Bubber P, Haroutunian V, Fisch G et al (2005) Mitochondrial abnormalities in Alzheimer brain: mechanistic implications. Ann Neurol 57:695–703. doi:10.1002/ana.20474
Cadenas E, Davies KJA (2000) Mitochondrial free radical generation, oxidative stress, and aging. Free Radic Biol Med 29(3–4):222–230. doi:10.1016/S0891-5849(00)00317-8
Cardoso SM, Proença MT, Santos S et al (2004) Cytochrome c oxidase is decreased in AD platelets. Neurobiol Aging 25:105–110. doi:10.1016/S0197-4580(03)00033-2
Chen CM, Lin JK, Liu SH, Lin-Shiau SY (2008) Novel Regimen through combination of memantine and tea polyphenol for neuroprotection against brain excitotoxicity. J Neurosci Res 86:2696–2704. doi:10.1002/jnr.21706
Contreras L, Gomez-Puertas P, Lijima M et al (2007) Ca2+ activation kinetics of the two aspartate-glutamate mitochondrial carriers, aralar and citrin. J Biol Chem 282(10):7098–7106. doi:10.1074/jbc.M610491200
Cortassa S, Aon MA, Marban E, Winslow RL, O’Rourke B (2003) An integrated model of cardiac mitochondrial energy metabolism and calcium dynamics. Biophys J 84:2734–2755. doi:10.1016/S0006-3495(03)75079-6
Da Cruz S, Parone PA, Martinou J-C (2005) Building the mitochondrial proteome. Expert Rev Proteomics 2(4):541–551. doi:10.1586/14789450.2.4.541
Dencher NA, Frenzel M, Reifscheider NH et al (2007) Proteome alterations in rat mitochondria caused by aging. Ann N Y Acad Sci 1100:291–298. doi:10.1196/annals.1395.030
Duff K, Eckman C, Zehr C et al (1996) Increased Amyloid-beta42(43) in brains of mice expressing mutant presenilin 1. Nature 383:710–713. doi:10.1038/383710a0
Fu H, Subramanian RR, Masters SC (2000) 14-3-3 proteins: structure, function, and regulation. Annu Rev Pharmacol Toxicol 40:617–647. doi:10.1146/annurev.pharmtox.40.1.617
Giacomello M, Drago I, Pizzo P, Pozzan T (2007) Mitochondrial Ca2+ as a key regulator of cell life and death. Cell Death Differ 14:1267–1274. doi:10.1038/sj.cdd.4402147
Green DR, Reed JC (1998) Mitochondria and apoptosis. Science 281:1309. doi:10.1126/science.281.5381.1309
Guo Q, Sopher BL, Furukawa K et al (1997) Alzheimer’s presenilin mutation sensitizes neral cells to apoptosis induced by trophic factor withdrawal and amyloid beta-peptide: involvement of calcium and oxyradicals. J Neurosci 17(11):4212–4222
Guo Q, Fu W, Sopher BL, Miller MW, Ware CB, Martin GM, Mattson MP (1999) Increased vulnerability of hippocampal neurons to excitoxic necrosis in presenilin-1 mutant knock-in mice. Nat Med 5(1):101–106. doi:10.1038/4789
Gygi SP, Rist B, Griffin TJ, Eng J, Aebersold R (2002) Proteome analysis of low-abundance proteins using multidimensional chromatography and isotope-coded affinity targets. J Proteome Res 1:47–54. doi:10.1021/pr015509n
Han DK, Eng J, Zhou H et al (2001) Quantitative profiling of differentiation-induced microsomal proteins using isotope-coded affinity tags and mass spectrometry. Nat Biotechnol 19:946–951. doi:10.1038/nbt1001-946
Hansen KC, Schmitt-Ulms G, Chalkley RJ et al (2003) Mass spectrometric analysis of protein mixtures at low levels using cleavable 13C-isotope-coded affinity tag and multidimensional chromatography. Mol Cell Proteomics 2:299–314
Hashiguchi M, Sobue K, Paudel HK (2000) 14-3-3ζ is an effector of Tau protein phosphorylation. J Biol Chem 275(33):25247–25254. doi:10.1074/jbc.M003738200
Hattori N, Kitagawa K, Higashida T et al (1998) Cl−-ATPase and Na+/K+-ATPase activities in Alzheimer’s disease brains. Neurosci Lett 254:141–144. doi:10.1016/S0304-3940(98)00654-5
Huber LA, Pfaller K, Vietor I (2003) Organelle proteomics: implications for subcellular fractionation in proteomics. Circ Res 92:962–968. doi:10.1161/01.RES.0000071748.48338.25
Karelson E, Fernaeus S, Reis K et al (2005) Stimulation of G-proteins in human control and Alzheimer’s disease brain by FAD mutants of APP714–723: implication of oxidative mechanisms. J Neurosci Res 79:368–374. doi:10.1002/jnr.20371
Keller A, Nesvizhskii AI, Kolker E, Aebersold R (2002) Empirical statistical model to estimate the accuracy of peptide identifications made by MS/MS and database search. Anal Chem 74:5383–5392. doi:10.1021/ac025747h
Kinnoshita Y, Uo T, Jayadev S et al (2006) Potential applications and limitations of Proteomics in the study of neurological disease. Arch Neurol 63:1692–1696. doi:10.1001/archneur.63.12.1692
Layfield R, Fergusson J, Aitken A et al (1996) Neurofibrillary tangles of Alzheimer’s disease brains contain 14-3-3 proteins. Neurosci Lett 209(1):57–60. doi:10.1016/0304-3940(96)12598-2
Leutner S, Czech C, Schindowski K et al (2000) Reduced antioxidant enzyme activity in brains of mice transgenic for human presenilin-1 with single or multiple mutations. Neurosci Lett 292:87–90. doi:10.1016/S0304-3940(00)01449-X
Li X, Burklen T, Yuan X et al (2006) Stabilization of ubiquitous mitochondrial creatine kinase preprotein by APP family proteins. Mol Cell Neurosci 31:263–272. doi:10.1016/j.mcn.2005.09.015
Lovell MA, Xiong S, Maresbery WR et al (2005) Quantitative proteomic analysis of mitochondria from primary neuron cultures treated with amyloid beta peptide. Neurochem Res 30(1):113–122. doi:10.1007/s11064-004-9692-5
MacCoss MJ, Wu CC, Liu H et al (2003) A correlation algorithm for the automated analysis of quantitative proteomics data. Anal Chem 75:6912–6921. doi:10.1021/ac034790h
Mattson MP (1998) Modification of ion homeostasis by lipid peroxidation: roles in neuronal degeneration and adaptive plasticity. Trends Neurosci 20:53–57. doi:10.1016/S0166-2236(97)01188-0
Mattson MP (2007) Calcium and neurodegeneration. Aging Cell 6:337–350. doi:10.1111/j.1474-9726.2007.00275.x
McCormack JG, Halestrap AP, Denton RM (1990) Role of calcium ions in regulation of mammalian intramitochondrial metabolism. Physiol Rev 70:391–425
Mootha VK, Bunkenborg J, Olsen JV et al (2003) Integrated analysis of protein composition, tissue diversity, and gene regulation in mouse mitochondria. Cell 115:629–640. doi:10.1016/S0092-8674(03)00926-7
Moreira PI, Santos MS, Oliveira CR (2007) Alzheimer’s Disease: a lesson from mitochondrial dysfunction. Antioxid Redox Signal 9(10):1621–1630. doi:10.1089/ars.2007.1703
Moseley AE, Williams MT, Schaefer TL et al (2007) Deficiency in Na, K-ATPase isoform genes alters spatial learning, motor activity, and anxiety in mice. J Neurosci 27(3):616–626. doi:10.1523/JNEUROSCI.4464-06.2007
Nelson O, Tu H, Lei T et al (2007) Familial Alzheimer’s disease-linked mutations specifically disrupt Ca2+ leak function of presenilin 1. J Clin Invest 117(5):1230–1239. doi:10.1172/JCI30447
Nesvizhskii AI, Keller A, Kolker E et al (2003) A statistical model for identifying proteins by tandem mass spectrometry. Anal Chem 75:4646–4658. doi:10.1021/ac0341261
Newman FI (1772) Musgrave, M. Lardelli (2007) Alzheimer’s disease: amyloidogenesis, the presenilins and animal models. Biochim Biophys Acta :285–297
Nicholls DG, Budd SL (2000) Mitochondria and neuronal survival. Physiol Rev 80(1):315–360
Ojaimi J, Masters CL, McLean C et al (1999) Irregular distribution of cytochrome c oxidase protein subunits in aging and AD. Ann Neurol 46:656–660. doi:10.1002/1531-8249(199910)46:4<656::AID-ANA16>3.0.CO;2-Q
Palmieri L, Pardo B, Lasorsa FM et al (2001) Citrin and aralar1 are Ca2+-stimulated aspartate/glutamate transporters in Mitochondria. EMBO J 20(18):5060–5069. doi:10.1093/emboj/20.18.5060
Papassotiropoulos A, Fountoulakis M, Dunckley T et al (2006) Genetics, transcriptomics, and proteomics of Alzheimer’s disease. J Clin Psychol 67:652–670
Pardo B, Contreras L, Serrano A et al (2006) Essential role of aralar in the transduction of small signals to neuronal mitochondria. J Biol Chem 281(2):1039–1047. doi:10.1074/jbc.M507270200
Parihar MS, Brewer GJ (2007) Mitoenergetic failure in Alzheimer disease. Am J Physiol Cell Physiol 292:C8–C23. doi:10.1152/ajpcell.00232.2006
Petrushanko N, Bogdanov E, Bulygina B et al (2006) Na-K-ATPase in rat cerebellar granule cells is redox sensitive. Am J Physiol Regul Integr Comp Physiol 290:R916–R925. doi:10.1152/ajpregu.00038.2005
Raha S, Robinson BH (2000) Mitochondria, oxygen free radicals, disease and ageing. Trends Biochem Sci 25:502–508. doi:10.1016/S0968-0004(00)01674-1
Reddy PH, McWeeney S, Park BS et al (2004) Gene expression profiles of transcripts in APP transgenic mice. Hum Mol Genet 13:1225. doi:10.1093/hmg/ddh140
Reinders J, Zahedi RP, Pfanner N et al (2006) Toward the complete yeast mitochondrial proteome: multidimensional separation techniques for mitochondrial proteomics. J Proteome Res 5:1543–1554. doi:10.1021/pr050477f
Rinholm JE, Slettalokken G, Marcaggi P et al (2007) Subcellular localization of the glutamate transporters GLAST and GLT at the neuromuscular junction in rodents. Neuroscience 145:579–591. doi:10.1016/j.neuroscience.2006.12.041
Rose AM, Valdes R Jr (1994) Understanding the sodium pump and Its relevance to disease. Clin Chem 40(9):1674–1685
Rutten BP, Van der Kolk NM, Schafer S et al (2005) Age-related loss of synaptophysin immunoreactive presynaptic boutons within the hippocampus of APP751SL, PS-1M146L, and APP751SL/PS-1M146L transgenic mice. Am J Pathol 167:161–173
Schlattner U, Dolder M, Wallimann T et al (2001) Mitochondrial Creatine Kinase and Mitochondrial Outer Membrane Porin Show a Direct Interaction That Is Modulated by Calcium. J Biol Chem 276(51):48027–48030
Schneider I, Reverse D, Dewachter I, et al (2001) J Biol Chem 276(15):11539–11544. doi:10.1074/jbc.M010977200
Schonberger SJ, Edgar PF, Kydd R et al (2001) Proteomic analysis of the brain in the Alzheimer’s disease: molecular phenotype of a complex disease process. Proteomics 1:1519–1528. doi:10.1002/1615-9861(200111)1:12<1519::AID-PROT1519>3.0.CO;2-L
Schuessel K, Frey C, Jourdan C et al (2006) Aging sensitizes toward ROS formation and lipid peroxidation in PS-1M146L transgenic mice. Free Radic Biol Med 20:850–862. doi:10.1016/j.freeradbiomed.2005.10.041
Selkoe DJ (2002) Alzheimer’s disease is a synaptic failure. Science 298:789–791. doi:10.1126/science.1074069
Selkoe DJ, Wolfe MS (2007) Presenilin: running with scissors in the membrane. Cell 131:215–221. doi:10.1016/j.cell.2007.10.012
Smine A, Xu X, Nishiyama K et al (1998) Regulation of brain G-protein Go by Alzheimer’s disease gene presenilin-1. J Biol Chem 273(26):16281–16288. doi:10.1074/jbc.273.26.16281
Spasic D, Tolia A, Dillen K et al (2006) Presenilin-1 maintains a nine-transmembrane topology throughout the secretory pathway. J Biol Chem 281(36):26569–26577. doi:10.1074/jbc.M600592200
Starkov AA, Fiskum G, Chinopoulos C et al (2004) Mitochondrial α-ketoglutarate dehydrogenase complex generates reactive oxygen species. J Neurosci 24(36):7779–7788. doi:10.1523/JNEUROSCI.1899-04.2004
Sun X, Beglopoulos V, Mattson MP, Shen J (2005) Hippocampal spatial memory impairments caused by the familial Alzheimer’s disease-linked presenilin 1M146V mutation. Neurodegener Dis 2:6–15. doi:10.1159/000086426
Suo Z, Wu M, Citron BA et al (2004) Abnormality of G-protein-coupled receptor kinases at prodromal and early stages of Alzheimer’s disease: an association with early β-amyloid accumulation. J Neurosci 24(13):3444–3452. doi:10.1523/JNEUROSCI.4856-03.2004
Swerdlow RH, Khan SM (2004) A “mitochondrial cascade hypothesis” for sporadic Alzheimer’s disease. Med Hypotheses 63:8–20M. doi:10.1016/j.mehy.2003.12.045
Tian G, Lai L, Guo H et al (2007) Translational control of glial glutamate transporter EAAT2 expression. J Biol Chem 282(3):1727–1737. doi:10.1074/jbc.M609822200
Tu H, Nelson O, Bezprozvanny A et al (2006) Presenilins form ER Ca2+ leak channels, a function disrupted by familial Alzheimer’s disease-linked mutations. Cell 126:981–993. doi:10.1016/j.cell.2006.06.059
Vlkolinsky R, Nigel Cairns N, Michael Fountoulakis M et al (2001) Decreased brain levels of 2’,3’-cyclic nucleotide-3’-phosphodiesterase in Down syndrome and Alzheimer’s disease. Neurobiol Aging 22:547–553. doi:10.1016/S0197-4580(01)00218-4
Vo TD, Palsson BO (2007) Building the power house: recent advances in mitochondrial studies through proteomics and systems biology. Am J Physiol Cell Physiol 292:C164–C177. doi:10.1152/ajpcell.00193.2006
Wang J, Xiong S, Xie C, Markesbery WR, Lovell MA (2005) Increased oxidative damage in nuclear and mitochondrial DNA in Alzheimer’s disease. J Neurochem 93:953–962. doi:10.1111/j.1471-4159.2005.03053.x
Ward M (2007) Biomarkers for Alzheimer’s disease. Expert Rev Mol Diagn 7(5):635–646. doi:10.1586/14737159.7.5.635
Xie Z, Askari A (2002) Na/K-ATPase as a signal transducer. Eur J Biochem 269:2434–2439. doi:10.1046/j.1432-1033.2002.02910.x
Yu L-R, Conrads TP, Uo T et al (2004) Global analysis of cortical neuron proteome. Mol Cell Proteomics 3:396–907. doi:10.1074/mcp.M400034-MCP200
Zoia C, Cogliati T, Tagliabue E et al (2004) Glutamate transporters in platelets: EAAT1 decrease in aging and in Alzheimer’s disease. Neurobiol Aging 25(2):149–157. doi:10.1016/S0197-4580(03)00085-X
Acknowledgment
This study was supported by National Institute of Health grant (NIH/NIA Grant # 1R01 AG25403-04). Our bioinformaics resources were supported by NCRR(NIH) Grant P 20 RR16481. The authors thank the University of Kentucky Mass Spectrometry Facility (http://www.rgs.uky.edu/ukmsf) for laboratory resources.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fu, YJ., Xiong, S., Lovell, M.A. et al. Quantitative Proteomic Analysis of Mitochondria in Aging PS-1 Transgenic Mice. Cell Mol Neurobiol 29, 649–664 (2009). https://doi.org/10.1007/s10571-009-9359-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-009-9359-5